Release Date: June 4, 2021
We are pleased to bring you the following new functionality in this week’s release. See details about feature enablement below.
Working with Documents
Video Annotation: Claim Links
With this release, users can create Claim Links when adding references on video documents by selecting approved Claims in order to define references in the resulting link annotations. This aligns with existing capability on non-video documents.
Share My Inbox Documents
You can now configure your Document Inbox to automatically share Inbox Documents where you are the Owner. This applies to both existing Inbox Documents and any new Inbox Documents you create. By clicking the Share Inbox button, you can select users and groups with whom to share your inbox.
This feature is particularly helpful where the user uploading files to the Document Inbox isn’t necessarily the user responsible for completing the document information and classifying it into the Library.
When configuring inbox sharing for specific users in Admin, Admins can define non-removable users and groups. Learn more about sharing inbox documents.
Document Inbox Auto-On for All Vaults
Document Inbox is now enabled for all vaults. This feature cannot be disabled since it is a core part of the document management functionality. However, we have introduced new ways to control who has access to its abilities, including a way to control exactly which users can create unclassified documents or see the Classify later option when uploading a new document. See the Document Inbox Enhancements release note below.
Relabel the Undefined Document Type to Unclassified
In vaults created after the 21R2 release, the Undefined document type is relabelled as Unclassified. We recommend that customers update their existing vault configurations with the new label.
Relabel the Unclassified Lifecycle to Inbox
In vaults created after the 21R2 release, the Unclassified document lifecycle is relabelled as Inbox. We recommend that customers update their existing vault configurations with the new label.
Document Inbox Enhancements
This release introduces features related to the Unclassified document type and Inbox. These enhancements affect the Document Inbox. Users must now have the Create Document permission on the Unclassified document type to view and drag and drop documents into the Document Inbox. Users without this permission without the Create Document permission can only view the Document Inbox if they have the View Document permission on a document using the Inbox lifecycle. Additionally, documents shown in the Document Inbox are now restricted to those assigned to the Inbox lifecycle.
Attachments for Unclassified Documents
Documents of the type Unclassified (previously Undefined) now include the Attachments section in the Doc Info pane. Now that the Unclassified document type is visible in Admin, you can also disable attachments on unclassified documents.
Prevent Reclassify for Checked-out Documents
A few releases back we started preventing documents from changing state whilst they are checked out. When a document is checked out, its source file is being edited, and it could cause process and security access issues if the state changes before it is checked back in. Reclassification will often affect the available document fields, and can even change the lifecycle state, so we have extended this concept to now prevent reclassification if the document is currently checked out.
Annotate UX Refresh
This enhancement adds a significant number of visual and usability improvements to annotations and document viewing in Vault. These enhancements also apply to video annotations. This feature has been postponed until a future release.
Doc Info UI Enhancements
With this release, the Doc Info panel is now divided into five panels: Information, Relationships, Files, Sharing Settings, and Timeline View. Instead of Mixed View, Content View, and Field View, users can now expand and collapse the entire Doc Info panel. Additionally, users can click and drag to resize the Doc Info panel. Vault maintains this panel width across all documents. Note that, when in Annotation mode, the Doc Info panel is set to 30% width and is not resizable. This feature has been postponed until a future release.
Vault Objects
Create Related Record in Record Create Dialog
In previous releases, users that wished to create a new record within a related record first had to create a new record if it did not already exist. Users can now create a related record directly in the create record dialog.
Hovercard on Record Picker Dialog Fields
Starting this release, users can now hover over fields on the record picker pop-up dialog, and Vault will display a hovercard with up to 1000 characters of text to help users decide which records to choose.
Lifecycle & Workflow
Read & Understood Workflows
Admins can now configure document workflows (formerly known as multi-document workflows) as Read & Understood workflows. This new method for creating and using Read & Understood workflows is only available to applications and customers with access to legacy Read & Understood workflows, and leverages the modern document workflow functionality. This includes the ability to start Read & Understood workflows for multiple documents, and when a user clicks into a Read & Understood task, they view the documents in the updated document and multi-document viewer.
Read & Understood workflows are a simplified version of workflows that support read and understood tasks. To create one, an Admin selects the Read & Understood while creating a document workflow.
Read & Understood workflows have a limited set of workflow steps. For example, Content Action steps are not available in Read & Understood workflow configuration. Read & Understood workflow tasks are always required and can only have a single verdict. Read & Understood workflows are only available as specific-lifecycle document workflows. Read & Understood workflow tasks can be made available to External and Read-Only users. Learn more about setting up Read & Understood workflows.
Display Next Workflow Start Dialog When Current Workflow Ends
Admins can now configure a workflow End step to automatically display a workflow start dialog when the current workflow ends. This feature, which was previously available for legacy workflows, is now available for object workflows and document workflows (formerly multi-document workflows).
The start dialog is shown to the last user whose action completes the workflow. Vault displays the dialog only if the user has permissions to start a workflow.
Auto-Removal of Documents from Workflows
In previous releases, documents could only be removed manually from document workflows (formerly known as multi-document workflows). With this release, Admins can now configure document workflows to automatically remove documents as part of a Content Action step.
Removed documents can remain in their current lifecycle state or change back to their start state. This action can only be configured with matching conditions. A Decision step is always recommended before the removal step to avoid situations where all documents could be removed. If every document matches the condition for removal, Vault displays an error and does not remove documents.
Document Workflow on Binders
Document workflows (formerly known as multi-document workflows) can now be started on binders. When a user starts a workflow on a binder, or a set of documents which includes a binder, the workflow addresses the binder itself and not the binder’s document contents. Note that Read & Understood workflows are not available on binders.
Unify Lifecycle Stage Group Configuration
Vault Admins can now create, access, edit and delete object and document lifecycle stage groups from one location at Admin > Configuration > Lifecycle Stage Groups.
This new page contains both document and object lifecycle stage groups. Administrators can search or filter for either type of stage group in this view.
Maximum Character Limit for Instructions
Object workflow and document workflow (formerly known as multi-document workflow) Admins can now enter a maximum of 500 characters for instructions in Start or Task steps. In previous releases, the limit was 200.
Rename Start Multi-Document Workflow to Start Document Workflow
As part of the One Workflow initiative, with this release, the Start Multi-Document Workflow action label has been changed to Start Document Workflow.
VQL for Multi-Document Workflows
This feature allows VQL developers to query for data associated with multi-document workflows. To utilize this feature, users must have the new Application: Workflow: Query permission. By default, all new and existing Vault Owners, System Administrators, and Business Administrators will have this permission.
Reporting & Dashboards
Contains Operator in Reports
This feature introduces the contains operator for report filters and prompts. Users can now filter report results based on whether the defined text can be found within another text or picklist field.
Run Report in Background
This feature provides several enhancements for large reports. Users may now run a report in the background and receive a notification when their report results are ready. Reports run in the background can execute for longer than other reports, allowing them to handle larger volumes of data.
Flash Reports Timeout Behavior
With this release, Vault automatically blocks flash reports that timeout three days in a row. Vault notifies flash report owners, who can then unblock the report by making edits to the report definition.
Reports with Multiple Down Objects in Dashboards
This feature allows users to add reports containing multiple objects with outbound references to the primary reporting object to dashboards.
Usability & UI Updates
Notifications: Support for Vault Mobile Push Notifications
This feature enables Vault Mobile to register with Vault for the user to receive mobile push notifications. Vault pushes notifications to Vault Mobile to notify users of new or pending workflow tasks. Users and Admins control whether push notifications are sent from a given Vault through a setting on the user profile. Learn more about mobile app registrations.
This feature supports a future release of Vault Mobile.
Checklists: Welcome Notification Templates
Checklist provisioning now supports welcome notification templates for each Checklist Type. The template includes tokens that reference fields on Checklist Design records, giving non-Admin designers a way to provide Checklist-specific text for welcome notifications sent to respondents.
Checklists: Duplicate Checklist Prevention
With this feature, a user can only create one checklist at a time for a given object record. This prevents users from accidentally creating a duplicate checklist while the first one is still being generated.
Localize Vault to Swedish
The Vault UI now supports Swedish. Users can update the language Vault displays for them by changing the language settings on the User Profile page. Vault also supports setting Swedish translations for labels in user-configurable data such as document types, fields, picklist values, and lifecycle names. Learn more about supported languages and localization settings.
Vault File Manager
Install Vault File Manager Without Affecting Checked-Out Files
With this release, updating Vault File Manager automatically or manually does not affect previously checked out files.
Auto-Logout of Vault File Manager After Inactivity
This feature addresses security concerns and ensures users are logged out of Vault File Manager. Vault File Manager automatically logs users out after eight (8) hours of inactivity.
Vault File Manager Automatically Opens Files
Prior to this release, customers could disable the ability for Vault to automatically open specific file types. With this release, all vaults now automatically open the file types specified in your Vault File Manager Security Policy records.
Administration
Scheduled Data Exports
With this release, admins can receive an automated daily CSV file export of their Vault object record data and document metadata directly to their Vault File Staging FTP folder or their Amazon S3 Bucket.
Feature Flag Confirmation Dialog in Vault
This feature adds a confirmation message that displays when a Vault Admin enables a feature that cannot be disabled once enabled on the Settings page.
Admins Can Edit All Picklist Labels & Picklist Value Labels
In previous releases, Admins could not edit the labels on all standard (__v) picklists and picklist values, even though this was possible via MDL and the bulk translation tools in Vault. We have removed this constraint.
Documentation Only: Limited Release Sandbox Feature Flag
In previous releases, Vault users were required to contact Veeva Support to access the Limited Release Sandbox feature. Starting in 21R1.3, Vault Admins can enable this feature in Admin > Settings > General Settings > Sandbox.
Display the Unclassified Document Type in Admin
With this release, the Unclassified document type, which is labeled as Undefined in existing vaults, now appears on the Business Admin pages. Admins can assign fields and document type groups to the Unclassified document types and can create document permissions on the document type. Note that Vault does not enforce required fields when a document remains in the Unclassified document type or the Inbox lifecycle.
Additionally, if an Admin assigns a document field to both the Unclassified document type and the Base Document type, it will no longer be directly assigned to Unclassified. All Base Document type fields will appear on the Unclassified document type, aligning it with normal document types.
Vault Java SDK
Vault Java SDK Service Account
Custom Vault Java SDK code now executes as the new Java SDK Service Account. Audit trails reflect this change, where events will be logged as java_sdk_service@{domain} on behalf of {username}. The Runtime behavior of existing code will not be impacted by this change, only the audit trail entries will be different.
This feature has been postponed to a future release and was removed from the 21R1.3 release in maintenance release 21R1.3.6 on June 17, 2021.
JSON Binding Annotations: User-Defined Model and Property
This feature introduces a new Vault Java SDK code type, User-Defined Model, which appears in Admin > Configuration > User-Defined Models.
User-defined models allow developers to serialize a model to JSON and deserialize JSON to a model. Learn more in the Vault Developer Portal release notes.
Audit Enhancements: On Behalf of User
Starting in 21R1.3, System and Application events that make data changes will be logged as System on behalf of {username}, which provides greater visibility into which user’s actions caused data to be created, updated, or deleted. These entries were previously logged simply as System. Existing audit entries will not be altered and only new audit entries will reflect this additional information.
Platform Data Model Changes
With every release, we update the data model to better support evolving needs and new feature functionality. With this release, we’ve added the following components to the Platform data model to support new features:
Added components to support the Dynamic Template to Create Requirements feature (RegulatoryOne):
- Added the following fields to the EDL (edl__v) and EDL Item (edl_item__v) objects:
- Registration Item (request__v)
- Registration Item Requirement (request_requirement__v)
- Requirement (requirement__v)
Added component to support the Share My Inbox Documents feature:
- Share Inbox Documents object (share_inbox_document__sys) is used for persisting inbox sharing rules. This object has its configuration locked down and has types enabled (user and group types). The following fields are enabled on the object:
- User(user__sys): Object reference to user object
- Shared with User (shared_with_user__sys): Object reference to user object
- Shared with Group (shared_with_group__sys): Text field storing the group name
- Locked (locked__sys): Yes / No object field
Added the new object Scheduled Data Exports (scheduled_data_exports__sys) to support the Scheduled Data Exports feature.
Added the following items to support the Checklists: Welcome Notification Templatesfeature:
- Added the following fields to the Checklist Design (checklist_design__sys) object:
- Welcome Email Subject (welcome_email_subject__sys)
- Welcome Email Text (welcome_email_text__sys)
- Welcome Notification Text(welcome_notification_text__sys)
- Added the same items as lookup fields on each Checklist runtime object and object type:
- Welcome Email Subject (welcome_email_subject__sys)
- Welcome Email Text (welcome_email_text__sys)
- Welcome Notification Text(welcome_notification_text__sys)
- Added a new notification template for each Checklist runtime object:
- Welcome: {checklist_runtime_object_label} (welcome_{checklist_runtime_object_name})
The Document Inbox Enhancements feature made the following data model updates:
- The ‘Undefined’ document type has not been automatically relabeled in existing vaults, but in all new vaults (and in Vault Help) it is now called ‘Unclassified’. We recommend customers update the label of this document type in admin.
- The ‘Unclassified’ document lifecycle has not been automatically relabeled in existing vaults, but in all new vaults (and in Vault Help) it is now called ‘Inbox’. We recommend customers update the label of this document lifecycle in admin.
- The ‘Unclassified’ document type (formerly known as ‘Undefined’) is now visible in the document types admin page, where you can edit its details, and assign document fields (it is now a child of Base Document, so will inherit all Base Document fields).
The Check Out to File Manager user permission is now called Vault File Manager.
A new Doc Info Panel MDL component is visible on the Field Layouts document admin page. This component has no functional benefit for now, but forms the basis of a future page layout engine, denoting which Panel a particular section or control belongs to.
Clinical Operations
Milestone-Specific Expected Documents
This feature was postponed to a future release.
Study Budgets
This feature adds support for tracking study-, country-, and site-level planned Budgets and Budget Categories, as well as the ability to track expenses against those Budgets and corresponding Budget Categories. Customers can now track Budget Category values on Fee Templates, Fees, and any Payable Items related to Fees with Budget Categories. Learn more about Study Budgets.
Clinical Operations & Regulatory Vault Connection: Streamlined Reuse Support
This feature selectively ignores streamlined document reuse functionality for CrossLink documents created via the Clinical Operations & RIM Vault Connection. This allows customers to leverage document reuse functionality for any document type, subtype, or classification, even documents within the scope of the Clinical Operations & RIM Vault Connection.
CDMS & Clinical Operations Vault Connection: Protocol Deviations
Protocol deviations can now be created in CDMS vaults and transferred to Clinical Operations vaults via a new integration available for the CDMS & Clinical Operations Vault Connection. This feature creates Protocol Deviation types of Issues in Clinical Operations vaults with the corresponding information from the CDMS vault.
Veeva eConsent Authoring in Clinical Operations
This feature allows users to create and maintain Veeva eConsent forms in their Clinical Operations Vault. Vault opens Veeva eConsent forms in the Veeva eConsent editor, where users can edit, save, and check them back into their vault.
Veeva eConsent Transfer with Site Connect
With this feature, Veeva Site Connect customers can exchange Veeva eConsent forms with SiteVault.
Site Connect: Additional Vault Clinical Docs Support
With this release comes support for transferring additional document types via Veeva Site Connect. Document types mapped to the following Vault Clinical Docs artifacts can now be exchanged between Clinical Operations vaults and SiteVault vaults:
- Protocol Synopsis
- Protocol Clarification
- Protocol Summary of Changes
- CRF Completion Requirements
- Contractual Agreement
- Regulatory Approval Notification
- Other Approvals
- Indemnity
- Insurance
- Clinical Study Report
New State Types for Soft Deletion
This feature adds the standard Delete Requested, Soft Delete One Version, and Soft Delete All Versions state types to all lifecycles to support workflow configurations for deletion.
Archive Study From Any State
This feature introduces a new Initiate Study Archival action that Admins can configure on any Study lifecycle state. Admins can enable this feature by inactivating the existing Initiate Study Archival action and then enabling the new Enable Study Archive from Any State setting on the Applications Settings page. Learn more about transitioning to the new Initiate Study Archival action.
Attachments for TMF Transfer
This feature allows customers on eTMF vaults to include document attachments when transferring documents via TMF Transfer. Any attachments on the documents in the source vault will now transfer to the target vault and remain as attachments on the corresponding transferred documents.
Conditional Actions on Trip Report Questions
This feature improves the entry of follow up items and Issues by allowing CRAs to easily see which Trip Report Question Responses require follow up items and Issues. For multiple-choice questions, Admins can specify which answers require users to create or link a related follow up item or Issue record. Learn more about configuring conditional actions on Trip Reports.
Add Yuzu Japanese CTN to the Application Settings Page
This feature allows Admins to enable and disable the Yuzu Enable Japanese CTN Feature from the Application Settings page. It also allows Admins to enable the Create CTN Data for Japanese Study Country feature, which generates CTN Data records for Study Site, Study Person, and Study Organization.
USN Picker Search Improvements
With this feature, Clinical Operations users can now search across multiple criteria in the USN Picker. Searched terms now match across the following columns: Site Name, USN, Country, State/Province, and Address.
Support for EU Regulatory Agency Issue Notification Tracking
This feature adds support for tracking EU Regulatory Notifications on the Issue (pdv__ctms) object. Within an Issue object record, users can specify a Notification Type (Serious Breach, Unexpected Event, or Urgent Safety Measure) and track when the notification should be reported to the EU/EEA Member States.
Clinical Operations Data Model Changes
With every release, we update the data model to better support evolving needs and new feature functionality. With this release, we’ve added and updated the following components to the Clinical data model.
Commercial
PromoMats & RIM Vault Connection
This feature creates a connection between PromoMats and RIM Submissions vaults for eCTD Compliance Package submissions to the FDA. The connection enables a seamless flow from Compliance Package generation in PromoMats and planning and tracking in RIM Submissions to the FDA through the gateway via RIM Submissions Publishing. This reduces the time required to get submissions ready for submission to the FDA. Note that the eCTD Compliance Package feature in PromoMats must be configured to use the connector. Please note the Promotional Material Type document field is now required on Post Marketing Compliance Packages by default and is visible to users without configuration if they are using the eCTD compliance package, regardless of whether or not they are using the Connection.
Note: The PromoMats & RIM Vault Connection functionality will be available in a future release. This feature supports the future functionality of the connector. We recommend waiting to configure the connection until the full functionality is released.
Modular Content: Approval Document
This feature leverages objects previously introduced to generate a document based on data in Content Modules. The generated approval document can be used to support the approval process for modules, providing standard document review features.
Support Long Text Request Details field for Medical Inquiry CRM Data Sharing
With this feature, users can map fields in the CRM Data Sharing functionality for Medical Inquiry to the long text type. Long text fields are required to share fields that are longer than a standard text field, which is limited to 1,500 characters.
Commercial Data Model Changes
With every release, we update the data model to better support evolving needs and new feature functionality. With this release, we’ve added the following components to the Commercial data model to support new features:
The following components have been added to PromoMats vaults to support the Modular Content: Approval Document feature:
- Converted the Text Element Content field on the Content Module Asset object (content_module_asset_v.text_element_content_v) from long text to rich text
- Added new Generate Approval Documents notification
- Added new Generate Approval Document user action
- Added new document type Approval Document(approval_document__v)
The following components have been added to PromoMats vaults to support the PromoMats & RIM Vault Connection feature:
- Added new object Submission (submission__v)
- Added the following new document fields:
- Actual Submitted Date (actual_submitted_date__v)
- Primary Submission (primary_submission__v)
- Submission Notes (submission_notes__v)
- Added a new object record for Promotional Material Document Type (promotional_material_document_type__v)
- Added new document type group PromoMats to RIM Connection
- Updated the shared document field Promotional Material Document Type to be required on Post Marketing Compliance Packages
- Added the new connection PromoMats to RIM (promomats_to_rim__v)
- Added the following new integrations:
- RIM Application Integration (rim_application_i__v)
- PromoMats Contact Integration (promomats_contact_i__v)
- PromoMats Compliance Package Integration (promomats_compliance_package_i__v)
- Added the following new integration points:
- RIM Application Inbound Integration Point(rim_application_iip__v)
- PromoMats Contact Outbound Integration Point (promomats_contact_oip__v)
- RIM Submission Inbound Integration Point (rim_submission_iip__v)
- PromoMats Submission Outbound Integration Point (promomats_submission_oip__v)
- PromoMats Compliance Package Outbound Integration Point (promomats_compliance_package_oip__v)
- Added the following new queue connections:
- PromoMats to RIM Outbound Queue(promomats_to_rim_outbound_queue__v)
- RIM to PromoMats Inbound Queue (rim_to_promomats_inbound_queue__v)
- Added the following new integration rules:
- RIM Submission Integration Rule (rim_submission_ir__v)
- RIM Application Integration Rule(rim_application_ir__v)
- RIM Applicant Update Integration Rule (rim_applicant_update_ir__v)
- RIM Applicant Integration Rule(rim_applicant_ir__v)
Quality
Enhanced Periodic Review for Documents
This Vault QualityDocs feature enhances the existing periodic review process by adding auto-start functionality. A new job type, Document Periodic Review, allows administrators to configure a job that identifies documents due for periodic review. When the job runs, Vault creates a Periodic Review object record for each document, and an Admin can configure a document workflow to automatically start a periodic review document workflow. This feature does not support legacy document workflows.
Once a user provides a periodic review verdict, an Admin can configure follow-up tasks to ensure that the proper steps have been taken based on the periodic review verdict. In addition, a new Associated Periodic Review section is available on Document Change Control (DCC), allowing users to add associated periodic reviews to a DCC for documents that are part of a DCC. This additional section provides full visibility of the periodic reviews that may have originated the need for a DCC. Learn more about setting up enhanced periodic reviews.
Process Navigator: Navigation Drawer and Favorites
This Vault QualityDocs feature enhances the Process Navigator with the following additions:
- The Navigation Drawer allows an end user to navigate process hierarchies using a left-hand navigation drawer. Instead of clicking through into each process hierarchy individually, the navigation drawer allows users to expand available hierarchies and jump directly to a specific process without having to traverse the entire tree. Users can navigate to different hierarchies of the same type using the navigation drawer without returning to the Process Navigator landing page.
- The Favorites capability allows users to favorite hierarchy records and documents by clicking on the Favorites star within the Process Navigator user interface. This allows users quick access to favorited hierarchy records and documents. Favorited hierarchy records and documents are displayed and searchable by clicking on the Favorites tab when the navigation drawer is expanded. Clicking on a favorited hierarchy record will take the user to the detail page of the record, while clicking on a favorited document will open the document in a pop-out viewer.
Learn more about using the Process Navigator.
On the Job Training Type
This Vault Training feature allows customers to electronically track qualification, certification and other skill or task-based assignments associated with On the Job Training (OJT) where an evaluator must verify the learner’s ability in a given area.
A checklist captures the hands-on learning actions and performance outcomes for a defined job position and task. As part of the evaluation process, the checklist captures, in real time, the step-by-step process followed, comments, and a pass/fail outcome. The checklist is then stored as part of a Learner’s Training Assignment.
Assign Effective Version for Training
With this Vault Training feature, if a Training Requirement references an Issued document, Vault Training can also assign an additional Training Assignment to the Learner containing the Effective version of the document. This allows the Learner to be trained on the Effective version of the document, in addition to the Issued version. Learn more about configuring Vault Document Training Requirements.
Allow Learner Choice For Substitute Training
This Vault Training feature allows a Learner to choose an appropriate substitute training from a list of available training requirements. A Training Admin can configure Substitute Training Rules with an option to allow the Learner to make a substitute selection. Once configured, the Learner has the option of selecting a substitute training or continuing with the primary training assignment. Learn more about configuring Learner choice for substitute training.
Generate Quality Document from Object Record Action
This Vault QMS feature allows administrators to configure User Actions or Entry Actions which create a document based on a Quality Event or Audit record. Documents created in this way are created from Formatted Output templates, allowing complex configurations and data from multiple related records to be brought into the generated document. Documents created by this function inherit Vault Document metadata from the starting record, are saved into the Document Library, and are linked to the record from which they were generated. This powerful feature enables automation for the generation of key artifacts such as Audit introduction letters, Audit summaries or Inspection copies for completed or closed Quality Events. Once created in the Library, these documents can be configured to support lifecycles or workflows for reviews or approvals, allowing robust processes to be linked together between your process records and generated artifacts.
Since quality document artifacts can include large amounts of data from many records, this operation can take some time to complete. We encourage careful consideration of where in your processes’ lifecycles to invoke or allow these actions.
This first release supports document creation via Formatted Output templates for Quality Events and Audit objects, and we’ll be investigating additional use cases in future releases. Learn more about setting up Quality document generation.
Relationship Automation for Quality Objects: Quality Events, Audits, CAPAs
With this Vault QMS feature, Vault Admins have a new tool to configure certain complex relationship objects to be automatically bi-directional once established. This feature is targeted at relationship objects which link two records of the same object together, like the Related Quality Event object in QMS. Additionally for these types of relationship objects, this feature allows such relationships to describe the nature in which the two records are linked. This allows for simple relationships, in which no nature is captured, or described relationships which capture the linking nature. This streamlines the way in which users identify related records, reduces the amount of effort required to link records of the same object together, and means that there is one page section to look at when reviewing all links between such records.
Sometimes links between records of the same type can contain information about how the two records are related. If the new Relationship Natures picklist field is used on Related Quality Event or Related Audit records, Vault allows Admins the ability to map “opposites” to describe the directionality of a relationship. For example, if a user wishes to link a deviation record DEV-0023 to DEV-9942 because investigation of DEV-0023 discovered DEV-9942, Admins could add values to the Relationship Natures picklist such as Testing Discovered and Discovered While Testing, configuring Vault to recognize these as opposite values. Then, when a user creates a new Related Quality Event record to link DEV-0023 to DEV-9942, and selects Testing Discovered as the nature of that link, Vault automatically creates an additional Related Quality Event record with a relationship nature indicating that DEV-9942 was Discovered While Testing DEV-0023. Any user who views either DEV-0023 or DEV-9942 would be able to understand that the two deviations are linked and how they are linked by looking at the same related record section on each. In previous releases, approximating this behavior required two page layout sections on Deviations, one showing inbound relationships, and one showing outbound relationships, as well as manually creating two Related Quality Event records to indicate both directions of the link.
This feature requires configuration to be used. This feature also introduces a new standard object called Related CAPA (related_capa__v). With this first release, the Related Quality Event, Related Audit standard objects, as well as the new standard object Related CAPA are supported. Learn more about setting up Quality Relationship Automation configurations.
Batch Release Process Support
In this release, Vault QMS customers will notice new objects and related components available in their vaults to support a GMP Batch Release / Disposition process. Vault provides objects which can be configured to allow batches to have associated disposition assessments, acting as a workspace to identify, collect and track content and tasks needed to arrive at a GMP disposition for a given lot or batch of product.
This initial release introduces the object model which can be configured and extended to support these business processes. We’ll be looking to further expand upon this area in future releases.
Reportability Assessment Management
This Vault Product Surveillance feature allows Admins to set up questions to determine the Severity of a complaint and thus determine the Reportability of the Adverse Event to health authorities. The Severity and Reportability are determined based on the answers provided by Complaint handling users. This feature also automates various scenarios of Reassessment by automatically creating, updating, or canceling Reportable Event records when the Severity of the incident changes.
Adverse Event Reporting Enhancements
This release brings the following enhancements to Adverse Event Reporting in Vault Product Surveillance:
- Follow-up submissions: Users can generate follow-up reports if new information comes to light after an initial report has already been submitted to the health authority.
- On-demand validation: Users can perform on-demand validation for a Reportable Event against the validation format of the given health authority.
- Notification for truncated fields:With this feature, users are notified if there is any truncation of information when it is compiled from reference objects while generating the Reportable Event XML/PDF.
Quality Data Model Changes
With every release, we update the data model to better support evolving needs and new feature functionality. With this release, we’ve added and updated the following components to the Quality data model.
Regulatory
Copy Into Content Plans
This feature allows users to copy a content plan section or content plan item from one content plan to another. When enabled, users working in the Content Plan Hierarchy Viewer can drag and drop a Content Plan or Content Plan Item record to another content plan when that content plan is open in the viewer in a separate browser window.
When users drag and drop a Content Plan Item record, Vault copies only that record to the target location and performs the copy automatically. When users drag and drop a Content Plan record, Vault copies that section, including its descendant Content Plan and Content Plan Item records and any missing submission join records, to the target location asynchronously. When the asynchronous copy is complete, Vault sends the user a notification with results. Learn more about copying into content plans.
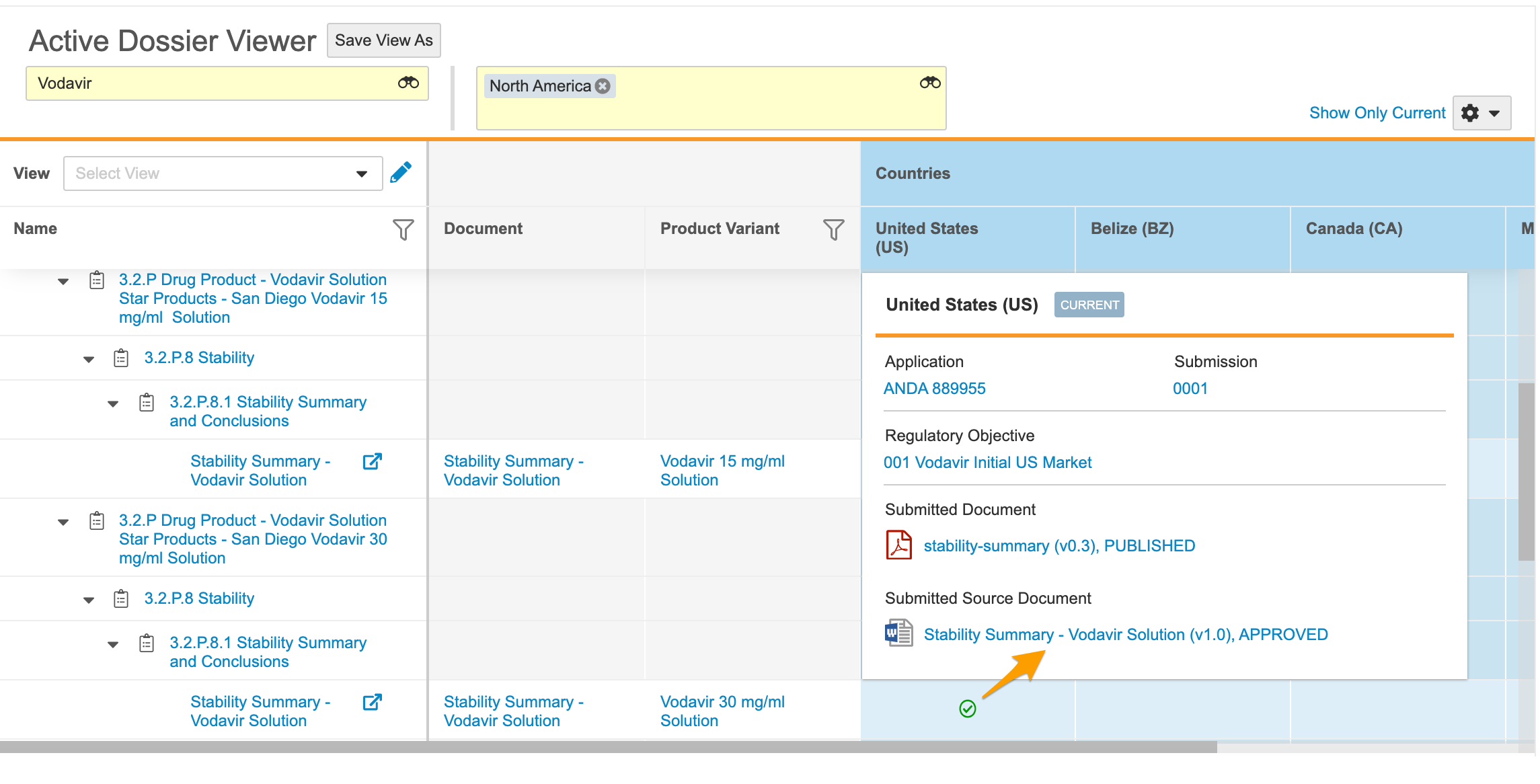
Active Dossier
RIM customers with RIM Submissions and Submissions Archive can configure Vault to track and manage documents that are active for a product in a given market. Leveraging the Submission and Regulatory Objective object lifecycles, Vault can populate the Active Dossier and subsequently calculate which documents are currently active based on the eCTD lifecycle or document versions. From the Submission or Regulatory Objective records, users can view or edit the Active Dossier with contextual filters applied based on the record’s relationships. Users can also view the Active Dossier directly with Product Family and Region filtering applied. Learn more about viewing and editing the Active Dossier.
RIM Reference Model
This release introduces a standard hierarchy reference model for RIM document types. Two new objects, RIM Reference Model and RIM Reference Model Mapping, are available with Vault-provisioned records. The RIM Reference Model Mapping object is used for the creation of CrossLink documents in RIM by the inbound Vault Clinical Operations connection and for the mapping of document types to the Active Dossier. Learn more about setting up the RIM reference model.
Auto-Create Commitment Application Joins
This release introduces a new setting on the Application Settings page that enables Vault to create Commitment Application join object records automatically when users create or update the Related Application field on Commitment records and click Save. Learn more about automatic record creation in RIM vaults.
Sequential Pagination of TOC
For Report Level Content Plans, users can now generate a Table of Contents (TOC) using a new user action. Vault generates the TOC utilizing Publishing Elements records configured by an Admin. Additionally, users can now create a TOC with overall page numbering when merging and publishing the Report Level Content Plan. Learn more about configuring and using TOC publishing for report level content plans.
Extend Content Plan Duplicate Submission Join Detection
This feature enhances the submission join duplicate detection that Vault uses in the Copy Content Plan action. Vault uses this duplicate detection to determine whether to use an existing submission join record on the target submission rather than copying the join record from the source submission. The duplicate detection now considers the XML fields on the submission join records.
Dynamically Adjust Content Plan Row Height to Fit Text
In the Content Plan Hierarchy, the row height for Content Plan rows now adjust dynamically to fit text so that cell text is never cut off vertically when wrapped. As a result, users can no longer manually adjust row height, since Vault will resize the row height automatically.
Submissions Archive Documents Created as System
With this release, users no longer need Create permission for binders and documents on the Submissions Archive document type. The document audit trail reflects the creation by the System user, and Vault assigns the document Owner role to the user who initiated the import.
Propagate Fields from Source to Archived Documents
With this release, Vault can populate any fields configured within a RIM Submissions Archive vault when a submission is published as long as the source document contains fields with the same namespace.
Archived Document Security in RIM Submissions
With this release, Vault does not include archived documents in published outputs as part of the Submissions Publishing or Report Level Content Plan Publishing processes.
US FDA eCTD Validation Criteria v3.9 Support
With this release, RIM Submissions Publishing vaults now supports the US FDA eCTD Validation Criteria v3.9.
Registration Verification Improvements
This feature provides new configuration options to enhance the registration verification workflow process. Admins can choose how to allocate workflow tasks and in which states the verification workflow should run. Additionally, Vault can now support verification when users edit Registration records or registered details directly. Previously, Vault only supported registration verification after users completed work in the Manage Registered Details wizard. Learn more about registration verification.
Activity Country Dependencies
This feature allows Admins to define country-to-country dependencies for planned changes. When Activity records are created, Vault will automatically create the applicable dependency records to ensure that the country dependencies are visible to country-level users. When a reference country activity reaches its approval or submission milestone, the owners of the dependent country activity will be notified that they can proceed with implementing the change in their market. Learn more about configuring country dependencies.
IDMP Process Automation
This feature introduces automation into the IDMP process. When users generate IDMP data from a Regulatory Objective record, Vault identifies applicable medicinal products, creates the product data submission for each applicable medicinal product, and generates the IDMP elements.
Learn more about about IDMP configuration and generating IDMP data.
Updates to IDMP Accelerators to Support UDI
This feature splits the Automatically Generate Registered Details Records setting on the Application Settings page into two separate settings for Registered Site Role records and Registered Packaged Product records. Vault now generates Registered Packaged Product records with the same object type as their source Registered Packaging records if there is a matching object type name. These changes will allow Vault to create Registered Packaged Medicinal Product records for IDMP and Registered Packaged Device Product records for UDI.
XEVMPD Attachment-Only Submission
This feature allows for the creation of attachment-only XEVMPD submissions. When the attachment-only submission is generated, Vault automatically submits it to the EMA. The EMA can then assign an EV Code to the document in the attachment-only submission, which can be used in subsequent product submissions.
Learn more about enabling and using attachment-only submissions.
XEVMPD Procedure Number
This feature updates the XEVMPD data aggregation algorithm to populate the MRP/DCP/EMA Number from the IDMP Procedure Number field on the Registration record.
21R2 RIM Data Model Changes
With every release, we update the data model to better support evolving needs and new feature functionality. With this release, we’ve added and updated the following components to the Regulatory data model.
In this release, we have also enabled the Required attribute on additional standard objects and fields. This allows organizations to configure these components according to their business requirements without needing to contact Veeva Support or Product Management to make these configuration changes. In order to utilize these newly-enabled attributes, Admins must perform the desired configuration to make them available for users.
Safety
Safety features are targeted for tentative availability on June 11, 2021.
E2B R2 and R3 Import to Inbox Item Configuration
Vault Safety now supports the ability to import single-case XML files in E2B(R2) and (R3) formats to Inbox Items. The system performs an E2B import upon receiving an AS2 Gateway transmission, or when a user runs the import action from an E2B document in the vault library. The Inbox Item displays the mapped E2B source data for the Case, Patient, Reporter, Adverse Events, Case Products, and Medical and Drug History in the Source Data pane. This feature also benefits from Vault Safety’s existing import to AER capabilities, including Study and Product matching, data mapping, and literature and attachments import.
Note: As of 21R2, AERs will enter a sunset period. No new functionality will be added to AERs. Using the new Inbox (Inbox Item) is optional in 21R2 (August 2021), recommended in 21R3 (December 2021), and mandatory in 22R1 (April 2022), when AERS will become obsolete.
Note: This feature is auto-on for Safety.AI customers who are already using the Create Inbox Item action released in 21R1 for form intake.
Learn More:
- Case Intake Overview: Case Intake Using an Inbox Item
- Import an Inbox Item
- E2B Case Import Data Mapping
Provision Additional EMA Supported Units of Measurement Auto-on
Vault Safety now includes additional units of measurement supported by EMA E2B(R3).
Learn More: About the Case Page: Units of Measurement
Default Sender User by Destination Configuration
Administrators can now configure a default sender user on all Transmission Profiles to be populated on system generated transmissions. For example, this allows defaulting different QPPVs for EMA and MHRA.
Learn More:
- Configure FDA Gateway: Configure FDA Transmission Profiles
- Configure EMA Gateway: Configure EMA Transmission Profiles
- Configure Health Canada Gateway: Configure Health Canada Transmission Profiles
- Configure MHRA Gateway: Configure the MHRA Transmission Profile
- Manage Transmission Profiles: Create Transmission Profiles
DSUR - Extended Definition of Death Auto-on
In Vault Safety, the List of Subjects Who Died During the Reporting Period Appendix now lists only the latest version of a fatal case received or approved during the reporting period, based on the value selected in the DSUR “Filter Case by” field. The appendix table constraints have also been updated to consider additional criteria for more accurate fatal case identification.
Note: This feature is auto-on for customers who have DSUR appendices enabled. Previously generated DSURs need to be regenerated to see the update.
Learn More: DSUR Table Data Mapping: List of Subjects Who Died During the Reporting Period
Feature Enablement Changes
Note the following features with enablement changes in this release:
| Feature | Previous Enablement | New Enablement (21R1.3/21R2) |
|---|---|---|
| Configurable Back Reporting | Support (21R1) | Auto-On |
| Multi-Case E2B Import via API/AS2 | Support (21R1) | Auto-On |
| VAERS Certification Enhancements | Support (21R1) | Auto-On |
| Multilingual MedDRA Browser and Autocode | Auto-On (21R1.2) | Config |
Deprecate Create AER Action Auto-on
The Create AER user action was replaced in 20R2 by Create Case to allow users to promote an Inbox Item directly to Case without creating an AER record. The Create AER action is being deprecated in 21R2.
SiteVault
Set Context in Vault Selector
With this feature, all SiteVault users will see their research organization and sites in the Vault Selector. Users with appropriate permissions can select a research organization to view data across all sites associated with the research organization.
Single Source of Truth for Person Profiles
This feature ensures that each person in a research organization only needs to manage one set of profile documents no matter how many sites they are associated with in SiteVault.
Prompt for Fields on Upload to Document Inbox
When uploading documents to the Document Inbox, users are now prompted to add values for common fields such as Study, Participant, and Organization.
Expanded Language Support
This feature provides additional support for users to view a translated version of SiteVault. See the About Supported Languages page for more information on the languages that are available.
Optional State Change for Send Document via Clinical Network Action
With this feature, Admins can configure an automatic document state change when a document is sent to a sponsor or CRO for a Connected Study.
New Exchangeable Document Types on Connected Studies
This feature adds support to transfer the following new document types from a sponsor or CRO’s vault for a Connected Study:
-
Ancillary Committee Response
-
Clinical Study Report
-
Contract
-
Insurance
-
Protocol Clarification
-
Procedure Manual
-
Protocol Summary of Changes
-
Protocol Synopsis
-
Regulatory Authority Response
Veeva eConsent: Patient Declines Consent
With this feature, sites using Veeva eConsent can receive a notification in SiteVault when a participant declines an eConsent form in MyVeeva for Patients. Moreover, site users can cancel a declined form or resend the form if the participant declined in error or changed their mind.
Veeva eConsent: Form Responses
This feature enables sites using Veeva eConsent to view, search, filter, and report on eConsent form responses collected from participants in MyVeeva for Patients during the eConsent process.
Veeva eConsent: Shareable eConsent Preview
This feature enables sites using Veeva eConsent to create a URL of an eConsent form. This enables users to share a preview of the eConsent as it would appear to a participant during the consent process with an external third party such as an internal review board (IRB).
SiteVault Data Model Updates
With every release, we update the SiteVault data model to better support evolving needs and new feature functionality. With this release, we’ve added the components below to support new features.
QualityOne
Training Management Phase 3
With the 21R1.3 release, we’ve improved QualityOne Training Management in the following ways:
- External Training Type:This release introduces the External Training type, which allows Learners to complete training on content or topics that occurred outside of Vault. For example, an organization may require that Learners complete a third-party online course or receive an external certification.
- Manager Access to Training Records: In many organizations, managers are responsible for the training completion status of the employees that report to them. This release introduces the ability for a Learner’s direct manager to have visibility into Learners’ Training Assignment records, as well as other training-related records. The Manager Groups feature must be enabled before configuring this feature in Training Management.
- Improvements to Learner Task Page: The Learner Task Page user interface is enhanced for Training Assignments containing multiple documents. Learners will arrive at a default landing page, requiring them to select a document. Vault now always displays the Learner Task Page for completed Training Assignments, along with a link to View Quiz.
- Direct Assignment to Multiple Learners: In previous releases, the Training Direct Assignment feature could issue a Training Assignment to only one Learner at a time. With this release, Direct Assignment now supports issuing a Direct Assignment Training Assignment to multiple Learners with a single request.
- Direct Assignment to Curriculum: With this release, the Direct Assignment action can also be performed on a Curriculum. This allows users to issue Training Assignments for that Curriculum to the Learners selected.
- Complete Training Assignment Workflow Visible in Admin: Previously, the Complete Training Assignment Workflow, which is used for Vault Document Training Assignment, was hidden in Vault Admin. In this release, the workflow is now visible. This allows Notification and Task Reminders to be configured for the workflow. Note: Modifying this workflow can cause issues delivering Training to Learners. Only certain workflow elements can be updated.
- Person Object - Remove User Restrictions When Adding a Learner Role: In previous releases, when a Learner Role was added to a Person record, Vault required an active User reference, regardless of the value of the Training Eligibility field. With this release, an active User reference is only required if Training Eligibility is set to Eligible.
- Training Requirement Object Type Limitation: To ensure that the Training feature continues to function as intended as we continue to add new features, Training Requirement record’s object type cannot be changed once the record is saved.
- Training Requirement ZIP File Limitation: To ensure Training Requirements function as intended, ZIP files cannot be added to the Related Training Materials page section for Training Requirements, regardless of object type.
- E-Learning Technology Standards Support: Many organizations include e-learning content in their training implementations. This format of training content can be dynamic, engaging, and easier for learners to absorb than other formats. With this Training Management release, we have introduced support for the e-learning technology standards AICC and SCORM. Content published in AICC or SCORM can be uploaded into a vault, just like any other document. Once uploaded into the Vault, training admins can associate the content to a Training Requirement as part of a training matrix.
- Substitute Training: This feature enables the Training feature to assign an alternative, or substitute, Training Assignment to a Learner in place of a required Training Assignment using Training Admin-defined substitution rules. When the Learner completes the Substitute Training Assignment, the Learner automatically gets completion credit for the required Training Assignment.
- Facilitated Training:This feature enables a Training Admin, or a user with proper access, to give completion credit for Training Assignments on behalf of Learners. This allows completion of Training Assignments for Learners that may not have access to Vault. For example, in some organizations, users could be contractors, external employees, temporary employees, or otherwise outside the primary organization.
- Training Nightly Job Performance & Robustness Improvements:This feature improves the performance and robustness of the Update Training Assignments job which drives Training Management automation. With this release, the job now continues to issue Training Assignment records even if an issue is encountered, skipping paths which have issues.
- Power Delete Action for the Training Assignment Object: During Training configuration and testing, you may need to delete a Training Assignment record and all of its related records. By default, you must delete each record individually. With this release, we have introduced the Power Delete Training Assignment Records record action, which completely deletes a Training Assignment record and its related records.
- Assignment Details Lifecycle State No Longer Updated: To improve accurate reporting and searching, the Assignment Details lifecycle state is no longer updated. In previous releases, the Assignment Details lifecycle state changed when the Training Assignment entered the Assigned, Completed, or Cancelled states. With this release, there is now a lookup field on Assignment Details records that references the parent Training Assignment record’s lifecycle state. This provides a real-time indication of the Training Assignment lifecycle state. The Assignment Details lifecycle state is no longer in sync with the Training Assignment’s lifecycle state.
- Classroom Training: Usability Enhancements: With this release, Classroom Training Assignments now have an associated auto-start workflow called View Classroom Training Assignment that provides easy access to the Classroom Training Assignment record for Learners. Once an Admin activates the View Classroom Training Assignment workflow, it is automatically started when a Classroom Training Assignment enters the Assigned state. When an Instructor changes a Classroom Training Assignment record’s state to Completed, the open Training Assignment workflow is automatically canceled with a configurable entry action.
- Training Assignment Lifecycle User Actions: With this release, Admins can now edit user actions on the Training Assignment Lifecycle. This feature is only intended to support the Download Formatted Output user action.
- Updated Training Requirement Document Picker Dialog: The dialog box when selecting documents in a Training Requirement or Training Requirement Impact Assessment has been updated to match the checkbox-style document picker.
- Learner Homepage: This release introduces the Learner Homepage, an intuitive, easy-to-use dashboard that provides the Learner with an overview of the Learner’s open assignments. Assignment cards display key information such as the related Curriculum, estimated completion time, due date, categories, and more. In addition, the Learner can access their completed Training Assignments.
- Mobile Training Task Completion: This release allows Learners to view and complete Vault Document Training Assignments from mobile device browsers. When a Learner accesses a Vault Document Training Assignment from a phone or a tablet browser, Vault displays the assignment in a mobile-friendly way that’s appropriate based on the device size. In the mobile interface, the Learner can view the document or documents, scroll through the document pages, view key document metadata, and complete the workflow task with e-signature from their mobile device.
- Hide Training Report from Document Actions Menu: This feature hides the Training Report from the document Actions menu for customers who do not utilize Read & Understood functionality in QualityOne vaults. This feature is enabled in Admin > Settings > Application Settings, and the Training Report can be made available again through the document Actions menu if a customer later chooses to utilize Read & Understood capabilities.
Learn more about Training Management features.
Inspection & NCR Product Model Compatibility
This feature ensures that customers using the new Product Hierarchy data model from 21R1 are able to use Inspection and Mobile NCR functionality.
HACCP Data Model
This feature offers an additional data model which allows organizations to manage Hazard Analysis and Critical Control Points (HACCP) to ensure food safety from biological, chemical, and physical hazards during production. Learn more about HACCP Management.
Note: This feature is currently available only to early adopters. Contact your Customer Success Manager for more information.
Incident Management - Track Labor Information
This feature extends the existing Incident Management object model with a new Labor Information object that enables tracking of labor hours and headcount for employees and contractors.
Note: This feature is currently available only to early adopters. Contact your Customer Success Manager for more information.
COA Textract Response File Download
This feature is an enhancement to the location-based header matching rule introduced in 21R1. Business Admins can create location-based header matching rules easily by removing dependencies of extracting the OCR response from AWS Textract console and storing the response in Vault. Once the response is stored in Vault, the file can be downloaded for OCR response extraction. Learn more about COA Textract Response File Download.
Note: This feature is currently available only to early adopters. Contact your Customer Success Manager for more information.
Support Localized Number Separator for COA
This COA enhancement extends the support to ingest various types of decimal and grouping separators found in a COA document such as period, comma, and single quotation. For example, Vault now identifies these number formats - “123.456”, “123,456”, “123’456”.
Note: This feature is currently available only to early adopters. Contact your Customer Success Manager for more information.
RegulatoryOne
Note that RegulatoryOne features are targeted for tentative availability on June 15th.
Regulatory Request Management
When configured, this feature provides the users with the ability to mitigate non-compliance risks by accessing accurate, up to date, and consistent information to include in responses to requests for information related to the regulatory compliance of a product or formulation. Requests can be external or internal. For each request, users can quickly create a request from predefined types and include relevant details, and then submit for review. Regulatory users can provide responses, associate related documents, view similar requests to ensure consistency in responses, and create any required secondary requests. Learn more about Regulatory Request Management.
Dynamic Template to Create Requirements
When configured, this feature provides users with the ability to automatically create a list of registration item requirements from an Admin-defined template. Requirements can be dynamic in the sense that Admins can configure criteria to include or exclude a requirement based on information specific to what is being registered. Admins can define tokens that describe a registration item’s type of information and leverage those tokens in requirement records. This feature provides Regulatory users with an efficient way to define and track what needs to be completed for a registration request. Learn more about configuring a Dynamic Template to Create Requirements.
Veeva Claims
Note that Veeva Claims features are targeted for tentative availability on June 15th.
Selective Creation of Local Adaptations from a Project
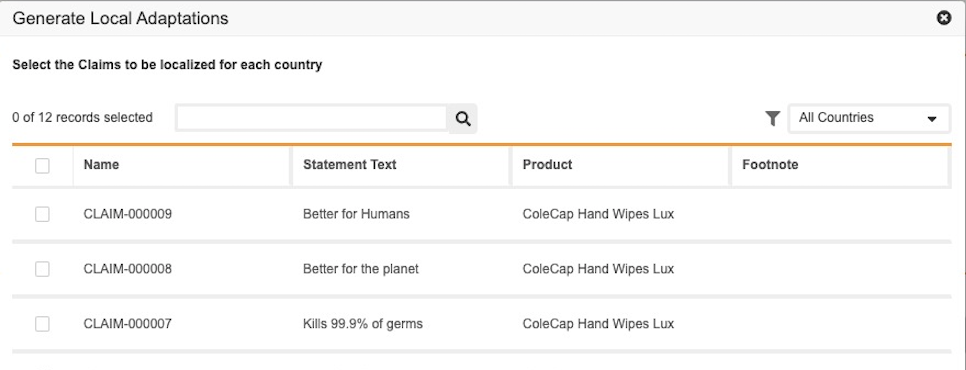
The feature allows users to create multiple Local Adaptations for the Claims and Countries of their choice by enabling them to selectively control which countries to automatically create local adaptations for through a new user action.
This feature reduces extra clicks caused by unwanted Local Adaptations generated when users can’t specify which Claim and Country combinations to create Local Adaptations for. When configured, users can now view the new action on the Project record(s) if there is at least one Claim and one Country in the project. After clicking the action, users can select the Claims under the Project in a new dialog for All or specific countries. After users complete their selection, clicking Continue creates new Local Adaptations for the Claims and Countries selected and links them to the existing Project. If Local Adaptations already exist for any of the Claims, the system will not generate duplicate records but will link the existing Local Adaptation record to the Project. Learn more about Selective Creation of Local Adaptations from a Project.
Enablement Details
| Name | Enablement | Application |
|---|---|---|
| Working with Documents | ||
| Video Annotation: Claim Links | Auto-on | Platform |
| Document Inbox Auto-On for All Vaults | Auto-on | Platform |
| Relabel the Undefined Document Type to Unclassified | Auto-on | Platform |
| Relabel the Unclassified Lifecycle to Inbox | Auto-on | Platform |
| Inbox Enhancements | Auto-on | Platform |
| Share My Inbox Documents | Auto-on | Platform |
| Attachments for Unclassified Documents | Auto-on | Platform |
| Prevent Reclassify for Checked-out Documents | Auto-on | Platform |
| Vault Objects | ||
| Create Related Record in Record Create Dialog | Auto-on | Platform |
| Hovercard on Record Picker Dialog Fields | Auto-on | Platform |
| Lifecycle & Workflow | ||
| Read & Understood Workflows | Configuration | Platform |
| Display Next Workflow Start Dialog When Current Workflow Ends | Configuration | Platform |
| Auto-Removal of Documents from Workflows | Configuration | Platform |
| Document Workflow on Binders | Auto-on | Platform |
| Unify Lifecycle Stage Group Configuration | Auto-on | Platform |
| Maximum Character Limit for Instructions | Auto-on | Platform |
| Rename Start Multi-Document Workflow to Start Document Workflow | Auto-on | Platform |
| VQL for Multi-Document Workflows | Auto-on | Platform |
| Reporting & Dashboards | ||
| Contains Operator in Reports | Auto-on | Platform |
| Run Report in Background | Auto-on | Platform |
| Flash Reports Timeout Behavior | Auto-on | Platform |
| Reports with Multiple Down Objects in Dashboards | Auto-on | Platform |
| Usability & UI Updates | ||
| Notifications: Support for Vault Mobile Push Notifications | Auto-on | Platform |
| Checklists: Welcome Notification Templates | Configuration | Platform |
| Checklists: Duplicate Checklist Prevention | Auto-on | Platform |
| Localize Vault to Swedish | Auto-on | Platform |
| Vault File Manager | ||
| Install Vault File Manager Without Affecting Checked-Out Files | Auto-on | Platform |
| Auto-Logout of Vault File Manager After Inactivity | Auto-on | Platform |
| Vault File Manager Automatically Opens Files | Auto-on | Platform |
| Administration | ||
| Scheduled Data Exports | Configuration | Platform |
| Feature Flag Confirmation Dialog in Vault | Auto-on | Platform |
| Admins Can Edit All Picklist Labels & Picklist Value Labels | Auto-on | Platform |
| Documentation Only: Limited Release Sandbox Feature Flag | Admin Checkbox | Platform |
| Display the Unclassified Document Type in Admin | Auto-on | Platform |
| Vault Java SDK | ||
| Vault Java SDK Service Account | Auto-on | Platform |
| JSON Binding Annotations: User-Defined Model and Property | Auto-on | Platform |
| Audit Enhancements: On Behalf of User | Auto-on | Platform |
| Platform Data Model Changes | ||
| Platform Data Model Changes | Auto-on | Platform |
| Clinical Operations | ||
| Study Budgets | Configuration | Vault Payments |
| Clinical Operations & Regulatory Vault Connection: Streamlined Reuse Support | Auto-on | eTMF |
| CDMS & Clinical Operations Vault Connection: Protocol Deviations | Configuration | CTMS |
| Veeva eConsent Authoring in Clinical Operations | Configuration |
CTMS, Study Startup, eTMF
|
| Veeva eConsent Transfer with Site Connect | Auto-on | SiteConnect |
| Site Connect: Additional Vault Clinical Docs Support | Auto-on | SiteConnect |
| New State Types for Soft Deletion | Auto-on | Study Startup, eTMF |
| Archive Study From Any State | Configuration | eTMF |
| Attachments for TMF Transfer | Auto-on | eTMF |
| Conditional Actions on Trip Report Questions | Configuration | CTMS |
| Add Yuzu Japanese CTN to the Application Settings Page | Auto-on | CTMS |
| USN Picker Search Improvements | Auto-on |
CTMS, SiteConnect, Study Startup, Vault Payments, eTMF
|
| Support for EU Regulatory Agency Issue Notification Tracking | Auto-on | CTMS |
| Clinical Operations Data Model Changes | Auto-on |
CTMS, SiteConnect, Study Startup, Vault Payments, eTMF
|
| Commercial | ||
| PromoMats & RIM Vault Connection | Configuration |
PromoMats, RIM Publishing
|
| Modular Content: Approval Document | Configuration | PromoMats |
| Support Long Text Request Details field for Medical Inquiry | Auto-on | MedComms |
| Commercial Data Model Changes | Auto-on |
MedComms, PromoMats
|
| Quality | ||
| Enhanced Periodic Review for Documents | Configuration | QualityDocs |
| Process Navigator: Navigation Drawer and Favorites | Auto-on | QualityDocs |
| On the Job Training Type | Configuration | Training |
| Assign Effective Version for Training | Configuration | Training |
| Allow Learner Choice For Substitute Training | Configuration | Training |
| Reportability Assessment Management | Auto-on | Surveillance |
| Adverse Event Reporting Enhancements | Auto-on | Surveillance |
| Relationship Automation for Quality Objects (QE, Audits) | Configuration | QMS |
| Generate Quality Document from Object Record Action | Configuration | QMS |
| Batch Release Process Support | Configuration | QMS |
| Quality Data Model Changes | Auto-on |
QMS, QualityDocs, Station Manager, Surveillance, Training
|
| Regulatory | ||
| Copy Into Content Plans | Admin Checkbox | RIM Submissions |
| Active Dossier | Configuration | RIM |
| RIM Reference Model | Configuration |
RIM Publishing, RIM Registrations, RIM Submissions, RIM Submissions Archive
|
| Auto-Create Commitment Application Joins | Admin Checkbox |
RIM Publishing, RIM Registrations, RIM Submissions, RIM Submissions Archive
|
| Sequential Pagination of TOC | Configuration |
RIM Publishing, RIM Submissions
|
| Extend Content Plan Duplicate Submission Join Detection | Auto-on | RIM Submissions |
| Dynamically Adjust Content Plan Row Height to Fit Text | Auto-on | RIM Submissions |
| Submissions Archive Documents Created as System | Auto-on |
RIM Submissions Archive
|
| Propagate Fields from Source to Archived Documents | Auto-on | RIM Publishing |
| Archived Document Security in RIM Submissions | Auto-on | RIM Publishing |
| US FDA eCTD Validation Criteria v3.9 Support | Configuration | RIM Publishing |
| Registration Verification Improvements | Configuration | RIM Registrations |
| Activity Country Dependencies | Configuration | RIM Registrations |
| IDMP Process Automation | Configuration | RIM Registrations |
| Updates to IDMP Accelerators to Support UDI | Auto-on | RIM Registrations |
| XEVMPD Attachment-Only Submission | Configuration | RIM Registrations |
| XEVMPD Procedure Number | Auto-on | RIM Registrations |
| 21R2 RIM Data Model Changes | Auto-on | RIM |
| SiteVault | ||
| Set Context in Vault Selector | Auto-on |
SiteVault Enterprise, SiteVault Free
|
| Single Source of Truth for Person Profiles | Auto-on |
SiteVault Enterprise, SiteVault Free
|
| Prompt for Fields on Upload to Document Inbox | Auto-on |
SiteVault Enterprise, SiteVault Free
|
| Expanded Language Support | Auto-on |
SiteVault Enterprise, SiteVault Free
|
| Optional State Change for Send Document via Clinical Network Action | Configuration |
SiteVault Enterprise, SiteVault Free
|
| New Exchangeable Document Types on Connected Studies | Auto-on |
SiteConnect, SiteVault Enterprise, SiteVault Free
|
| Veeva eConsent: Patient Declines Consent | Auto-on |
SiteVault Enterprise, SiteVault Free
|
| Veeva eConsent: Form Responses | Auto-on |
SiteVault Enterprise, SiteVault Free
|
| Veeva eConsent: Shareable eConsent Preview | Configuration |
SiteVault Enterprise, SiteVault Free
|
| QualityOne | ||
| Training Management Phase 3 | Configuration | QualityOne Training |
| Inspection & NCR Product Model Compatibility | Configuration | QualityOne |
| HACCP Data Model | Configuration | QualityOne |
| Incident Management - Track Labor Information | Auto-on | QualityOne |
| COA Textract Response File Download | Configuration | QualityOne |
| Support Localized Number Separator for COA | Auto-on | QualityOne |
| RegulatoryOne | ||
| Regulatory Request Management | Configuration |
RegulatoryOne Compliance Management
|
| Dynamic Template to Create Requirements | Configuration |
RegulatoryOne Registration & Dossier Management
|
| Veeva Claims | ||
| Selective Creation of Local Adaptations from a Project | Configuration | Veeva Claims |
See the following explanations for enablement options:
| Enablement | Description | Auto-On | Automatically activated and no configuration is required before using the feature; note that in some cases, a new feature is dependent on another feature that must be enabled or configured. | Admin Checkbox | Admins must turn on the feature with an Admin checkbox. Note that some "Auto-On" features have a checkbox setting that hides the feature; these will show "Auto-On." | Configuration | Admins must configure the feature (separately from an Admin checkbox) before it is available to use or is active; for example, an Admin must add document templates before users can create documents from templates. | Support | On/off option controlled by Support. |
|---|