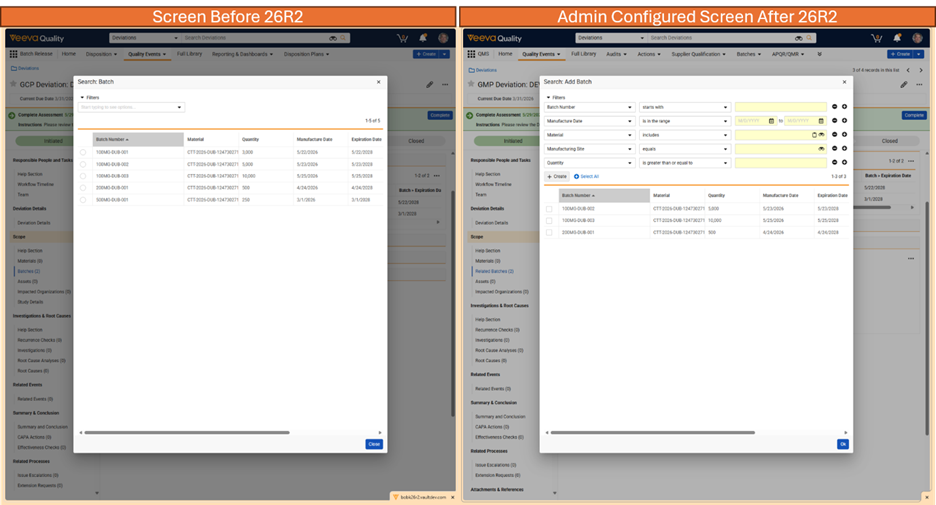
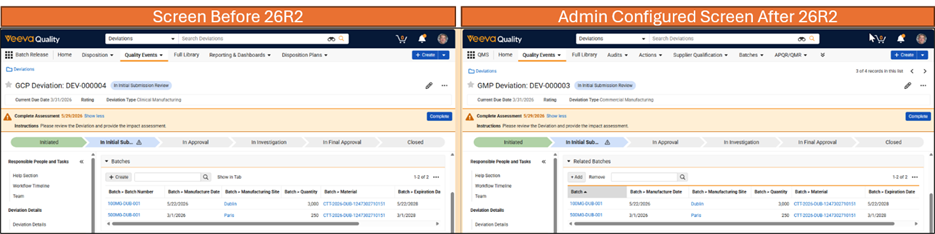
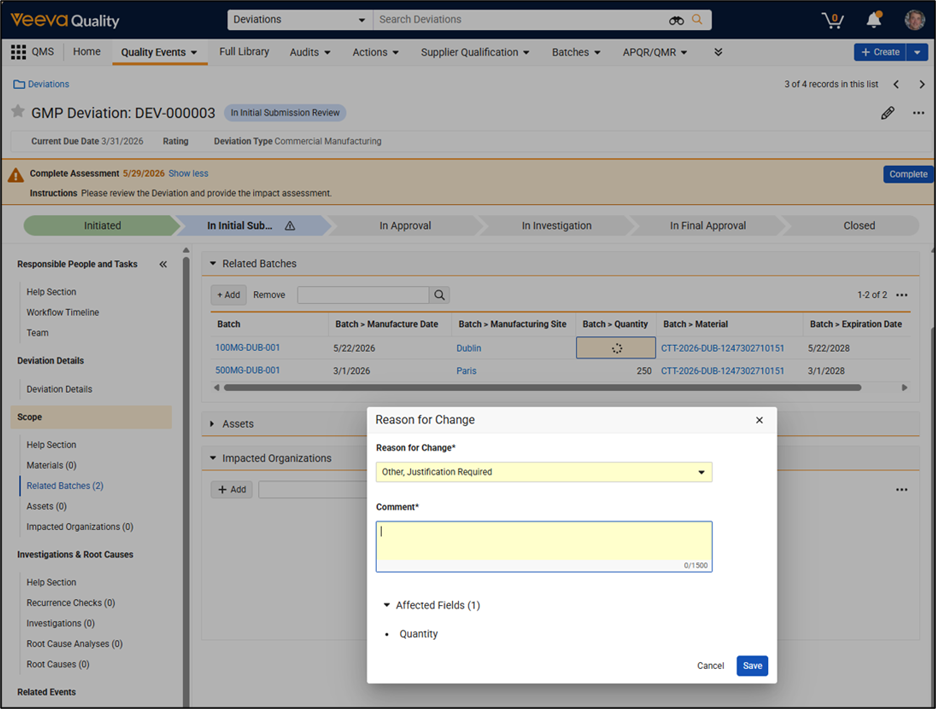
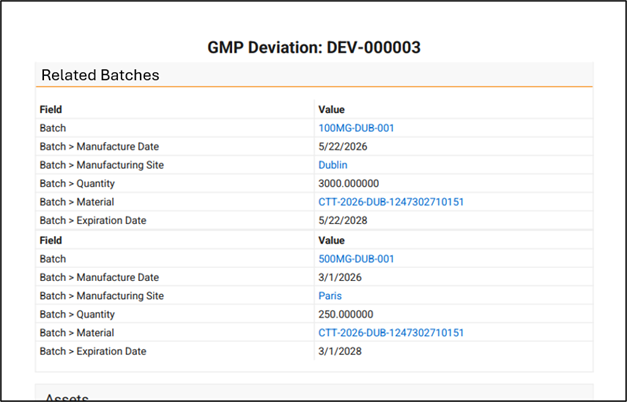
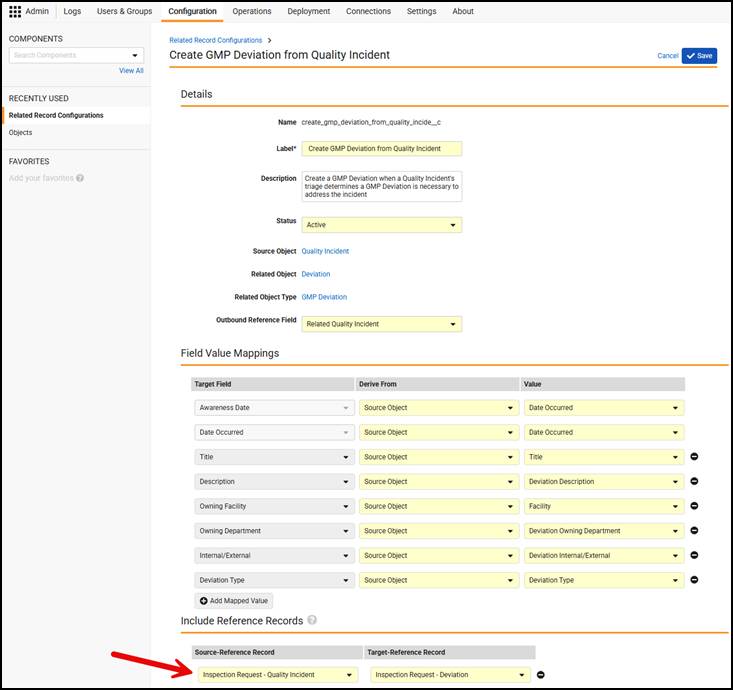
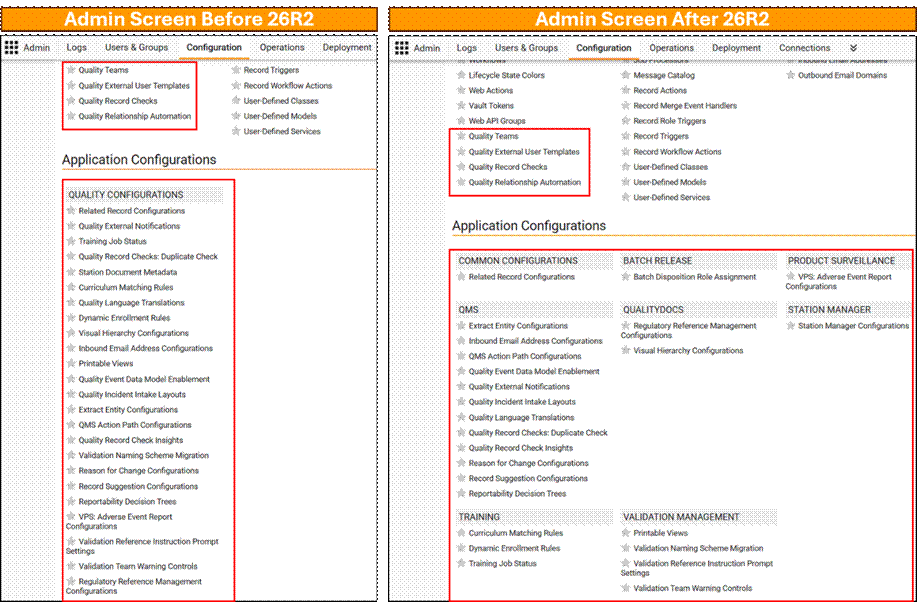
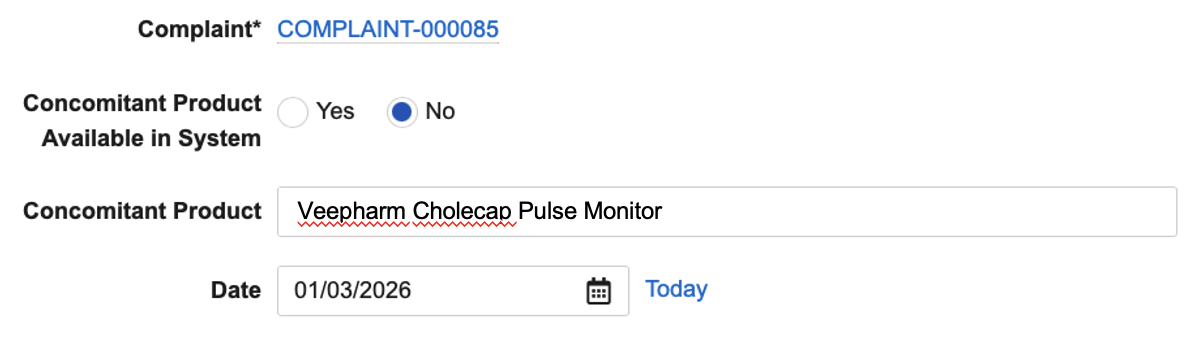
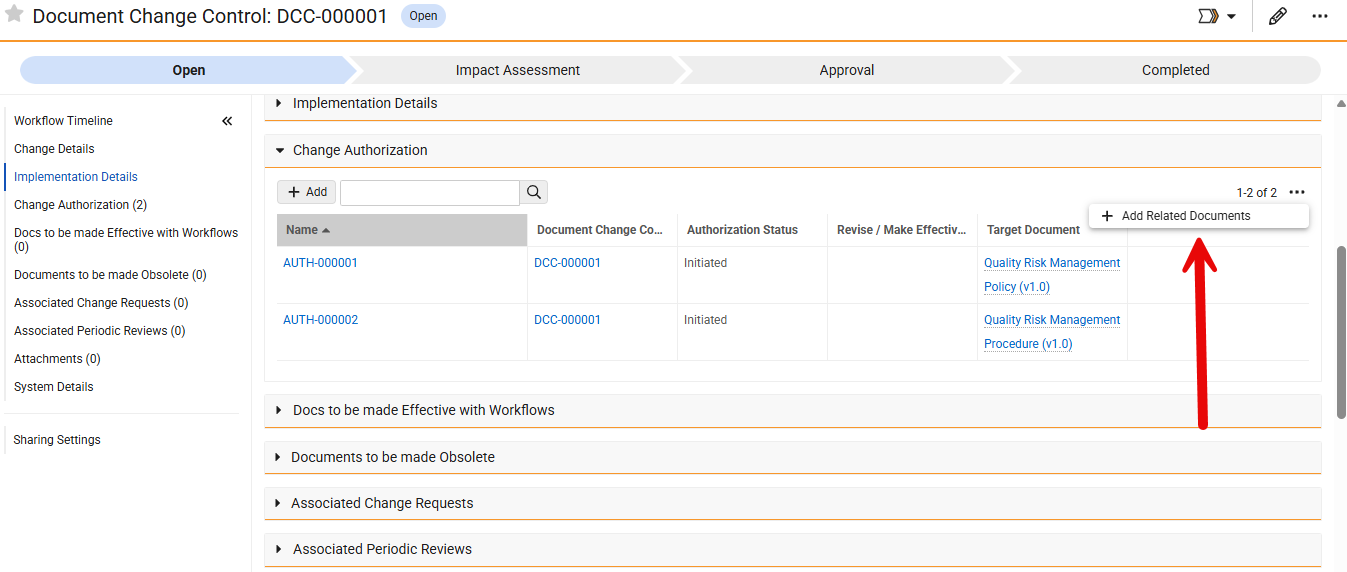
Limited Release Dates: May 1, 2026 (26R1.2); June 12, 2026 (26R1.3); June 26, 2026 (26R1.4) | General Release Date: August 7, 2026
We are pleased to bring you new functionality with each limited release. These release notes are updated with upcoming new features one week before the limited release date. See the following explanations for enablement options:
- Auto-on: Automatically activated and no configuration is required before using the feature; in some cases, a new feature is dependent on another feature that must be enabled or configured.
- Admin Checkbox: Admins must turn on the feature with an Admin checkbox. Some “Auto-On” features have a checkbox setting that hides the feature; these will show “Auto-On.”
- Configuration: Admins must configure the feature (separately from an Admin checkbox) before it is available to use or is active; for example, an Admin must add document templates before users can create documents from templates.
- Available for Use: Used only by the eConsent, eCOA, and SiteVault applications. Sponsors must make a study-specific configuration change to implement new capabilities.
Platform
Highlights
Fast SearchAuto-on26R1.3
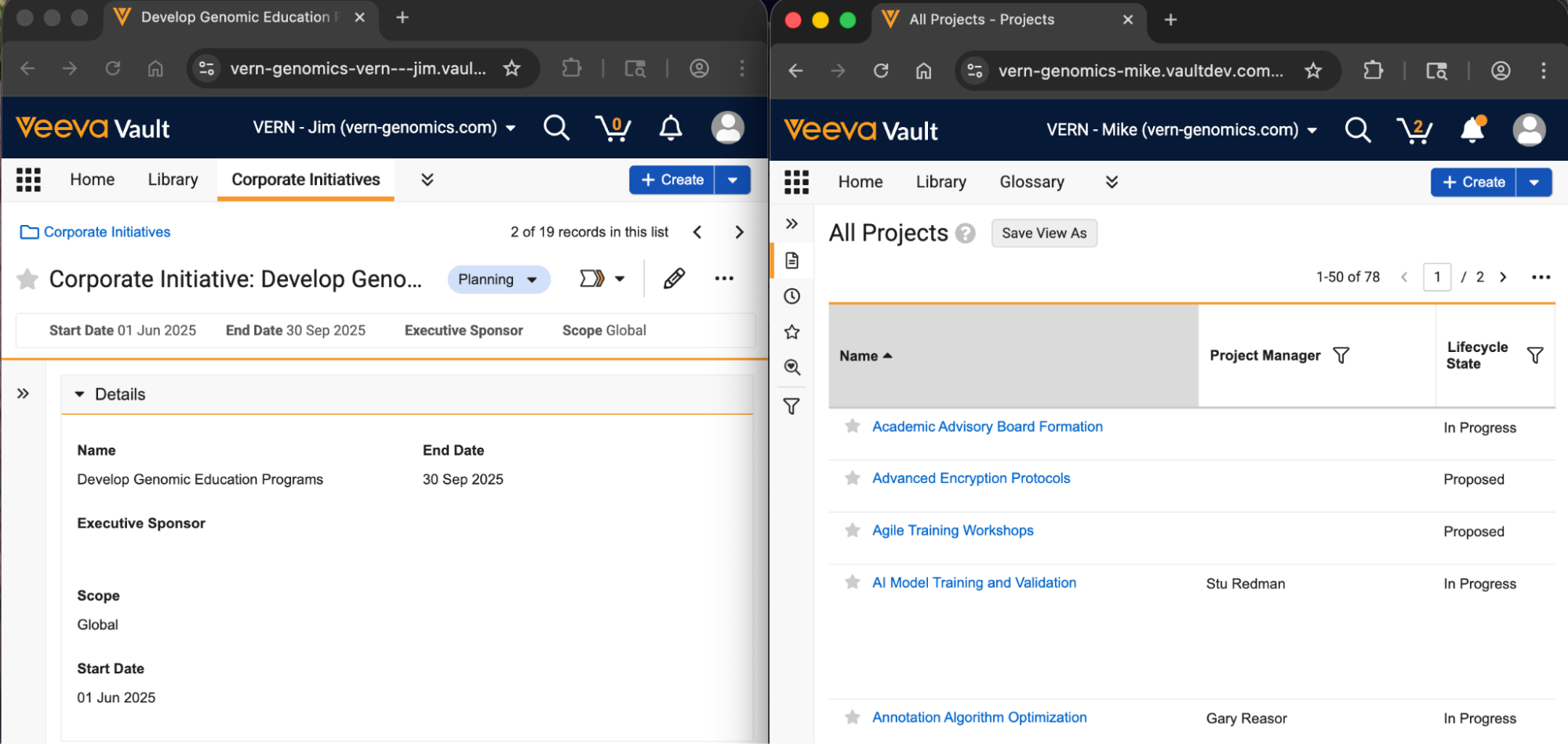
When users click into the search bar on a document tab, the suggested documents shown in the dropdown for quick access will now load faster and provide more relevant documents to users, making it easier for users to access necessary content.
Prior to 26R2, this list showed documents based on the most recently viewed documents. With 26R2, the documents shown are based on all interactions a user might have with a document, such as edits and annotations.
The performance of loading the list of documents is also improved, both when users initially click into the search box:
As well as when users begin typing text into the search box:
Additionally, this feature supports the Improved Document Viewer by providing users options to open documents in a mini-browser window or in a new split-screen view when hovering over a document:
Learn more about Searching Vault.
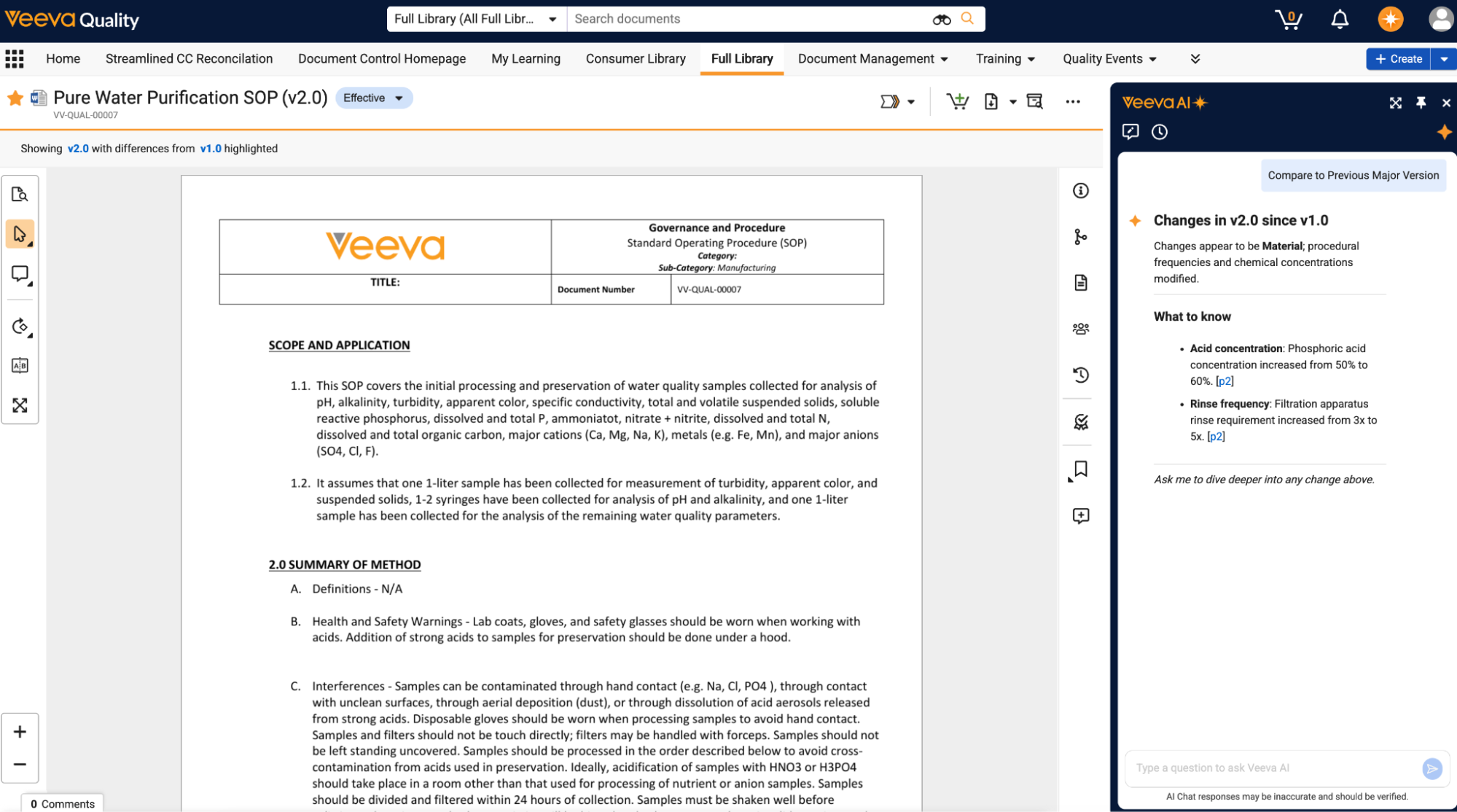
Improved Document ViewerAuto-on26R1.3
The document viewer now provides a simpler user experience, focused on displaying the most common document actions to users.
This includes a new vertical toolbar that includes actions like Full Screen, Zoom, and Find in Document:
Zoom controls have also been improved by introducing a new zoom slider and allowing users to enter an explicit zoom percentage:
For standard document sizes (letter, legal, A4), the zoom defaults to 100%.
Actions that open additional panels are now included on the Doc Info pane toolbar with other panel options. This includes Bookmarks, Destinations, Glossary, Thumbnail View, and Annotations:
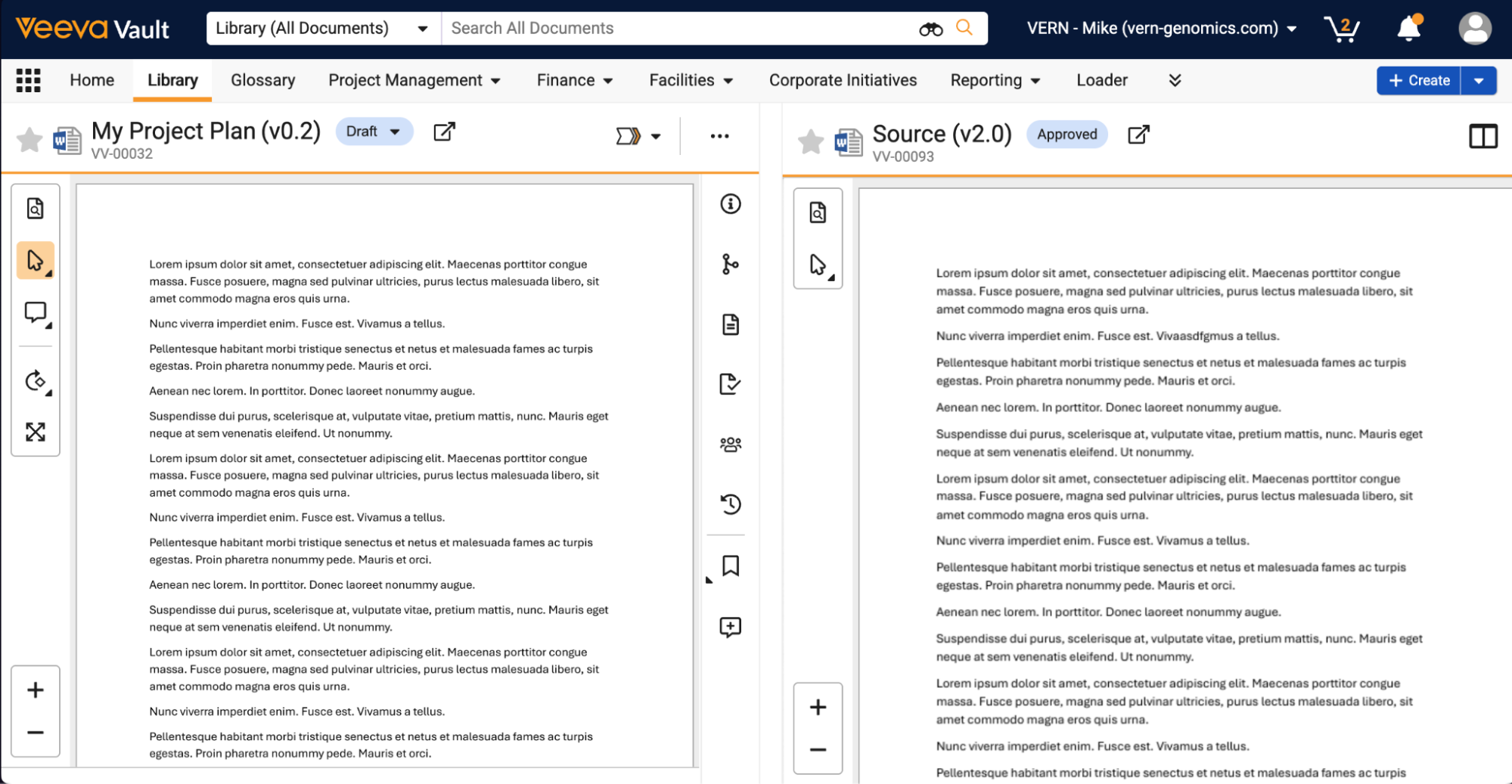
Users can also now easily view multiple documents at once by opening documents in a mini-browser window or in a side-by-side viewer experience through Fast Search, relationships, attachments, and document versions:
Additionally, for customers with Vault AI enabled, clicking the new Version Comparison button includes a generated AI summary of changes in Vault AI Chat that shows whether there were material changes since the last major version and summarizes important changes to review.
These changes are also reflected in the external viewer used when using Send as Link to Non-Vault Users and other application-specific areas.
If Lifecycle Stages are configured, they will now only be shown on non-steady state versions.
Responsive UIAuto-on26R1.2
The Vault user interface now automatically scales to a user’s browser width, collapsing sidebars to maximize space and adjusting field layouts to reduce horizontal scrolling.
Even without adjusting browser width, users can manually collapse and expand sidebars, allowing them to focus on content over navigation, if desired.
If a sidebar is manually collapsed, Vault remembers that as a user preference across tabs.
Also, Vault now respects font size changes made within a user’s browser preferences, ensuring a consistent experience across different sites and web applications users may leverage.
Learn more about Supported Browsers.
Copy Link for RecordsAuto-on26R1.2
Vault supports a new Copy Link action for object records, allowing users to copy a clean and shortened URL to share with others. The copied link automatically redirects users to the record detail page when pasted into a browser web address bar. This new action supports new shortcut keys.
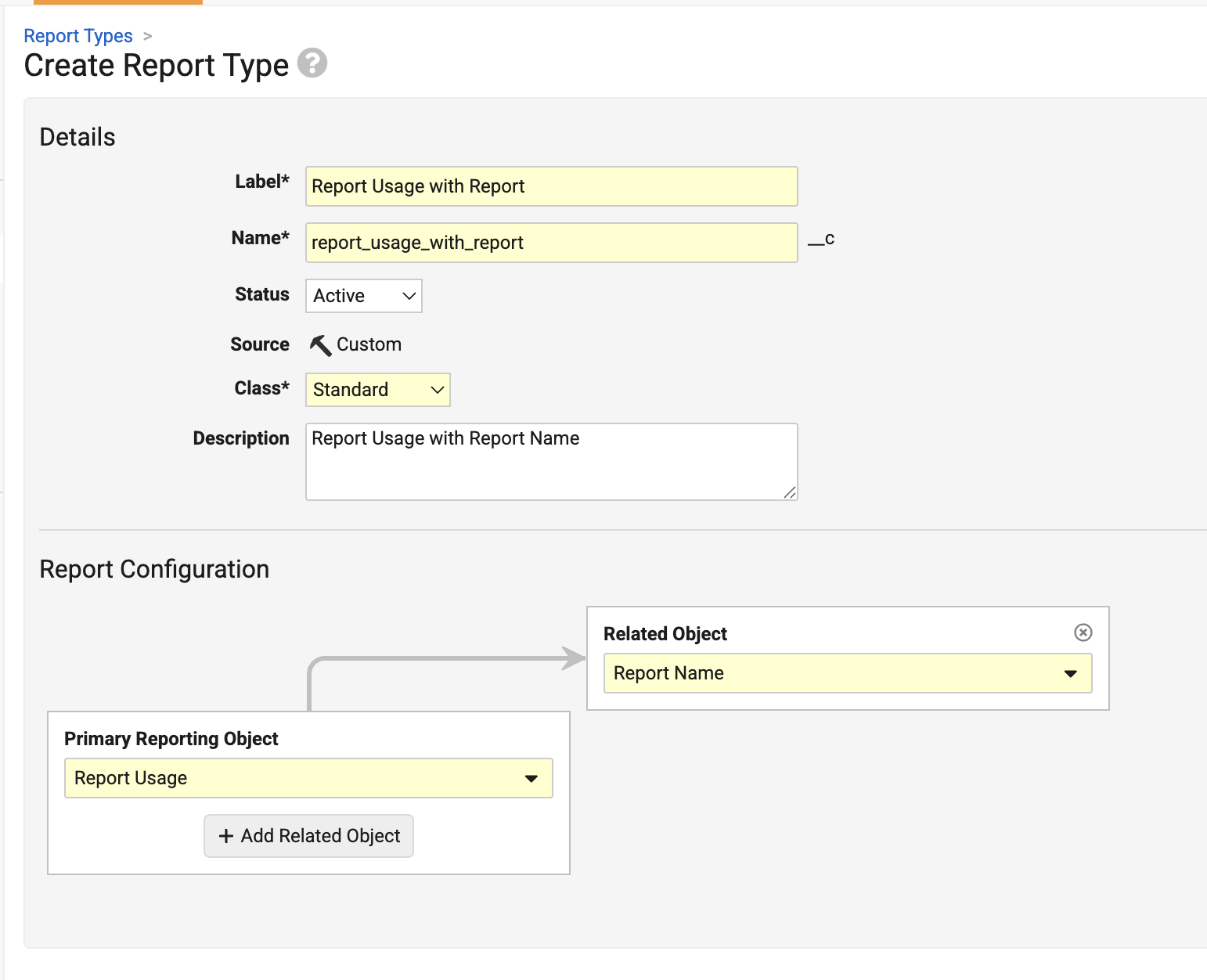
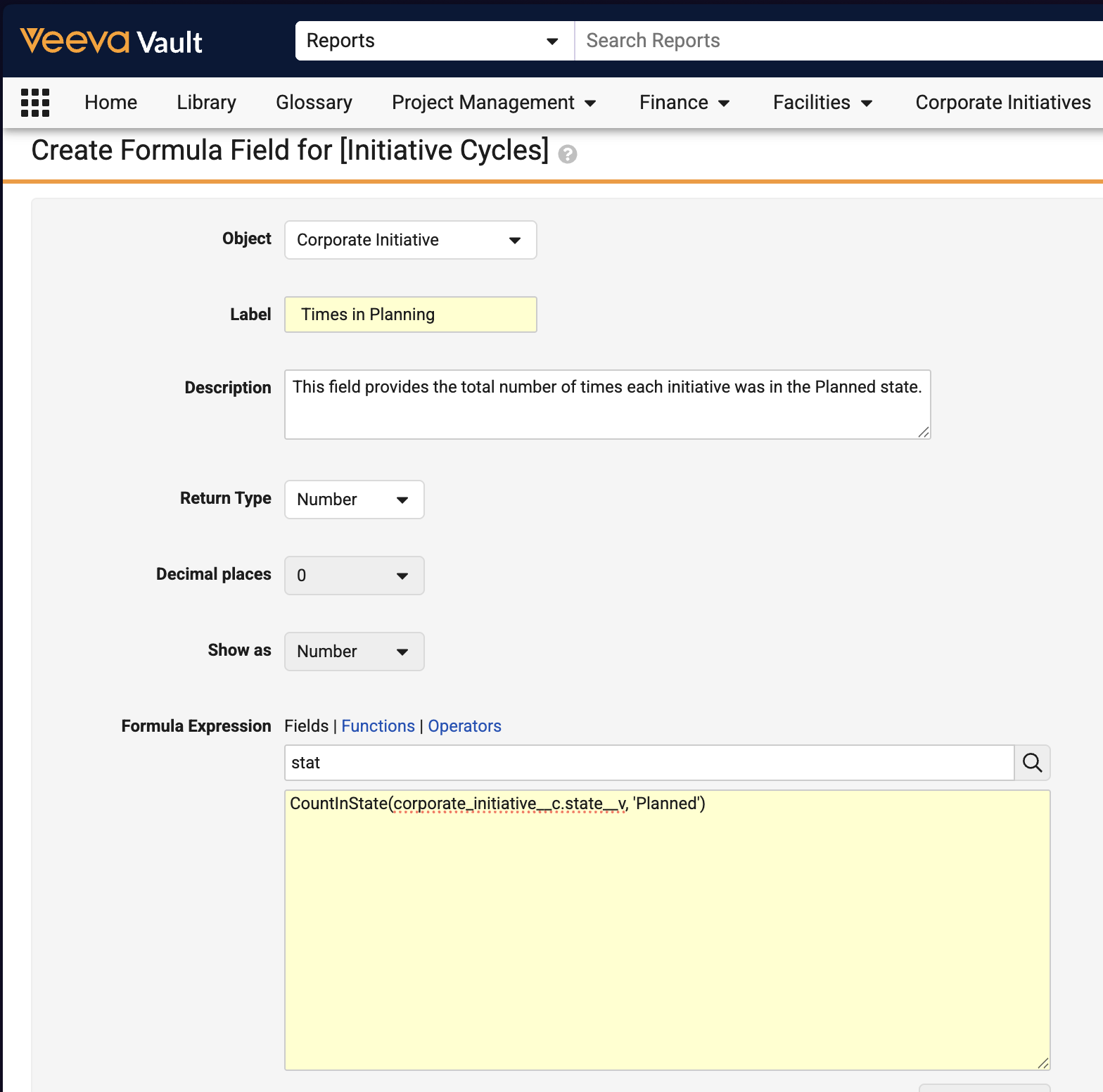
Report UsageConfiguration26R1.3
Vault now supports analyzing report runs and usage, making it easier to perform regular report maintenance activities, such as removing unused reports.
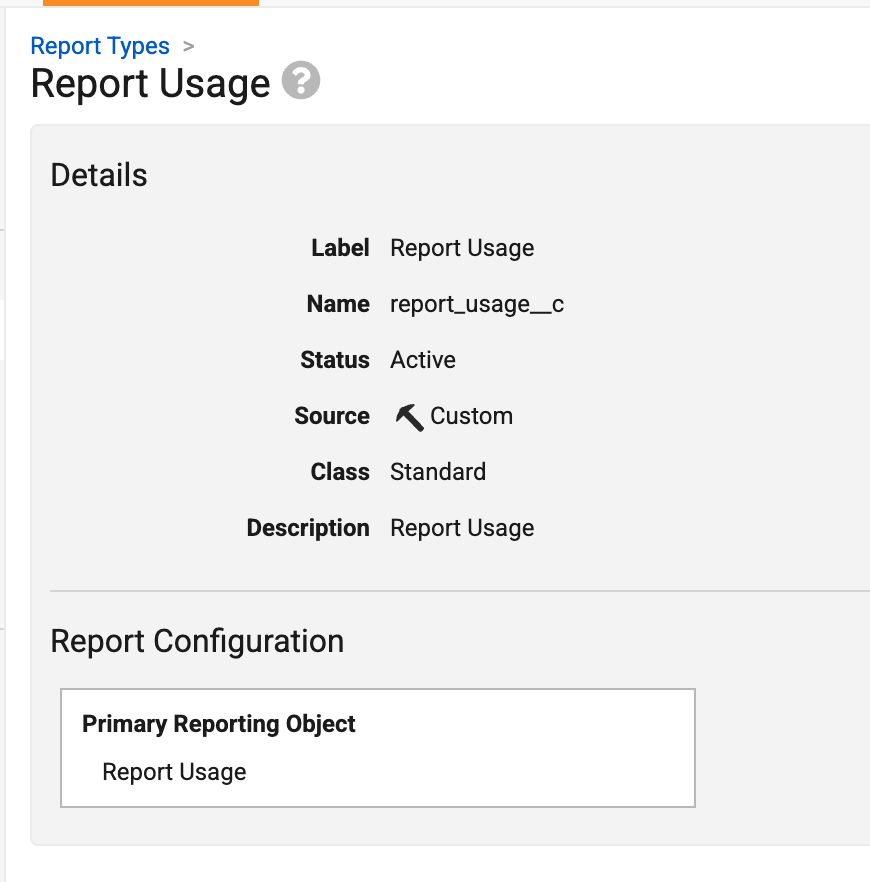
To support this enhancement, a new Report Usage (report_usage__sys) object is introduced. Admins can create a report type based on this new object:
The following data is available for analysis:
- Report Name
- Report API Name
- Report ID
- Report Run Type, including:
- Ad Hoc
- Click and Run
- Flash Report
- Run-in-Background
- Dashboard
- Last Ran By
- Last Ran Date
- Rows Returned
- View Count
This enhancement also allows Admins to report on the Report (report__sys) object itself, allowing reports to be included along with Report Usage:
As part of this change, the existing Last Ran field on reports has been deprecated and is no longer an available column when viewing reports in a list view. Last Ran is still displayed when viewing flash reports or dashboards.
Learn more about Configuring Report Types in Vault Help and Managing Reports - Best Practice Series in Veeva Connect.
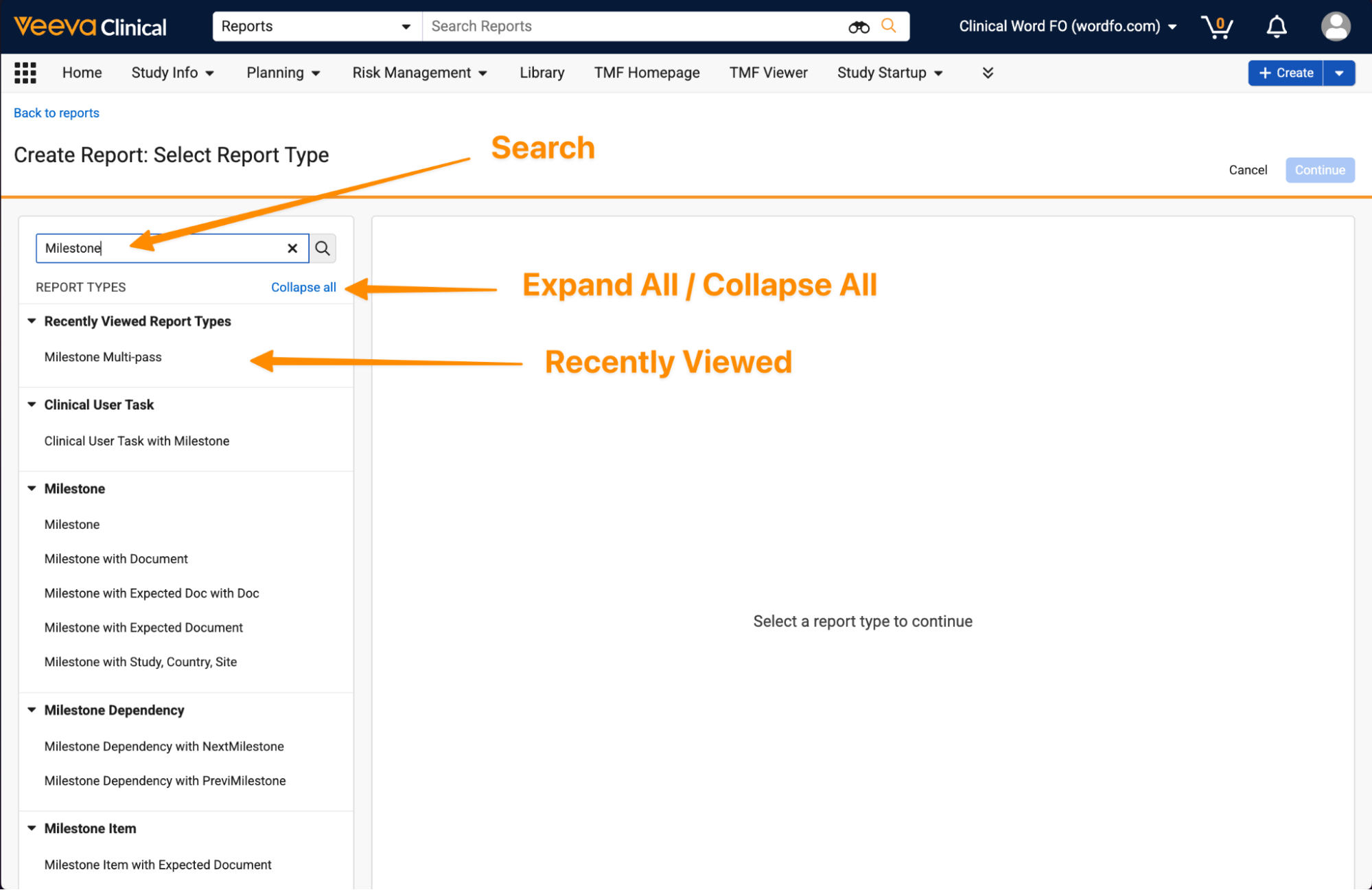
Enhanced Create Report PageAuto-on26R1.3
The Create Report page has been improved to allow users to more easily identify the right report type when building reports. These improvements include the ability to search report types and their underlying objects, easily access recently viewed report types, and expand or collapse the report type list:
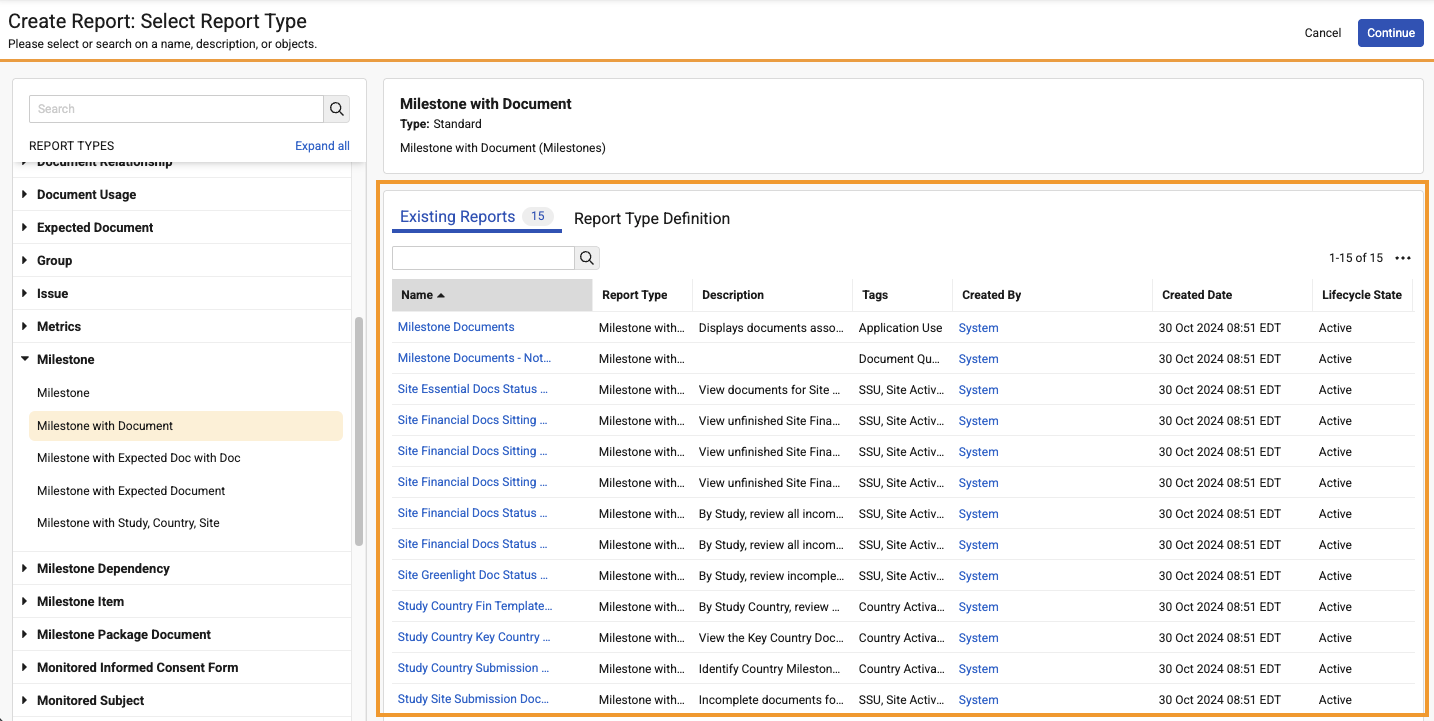
When a report type is selected, users can see existing reports using that report type:
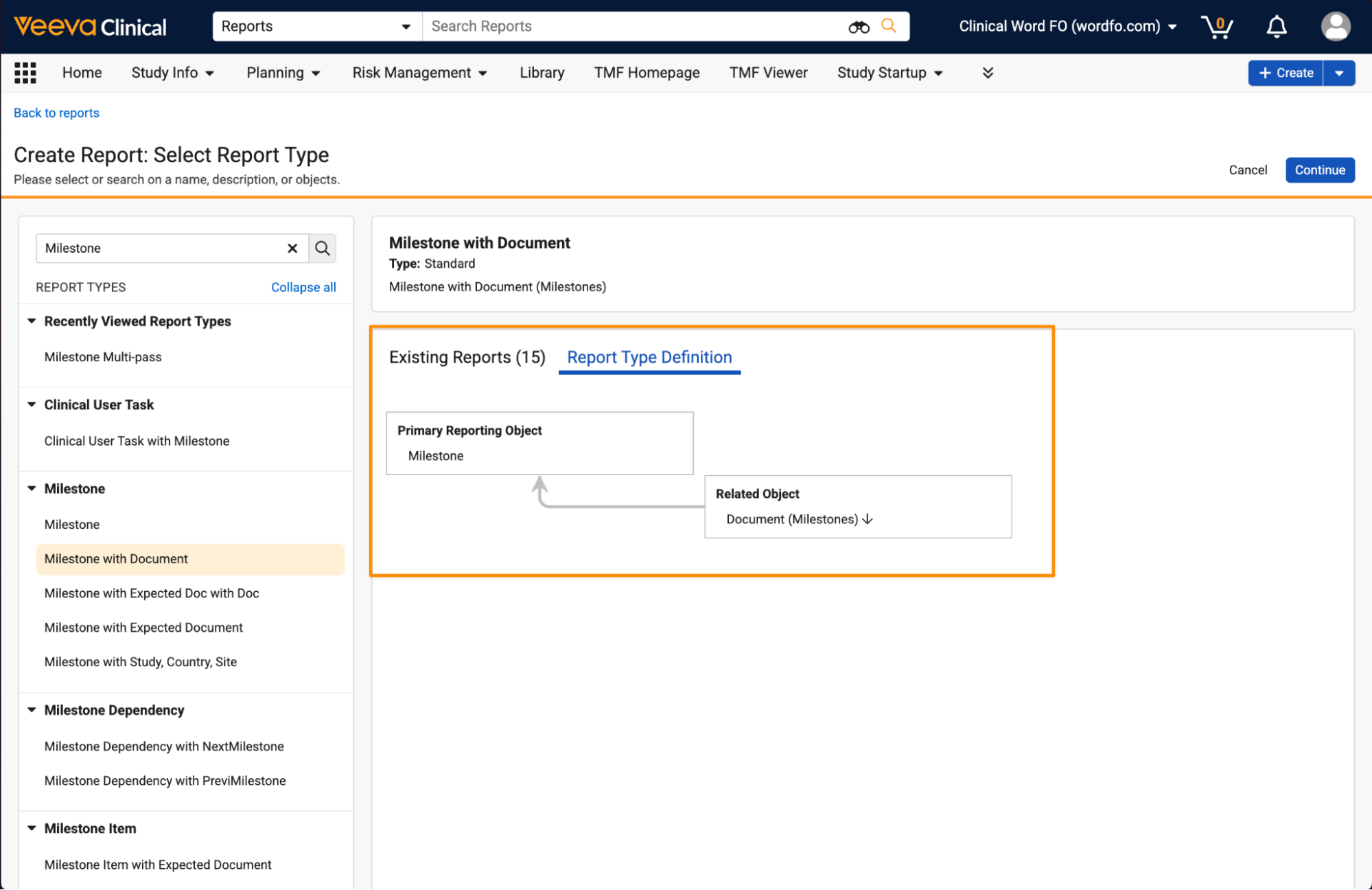
Users can also view the report type definition to see the structure of the report type and its entities:
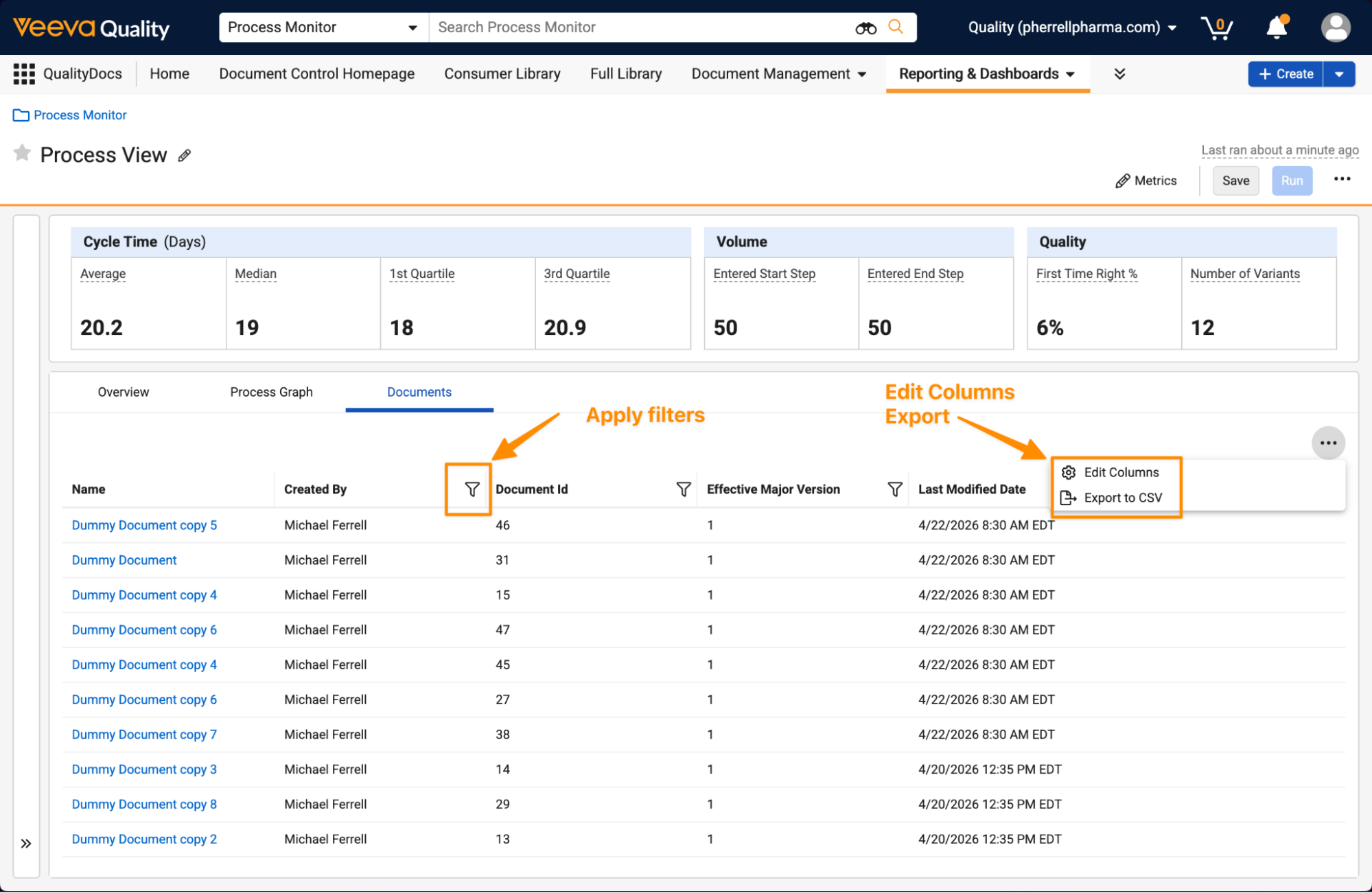
Once a report type is selected, users are prompted with an Update Report Configuration dialog, where they can name the report, add tags, and adjust the report format. For reports that include documents, this also allows users to filter by Type, Subtype, and Classification, as well as choose whether to include all versions.
This same dialog can be reopened and updated when clicking the Edit icon by the report name.
Learn more about Creating Reports.
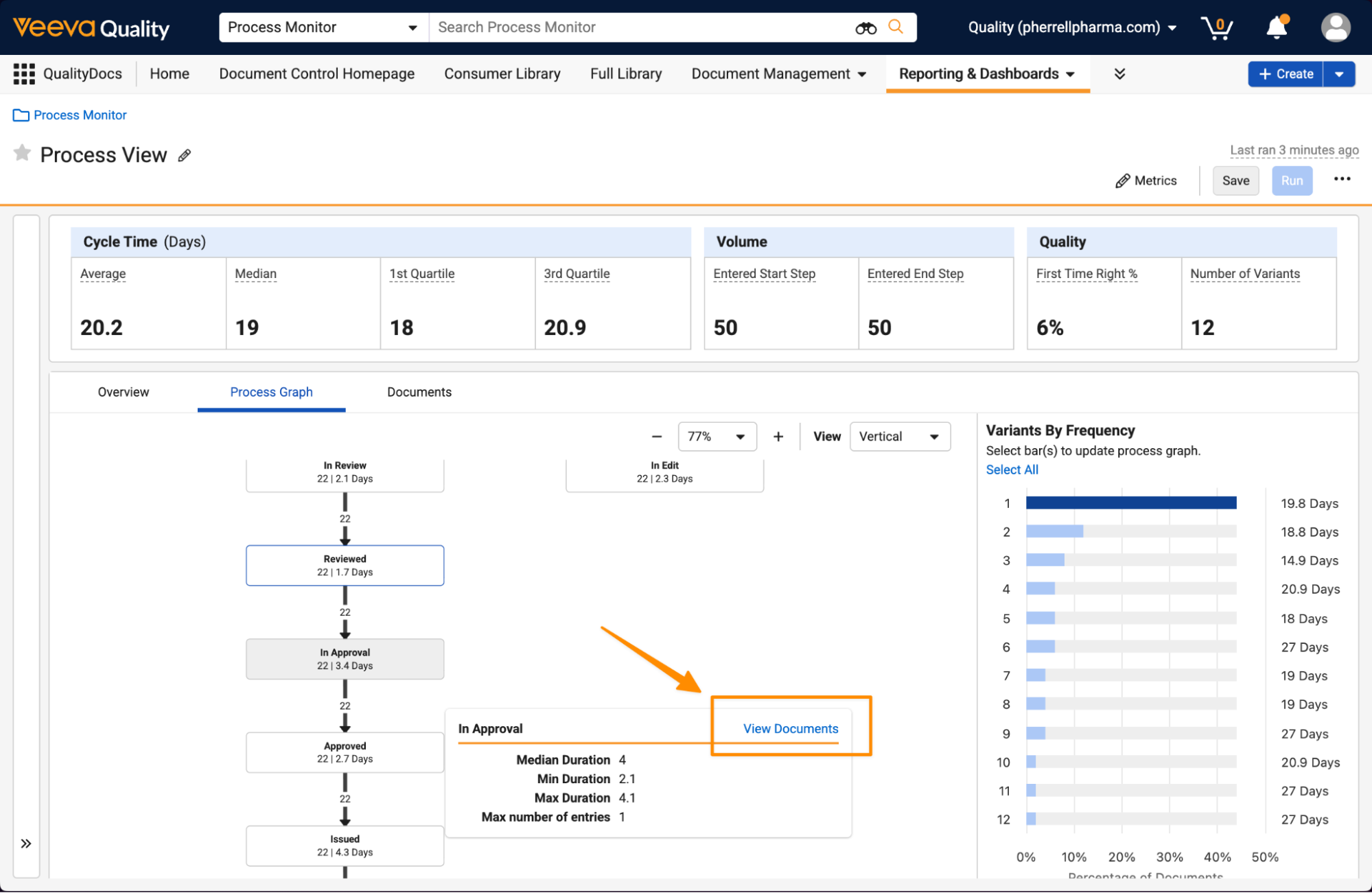
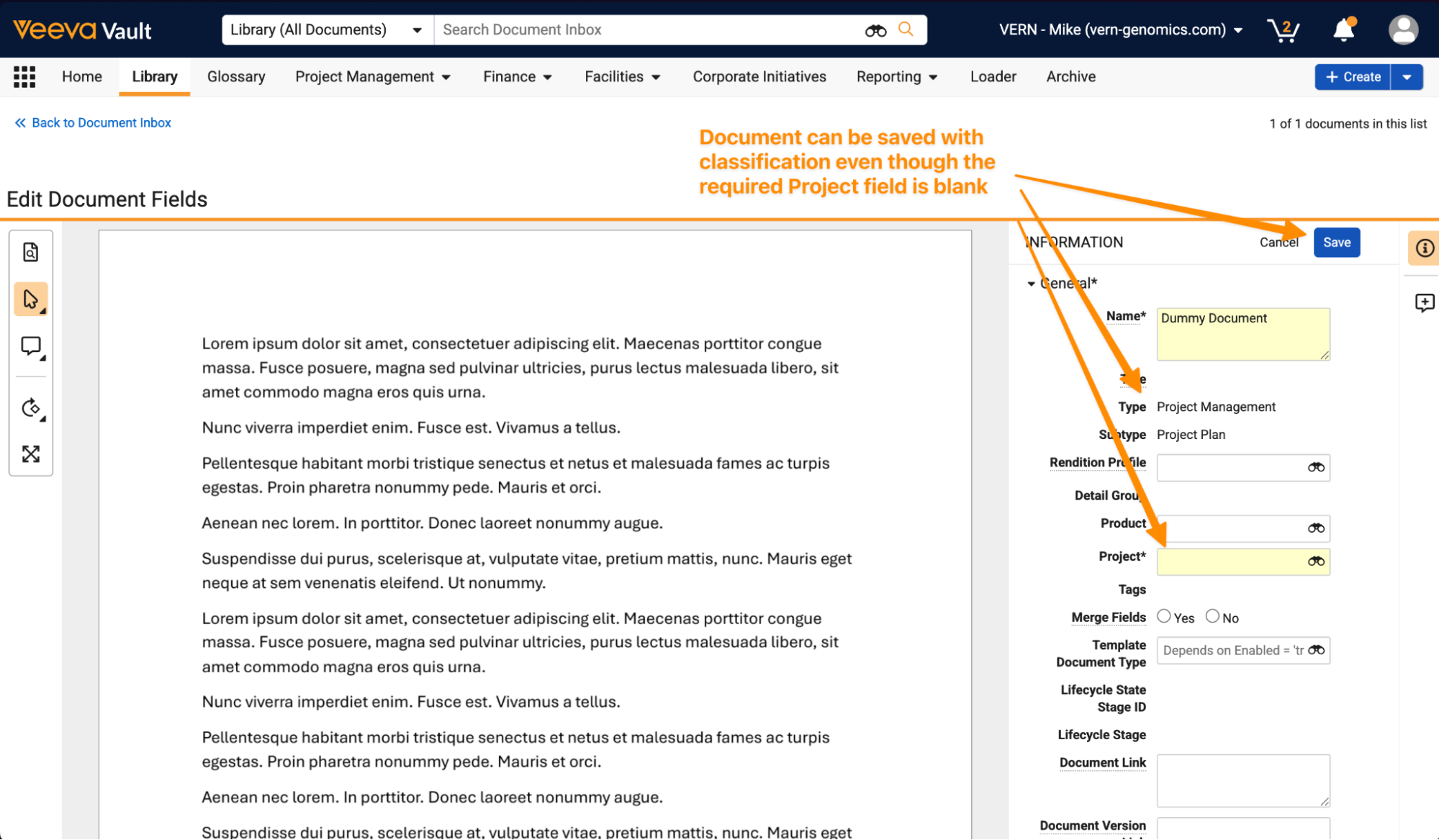
Process Monitor: Drill DownAuto-on26R1.3
Process Monitor now provides an option to explore specific records or documents that are included in a process directly in the Process View itself. This enhancement makes it easy for users to view details of what records are contributing to cycle times across process variants.
This can be accessed as a new tab within the Process View:
Within this tab, users can edit columns, apply filters, or export details to CSV:
Users can also easily navigate to prefiltered lists of records from states or transitions in the Process Graph tab.
Learn more about Process Monitor.
Managing Documents
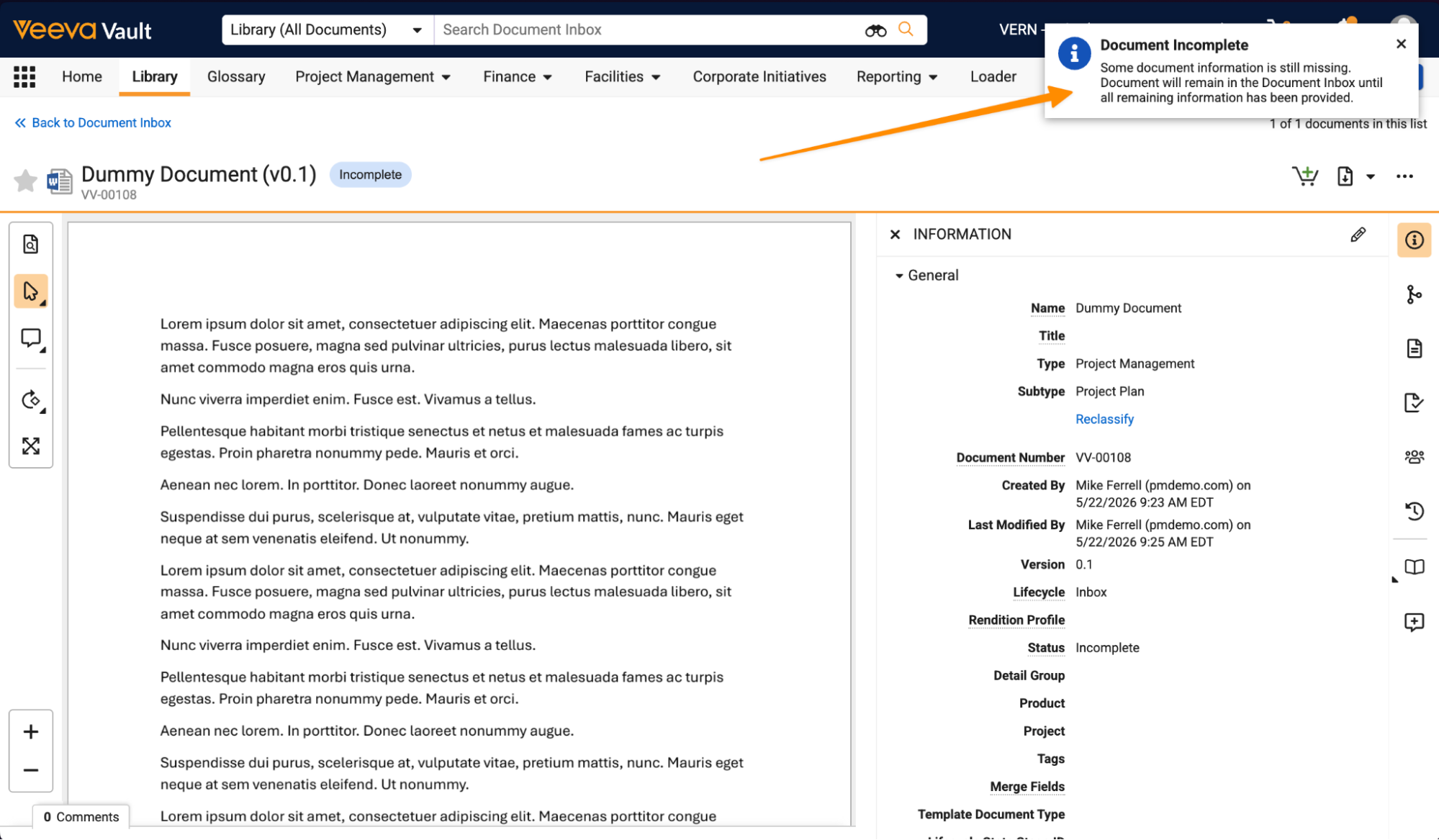
Document Inbox Enhancements: Edit Document TypeAuto-on26R1.3
Users can now classify Incomplete documents in the Document Inbox without providing all the required document field information. This allows users to still be able to make progress on documents even if not all required information is known yet.
Documents remain Incomplete in the Document Inbox until all required fields are completed. Vault displays a banner to make sure the user is aware that the document is not fully complete yet:
As part of this feature, labels related to the Document Inbox have been updated:
- Unclassified is now Incomplete: The Unclassified lifecycle state label has been updated to Incomplete to better reflect the document’s status.
- Complete is now Edit: The action to finalize a document in your Document Inbox has been renamed from Complete to Edit.
Learn more about Unclassified Documents.
Collaborative Authoring: Increase Participant Limit for Automatic MentioningAuto-on26R1.3
When using the option to automatically check out documents to collaborative authoring as part of a workflow, Vault now supports granting up to 100 workflow participants access to the document on SharePoint. This is an increase from the prior limit of 15 workflow participants.
Granting this access on check out enables these users to be @mentioned in the document before they have joined the collaboration session.
If more than 100 participants are chosen when the workflow begins, no participants are added to the document on SharePoint until they have clicked Edit in Microsoft Office.
Learn more about Collaborative Authoring Workflows.
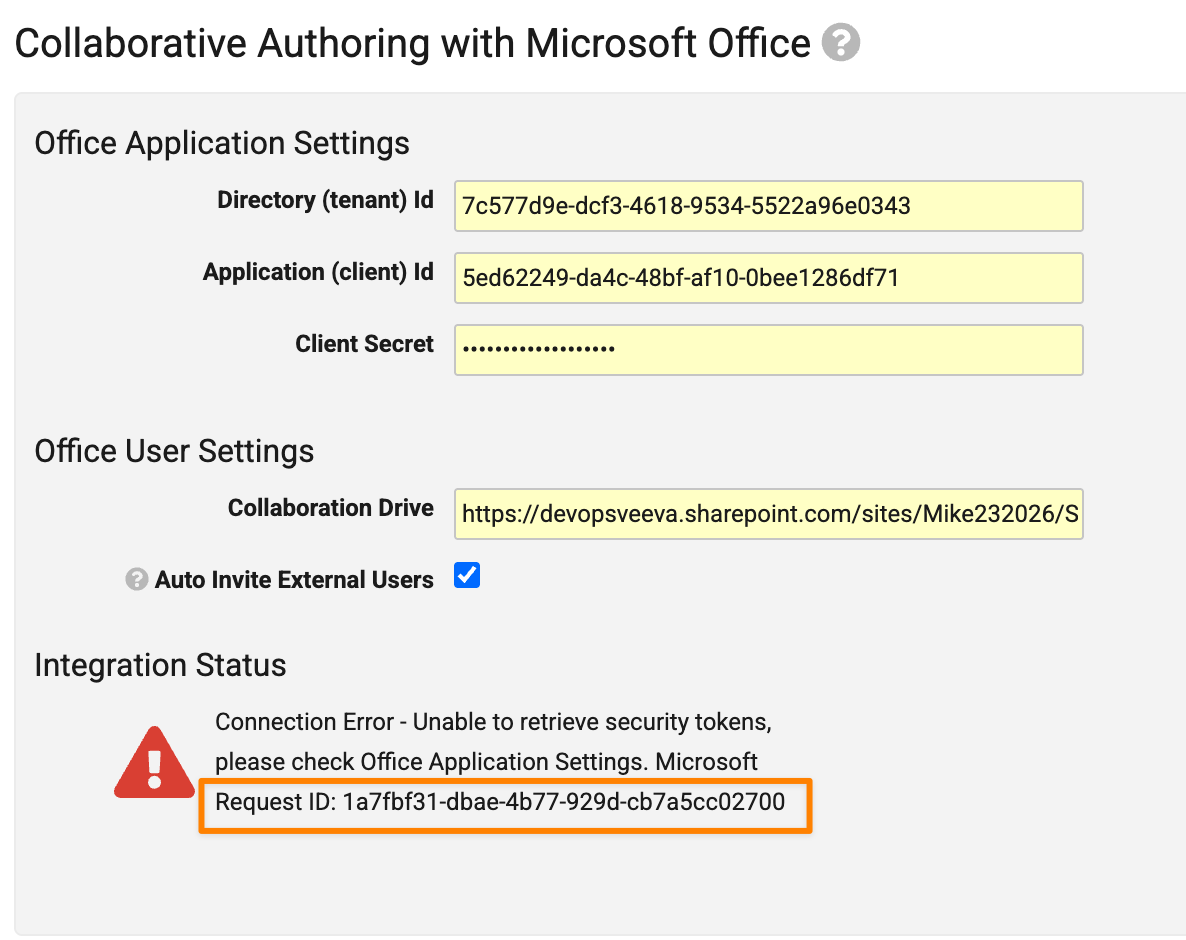
Collaborative Authoring Logs for OAuth ErrorsAuto-on26R1.3
The Microsoft Request ID is now included when an Integration Status error exists in Admin > Settings > Checkout Settings > Collaborative Authoring with Microsoft Office. Previously, the Request ID was only included in the Collaborative Authoring Error Logs, but this would not capture errors in setting up the configuration itself. In cases where the issue may not be related to Vault functionality, Admins can provide the Request ID when raising support tickets with Microsoft to streamline issue identification and resolution.
Token refresh errors are now also included in Admin > Logs > Collaborative Authoring Error Log.
Learn more about Configuring Collaborative Authoring.
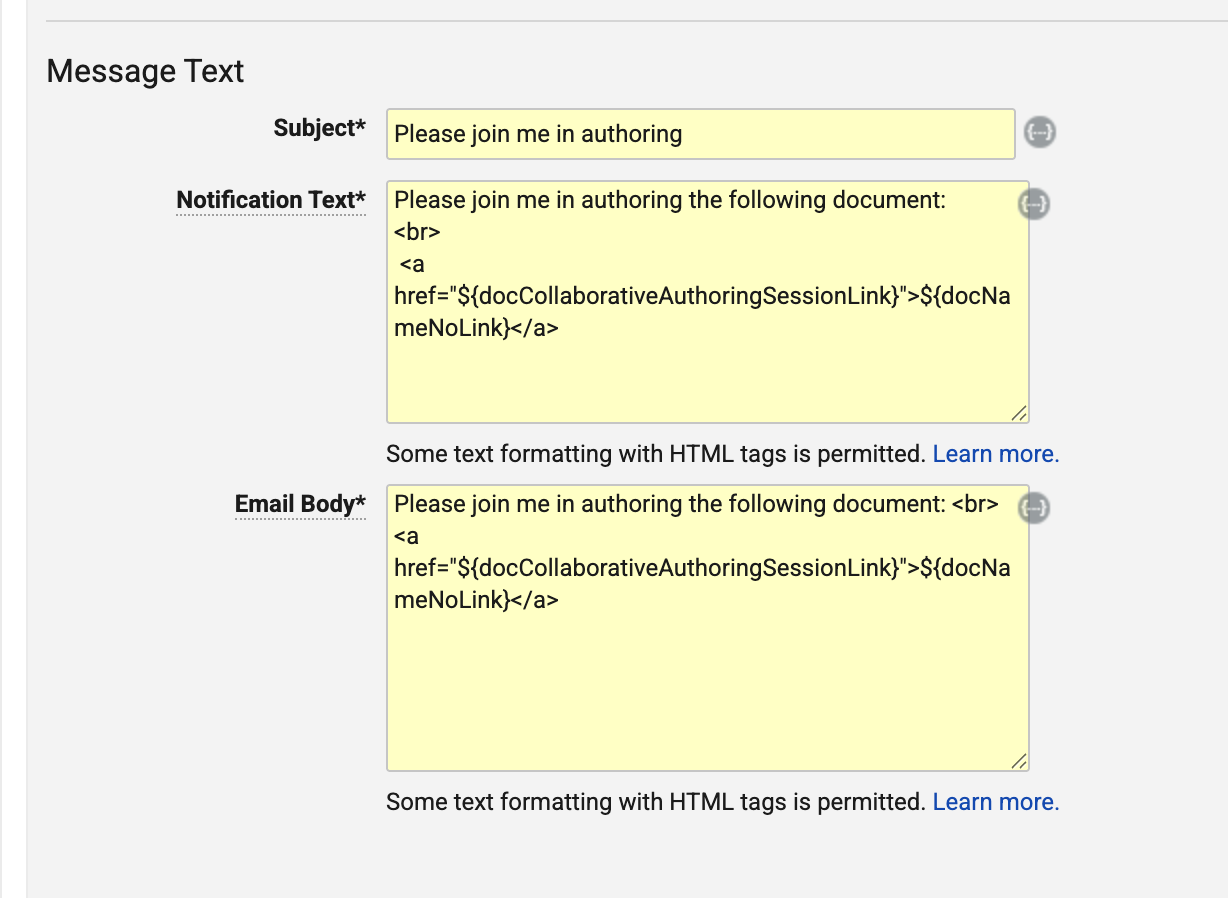
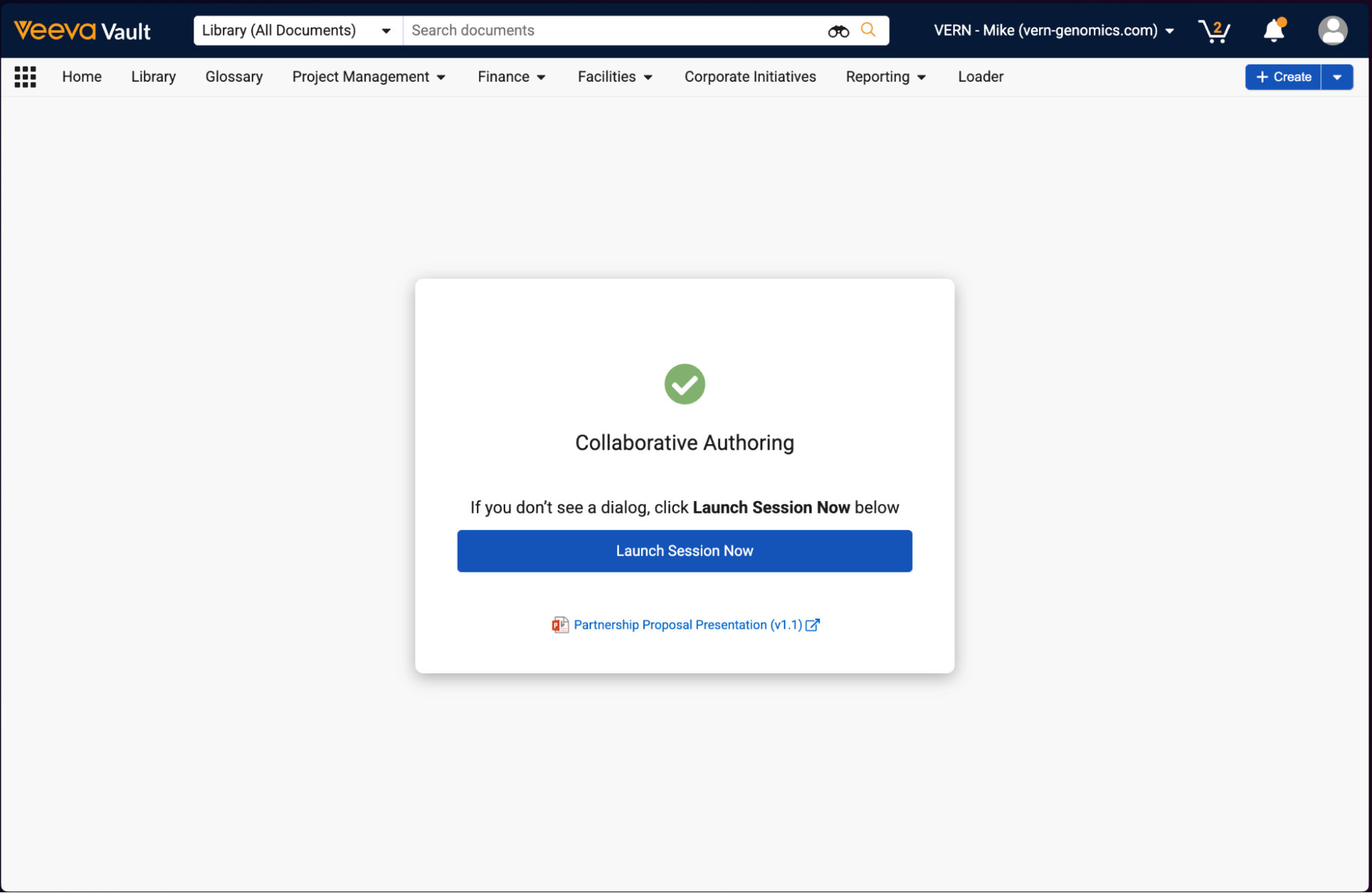
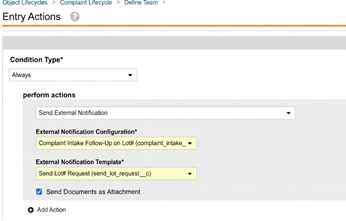
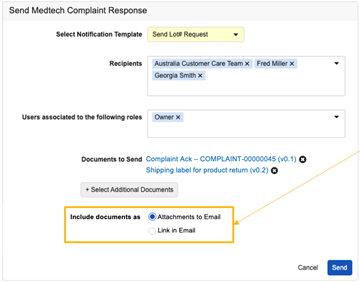
Launch Collaborative Authoring from LinkConfiguration26R1.2
For Document Notification Templates for single document workflows, Admins can now configure templates to include links to join collaborative authoring sessions directly from email notifications. This enhancement can streamline the user experience by allowing authors to more quickly work on documents that they need to author.
To enable this feature, Admins can use a new token, ${docCollaborativeAuthoringSessionLink}, in HTML link syntax.
For instance, the following notification would show the Document Name as a hyperlink that launches collaborative authoring:
<a href="${docCollaborativeAuthoringSessionLink}">${docNameNoLink}</a>
When recipients click one of these links from an email, they are required to authenticate (unless they are already authenticated in that Vault), and are then routed to a new landing page that automatically opens the document in collaborative authoring.
This enhancement applies to workflows run on single documents only (where Use workflow for single document is enabled) that are configured to use Document Notification Templates. With the HTML syntax, if including a different token to display as the link (such as ${docNameNoLink} above), the token must not resolve to its own hyperlink.
Learn more about Email & Messages Administration and Collaborative Authoring.
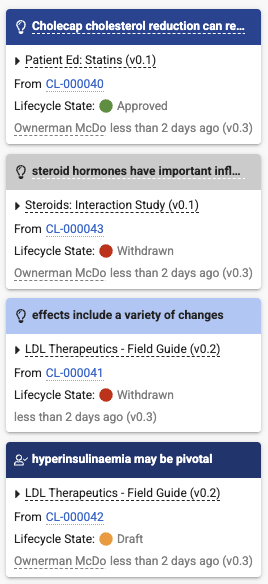
Dynamic Styling for Claim-Based Link AnnotationsAuto-on26R1.2
Reviewers can now quickly gauge how a Claim-based link annotation was created, as well as the current lifecycle state of the associated Claim record.
Vault now displays distinct annotation banner colors based on the method of creation. In addition, for Auto Link annotations only, when the related Claim is withdrawn, the annotation banner color changes to silver.
Vault also now displays a colored circle indicator to better highlight the related Claim’s lifecycle state.
Banner colors:
- Dark blue for Claim Link, Approved Link, Document Link, and Permalink Link
- Blue for Auto Link
- Light blue for pending Suggested Link
- Silver for Auto Link when the Claim record is withdrawn
Circle indicator colors:
- Green for Approved
- Red for Withdrawn
- Orange for all other states
Learn more about Claim Link Annotations.
Make a Copy: Exclude Inactive Record ValuesAuto-on26R1.3
When using Make a Copy to copy a document, inactive object record values in object reference fields are no longer copied to the new document. This ensures consistency with how picklist fields behave, and ensures that inactive values are handled the same for copies as they are for new documents.
Learn more about Copying Documents.
Managing Data
Document Fields in Object Formula FieldsConfiguration26R1.2
Admins can configure formula fields on objects to use document fields if the object references one or more documents. This feature allows Admins to use or display data from a document selected in a document reference field, such as Major Version, Minor Version, Document Number, and other supported document field types.
Learn more about Configuring Formula Fields in Objects.
Control Binding for Object FieldsAuto-on26R1.2
This feature allows Admins to bind object fields to a control. In the past, Admins could only configure controls on a layout. An object field with control binding is always displayed using that control in record detail views, list views, and related object sections.
Learn more about Configuring Vault Objects.
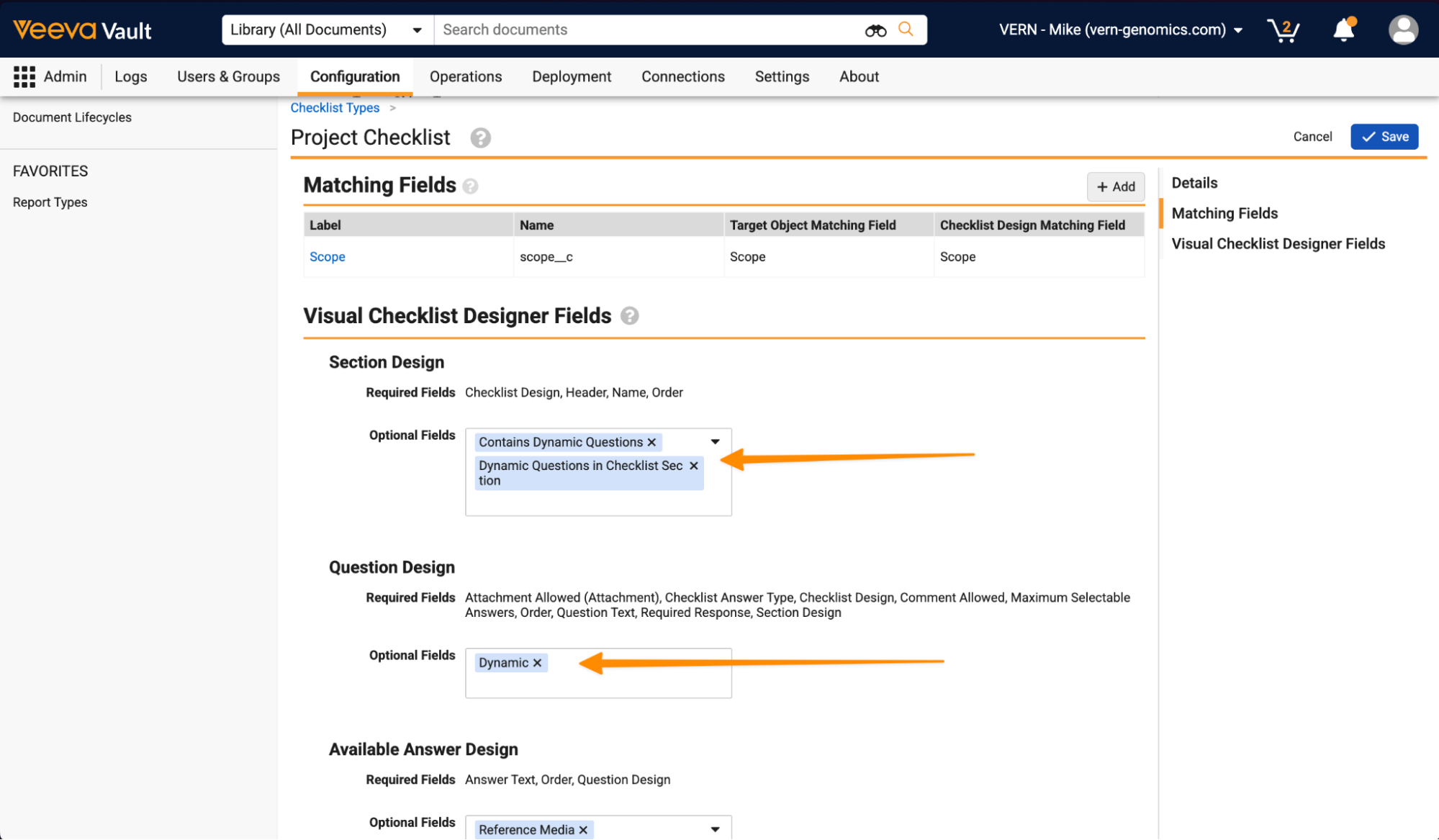
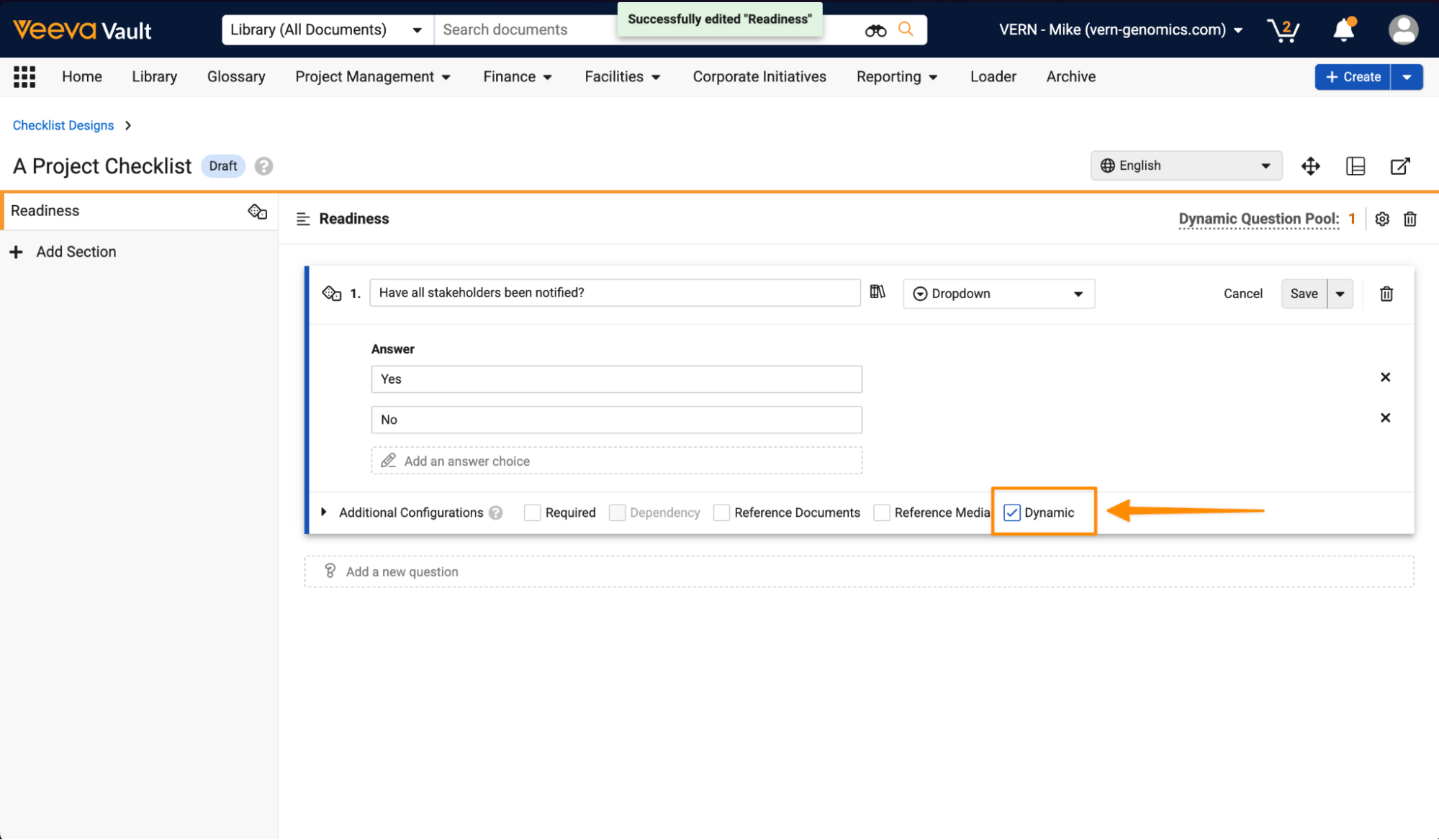
Checklists: Dynamic QuestionsConfiguration26R1.3
Admins can now create a pool of dynamic questions for each section in a Checklist Design. From this question pool, only a random subset of these dynamic questions are created in the instantiated checklist.
To leverage this feature, the Checklist Type must include:
- Dynamic Questions in Checklist Section and Contains Dynamic Questions in Section Design Optional Fields
- Dynamic in Question Design Optional Fields
Once enabled, when configuring Checklist Designs, Admins can determine per Section if dynamic questions are included and how many to include in the instantiated checklist:
Then questions can be added to the dynamic question pool using the Dynamic checkbox:
A Section must contain more available dynamic questions than are expected to be displayed to users.
This feature is not supported for Quiz functionality in QualityOne Vaults.
Learn more about Configuring Checklists.
Where Used Object ActionAuto-on26R1.3
Admins can now use the Where Used action available on the object record Actions menu. When executed, this action generates a CSV report listing all references to the target record, including both blocking and non-blocking references.
Word Formatted Output: Support for GetRelatedRecords FormulaConfiguration26R1.2
When configuring Word Formatted Output templates, Admins can now leverage the GetRelatedRecords() formula in HideRowIf() and HideTableIf() syntax.
This feature allows scenarios where an Admin may need to hide a row or table based on whether or not a particular related record exists. For example, the following formula hides the Impact Assessment Summary row if no Impact Assessment records are associated to the Change Control object by combining CountA() and GetRelatedRecords():
${HideRowIf(CountA(GetRelatedRecords($impact_assessments__vr))=0)}
Learn more about Managing Word Formatted Output Templates.
User Experience
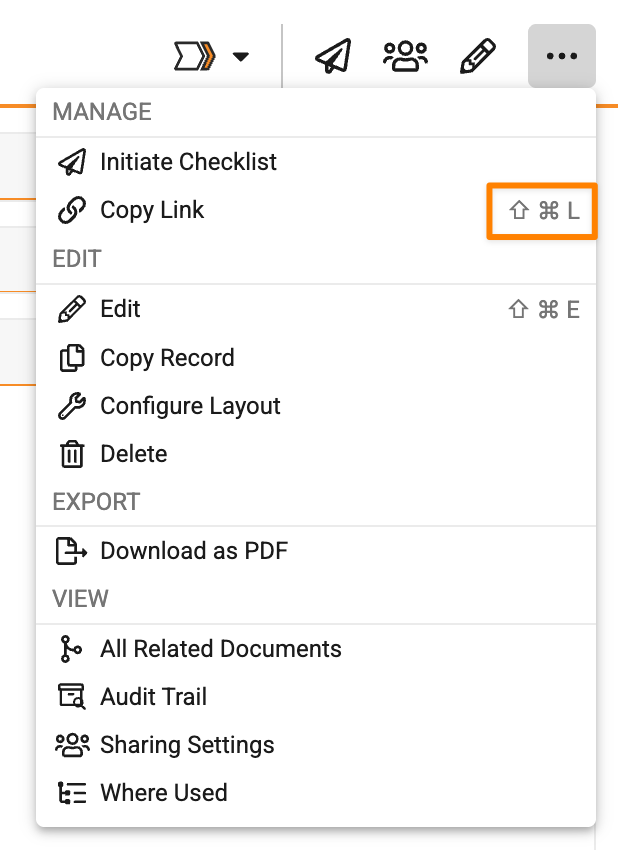
Copy Link Keyboard ShortcutAuto-on26R1.3
Users can now copy the link to a document or record using a keyboard shortcut, in addition to using the Copy Link option in the All Actions menu. This makes it fast and easy for users to copy a link to use elsewhere.
The keyboard shortcut is Shift + Ctrl + L (Windows) or Shift + Cmd + L (Mac).
Additionally, Vault displays this shortcut next to the Copy Link action:
For Vaults that don’t have Copy Link enabled on documents, this shortcut copies the URL as it currently shows in the browser.
Learn more about Vault Keyboard Shortcuts.
Timeout ImprovementsAuto-on26R1.3
When a user re-authenticates to Vault after a log out due to inactivity, they will now do so in a new window, maintaining the page they were actively working on. This makes it easier for users to immediately pick back up on their work, without having to re-navigate to the right place.
Additionally, when a user is working in multiple Vaults on the same domain in different browser tabs or windows, activity in any Vault will prevent timeout in all tabs and/or windows.
Note: This feature will be completed as part of the 26R2 general release, though it will not be available as part of the initial 26R2 release deployment. The feature will be enabled in all Vaults during the week of September 23. The feature will be available in pre-release.
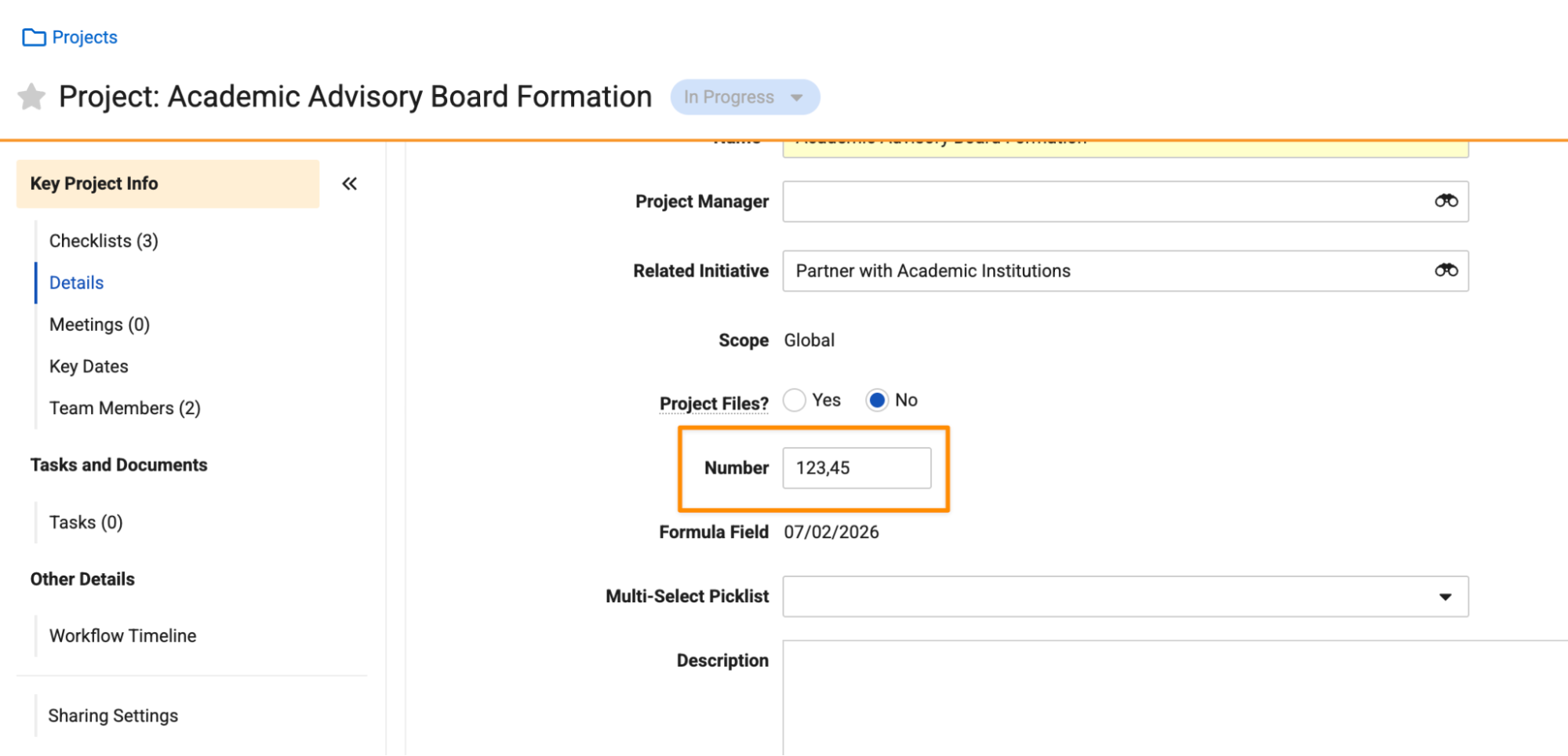
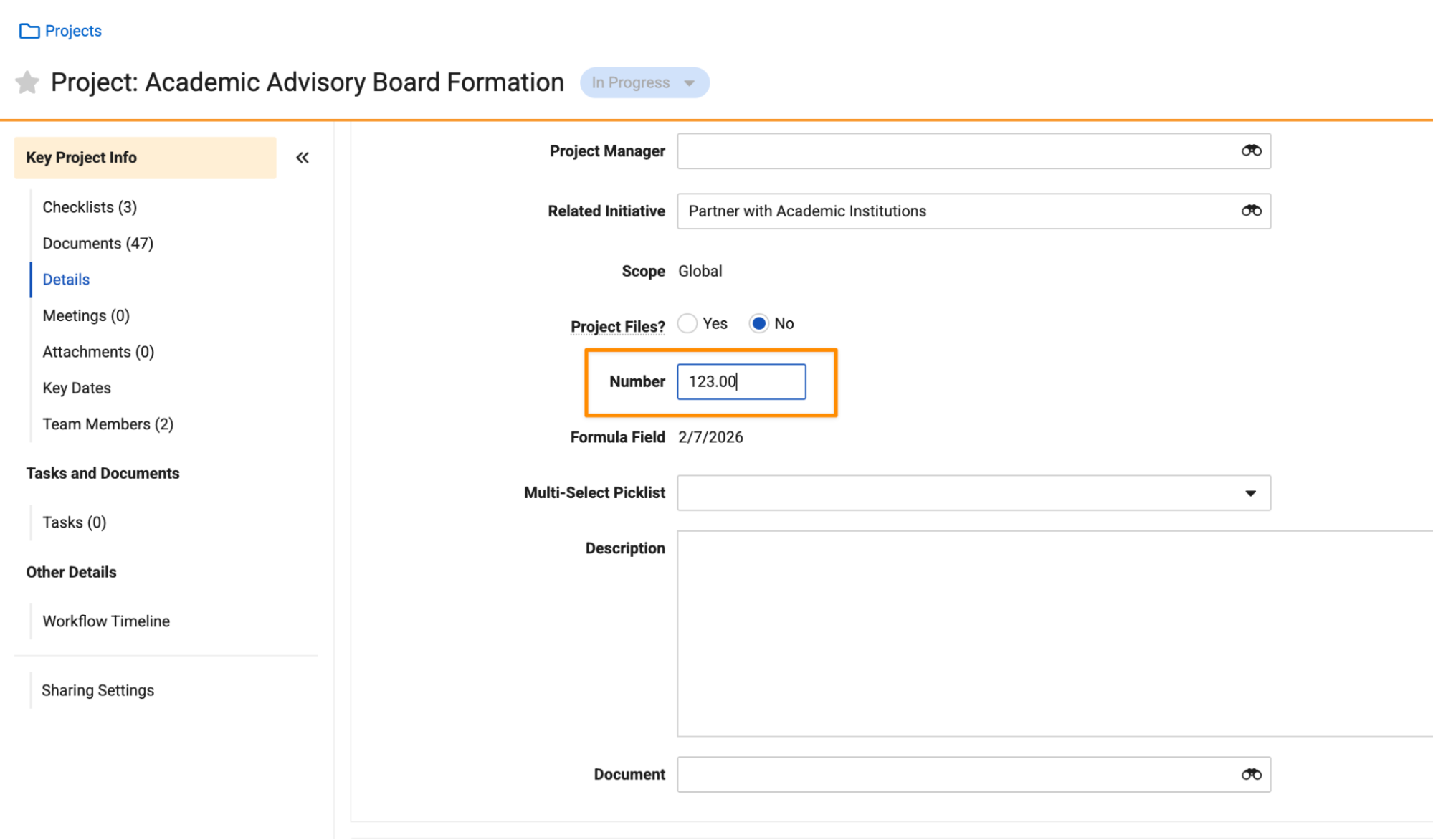
Improved Localization for Number FieldsAuto-on26R1.3
This feature leverages ICU (International Components for Unicode) formatting to ensure that numbers, currencies, and percentages are displayed and edited using the appropriate format of a user’s locale. By adopting these global standards for decimal and grouping separators, Vault now provides a more consistent and intuitive experience for viewing and editing numbers across the platform.
Prior to 26R2, Number fields were displayed to users based on their locale, but data entry did not respect their locale.
For instance, some locales use a comma as a decimal separator, while others use a period. Prior to 26R2, all users, regardless of locale, had to use a decimal. If a comma was entered, Vault would identify it as an invalid character.
Going forward, if a user’s locale uses a comma (based on ICU) as a decimal separator, Vault expects that character when entering numbers, not just when viewing numbers.
For example, a user with a France (French) locale would enter a number using a comma separator:
A user with a United States locale would enter the same number using a period separator:
Additionally, this feature ensures that digit grouping characters are displayed consistently based on all locales. For example, in the United States, one million is entered as “1,000,000”, while in Germany, it is entered as “1.000.000”.
This applies to Number fields across objects, documents, workflows, reporting, and Process Monitor.
Learn more about Language & Region Settings.
Process Optimization
Layout Profiles Supported in Action TriggersConfiguration26R1.2
Admins can evaluate Layout Profiles in a condition and use Update Records and Update Current Record Action Triggers to set a Layout Profile.
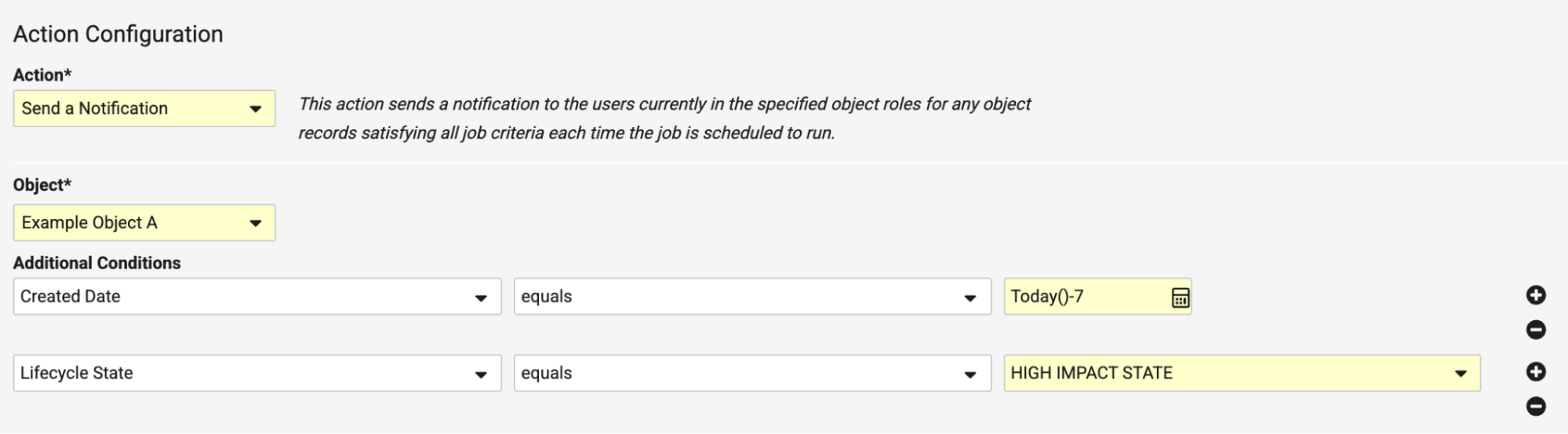
Relative Date Filters in JobsAuto-on26R1.3
Admins can now configure job definitions with dynamic date-based expressions on DateTime fields using Additional Conditions, enabling automated execution based on relative date filters in addition to manual, fixed-date updates.
Analytics
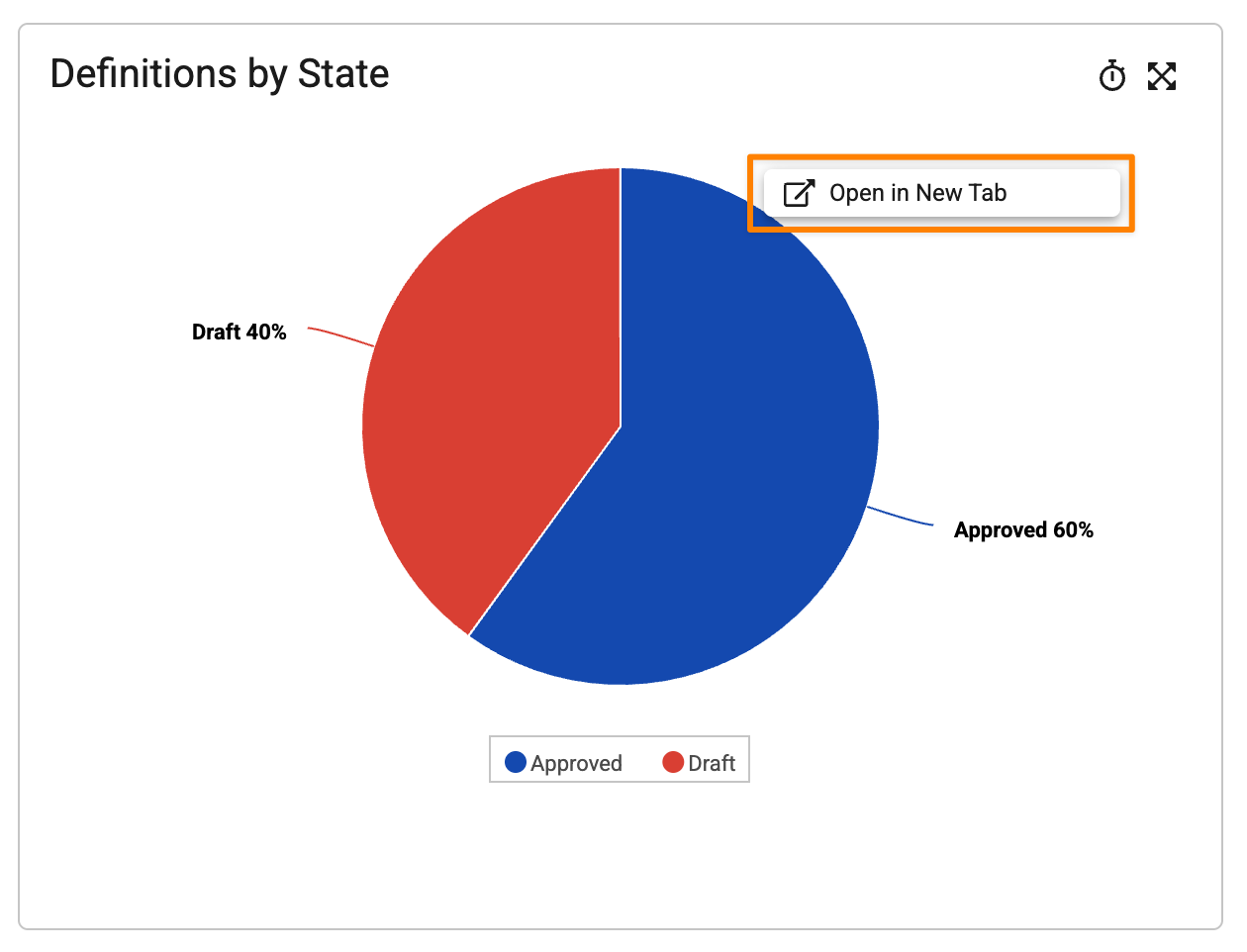
Open Chart in New TabAuto-on26R1.3
When viewing a dashboard, users can now right-click on charts to open the underlying report in a new browser tab:
This enhancement makes it easier for users to view the details of dashboard data while maintaining a browser tab with the full dashboard.
Learn more about Viewing and Sharing Dashboards.
Process Monitor: Saved ResultsAuto-on26R1.3
Process Views now run in the background and save your results, allowing users to navigate away from the page while data is retrieved. Prior to 26R2, if a user navigated away before a Process View finished running, they would need to rerun the view.
Process Views can often pull a large amount of data, and this enhancement ensures that users can move on to other work while the data is retrieved. Vault sends a notification once the Process View is ready to view.
The data is saved on a Process View for 24 hours. When users return to a Process View that has previously run in the last 24 hours, the Process View displays when the data was last updated:
When exporting data, if there have been no changes to the included columns since the last time the view ran, the export occurs synchronously. If changes have been made to the included columns, the export reruns the Process View and a notification is sent once the run is complete and the export is available for download.
Learn more about Process Monitor.
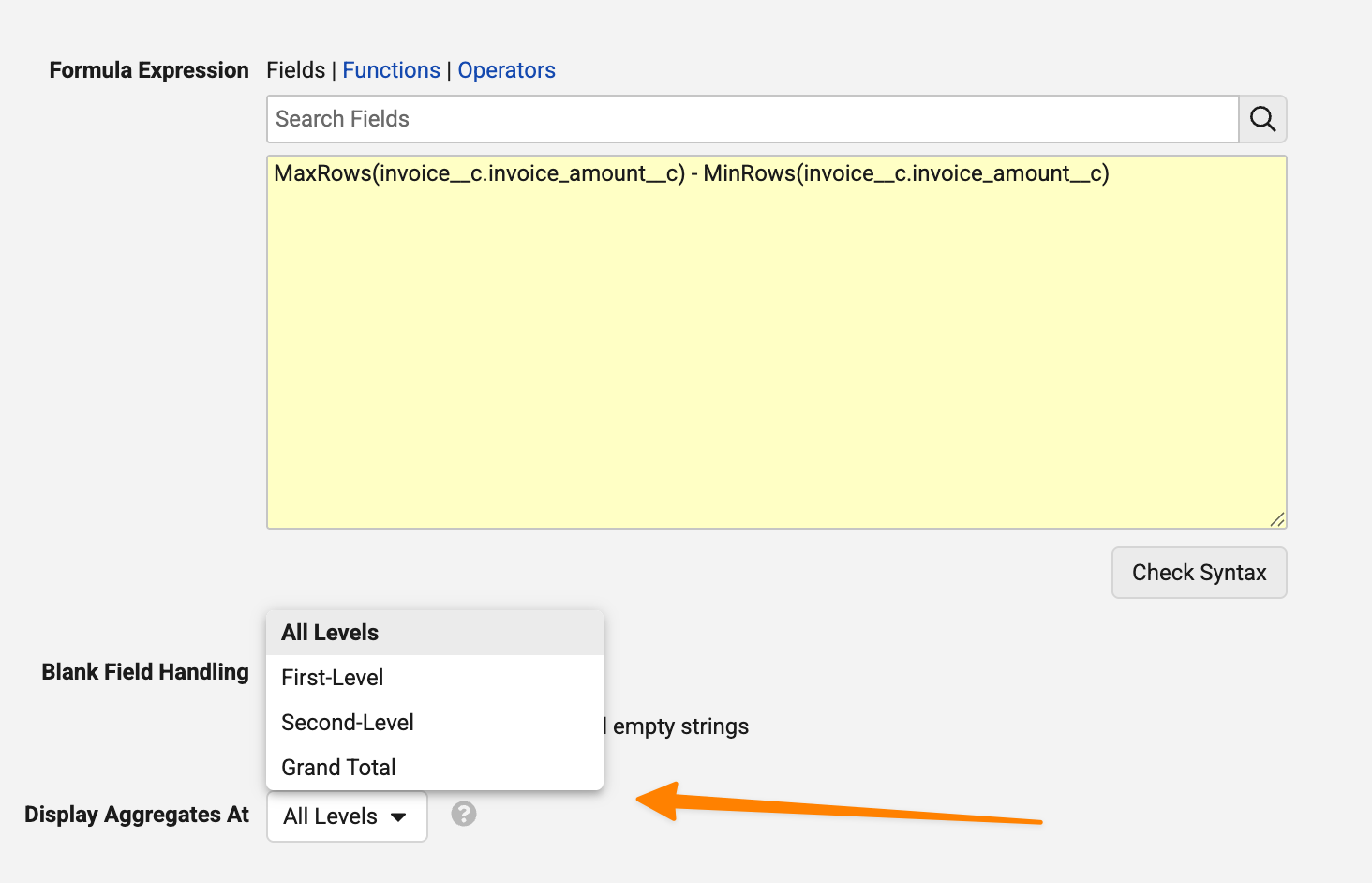
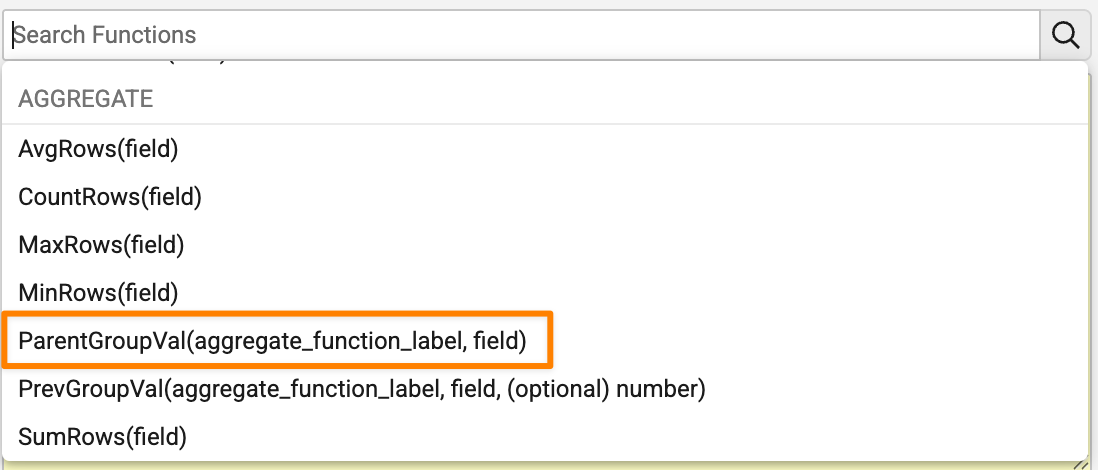
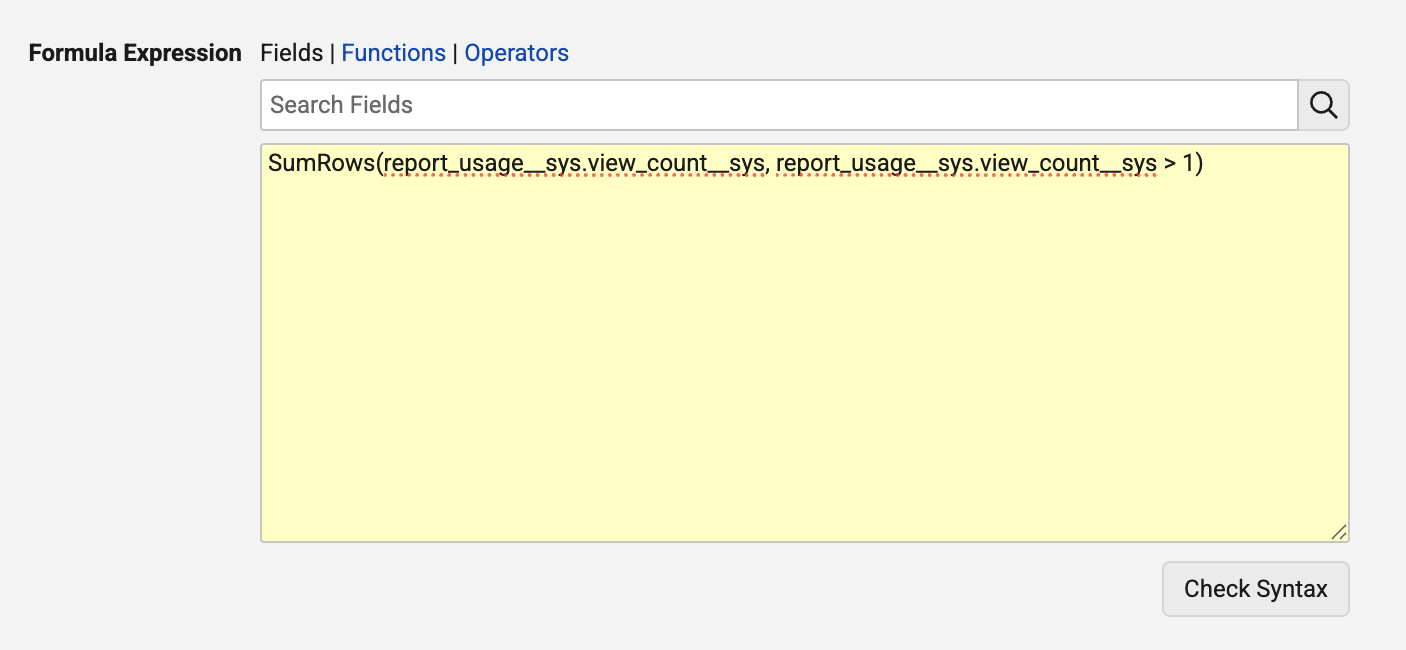
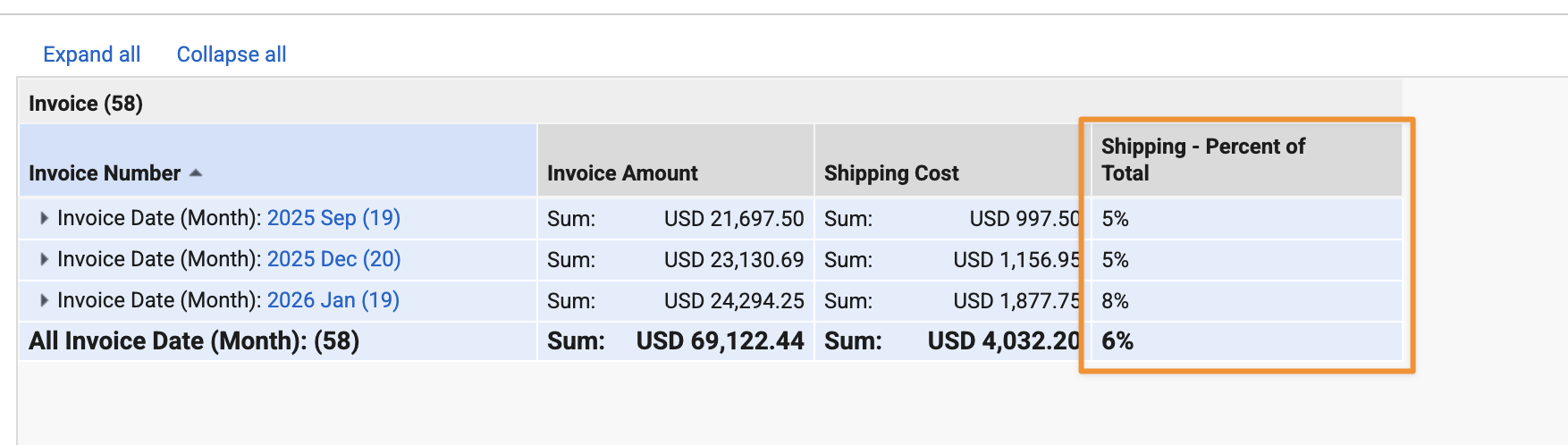
Enhanced Aggregate FormulasAuto-on26R1.3
A number of enhancements are introduced to the aggregate functions in report formulas functionality that was released in 26R1. These include:
- Aggregate functions are now supported in Matrix reports
- A new Display aggregate at option is available to determine which grouping an aggregate formula field should display on
ParentGroupVal()is added as a new function to support calculations across two levels of grouping. It is useful to calculate percentages within a group.
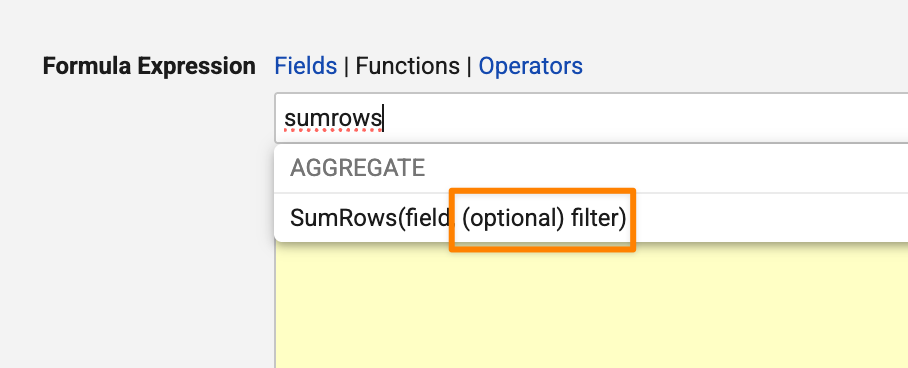
Aggregate functions now support an optional filter parameter. This allows you to conditionally include or exclude records from calculations based on a boolean expression. For example: SumRows(field, field>1) or CountRows(field, if(text(state__v)=Effective, true, false)).
MinRows()andMaxRows()now support Date and DateTime fields. Previously, only Number fields were supported.- Up to 15 aggregate functions can be used in a single formula field, with an overall limit of 30 aggregate functions across all formula fields. Previously, this limit was 3 per formula field.
CountRows()now supports all field types. Previously, only the Name field was supported.
These changes provide even more flexibility to report creators and editors in performing calculations on aggregated data.
Learn more about Aggregate Formula Fields.
Add Description to Report Formula FieldsAuto-on26R1.3
Report creators and editors can now add descriptions to formula fields, making it easy for other report users to understand these fields and their purpose within a given report.
The description is visible to report viewers by hovering over the formula field column name:
Learn more about Report Formula Fields.
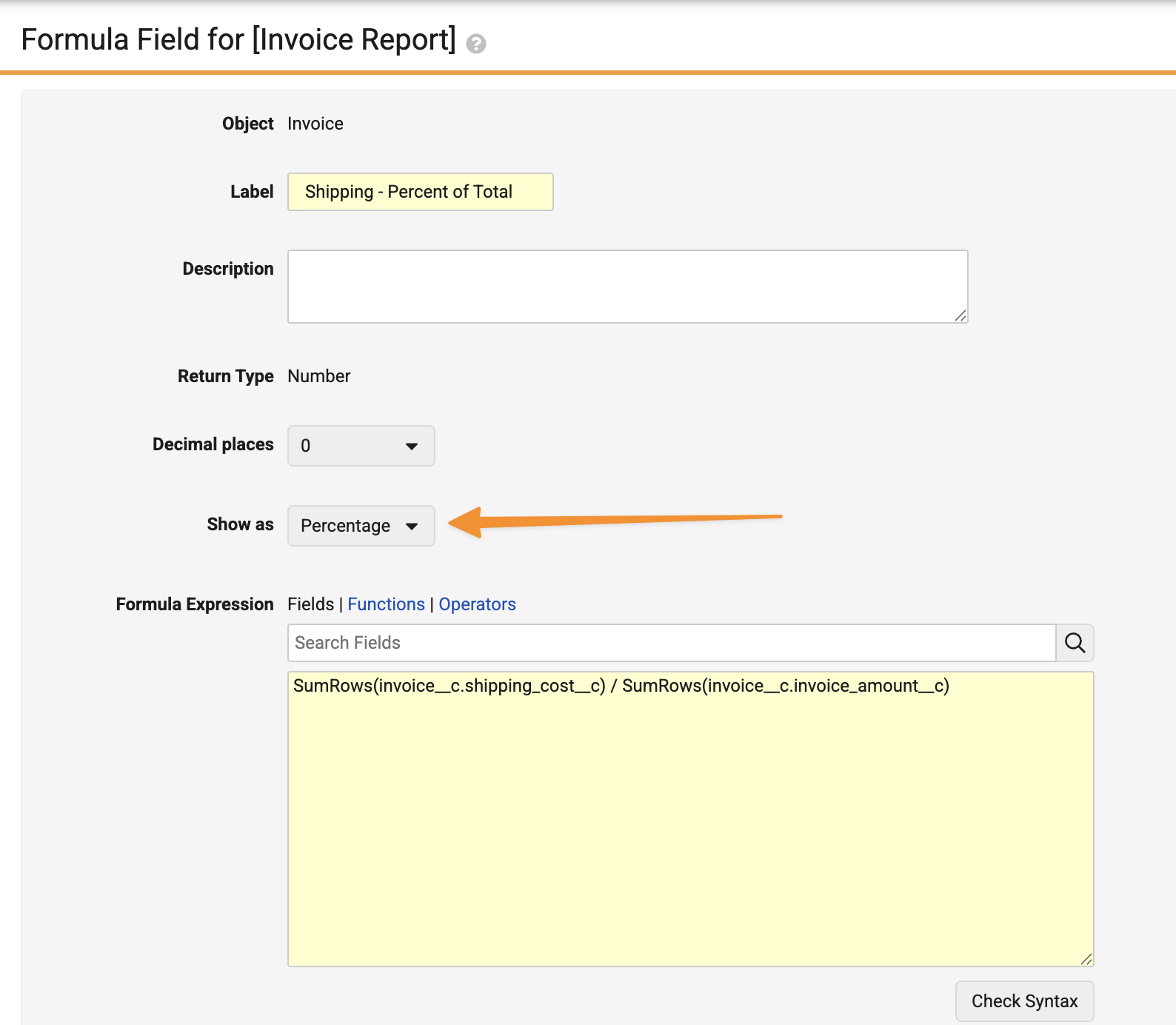
Show Report Formula Fields as a PercentageAuto-on26R1.3
When creating formula fields with a Return Type of Number, users can now choose whether to display results as a number or a percentage:
Learn more about Report Formula Fields.
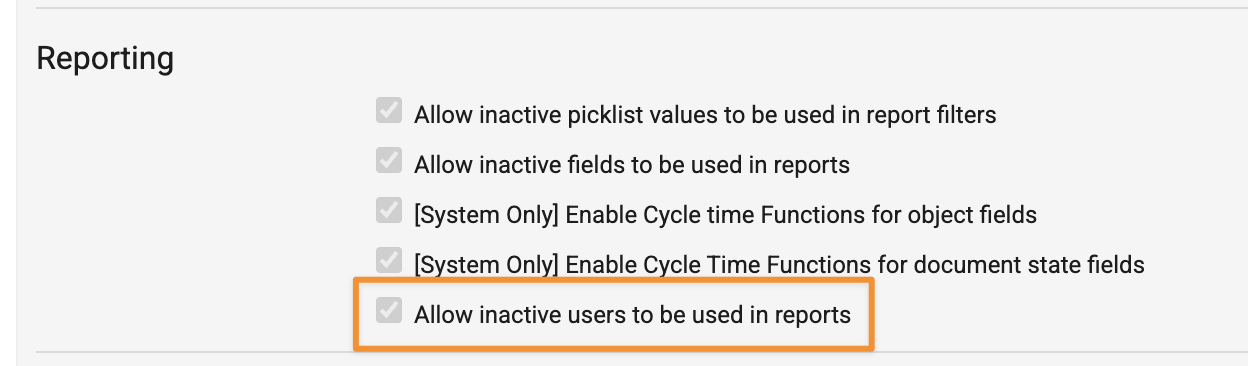
Exclude Inactive Users in Report FiltersConfiguration26R1.2
Reports can now exclude inactive users from being available in report filters and prompt dropdowns. This can be controlled for all reports with the Allow inactive users to be used in reports Admin checkbox in Admin > Settings > General Settings:
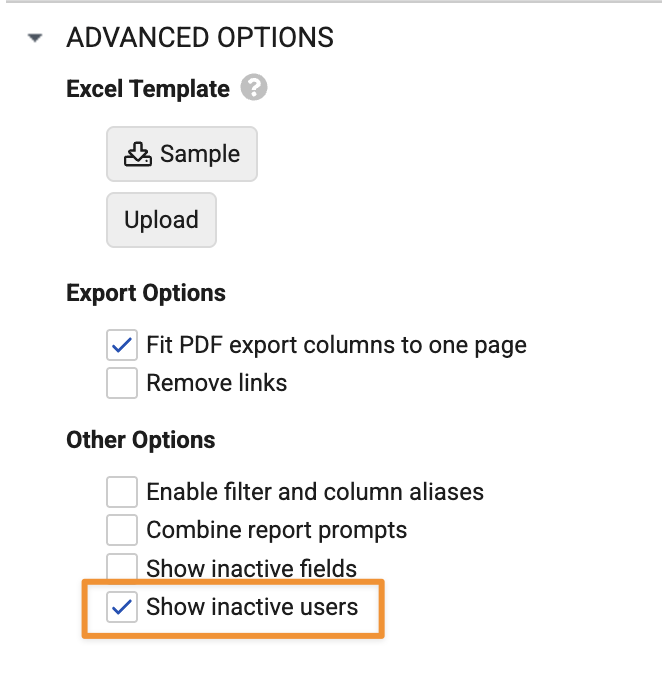
If enabled at the Vault level, users can still choose to exclude inactive users on a case-by-case basis using a new Show inactive users checkbox in Advanced Options when creating and editing reports:
Both checkboxes are selected by default to maintain current behavior.
Learn more about Creating Reports.
Permissions & Access
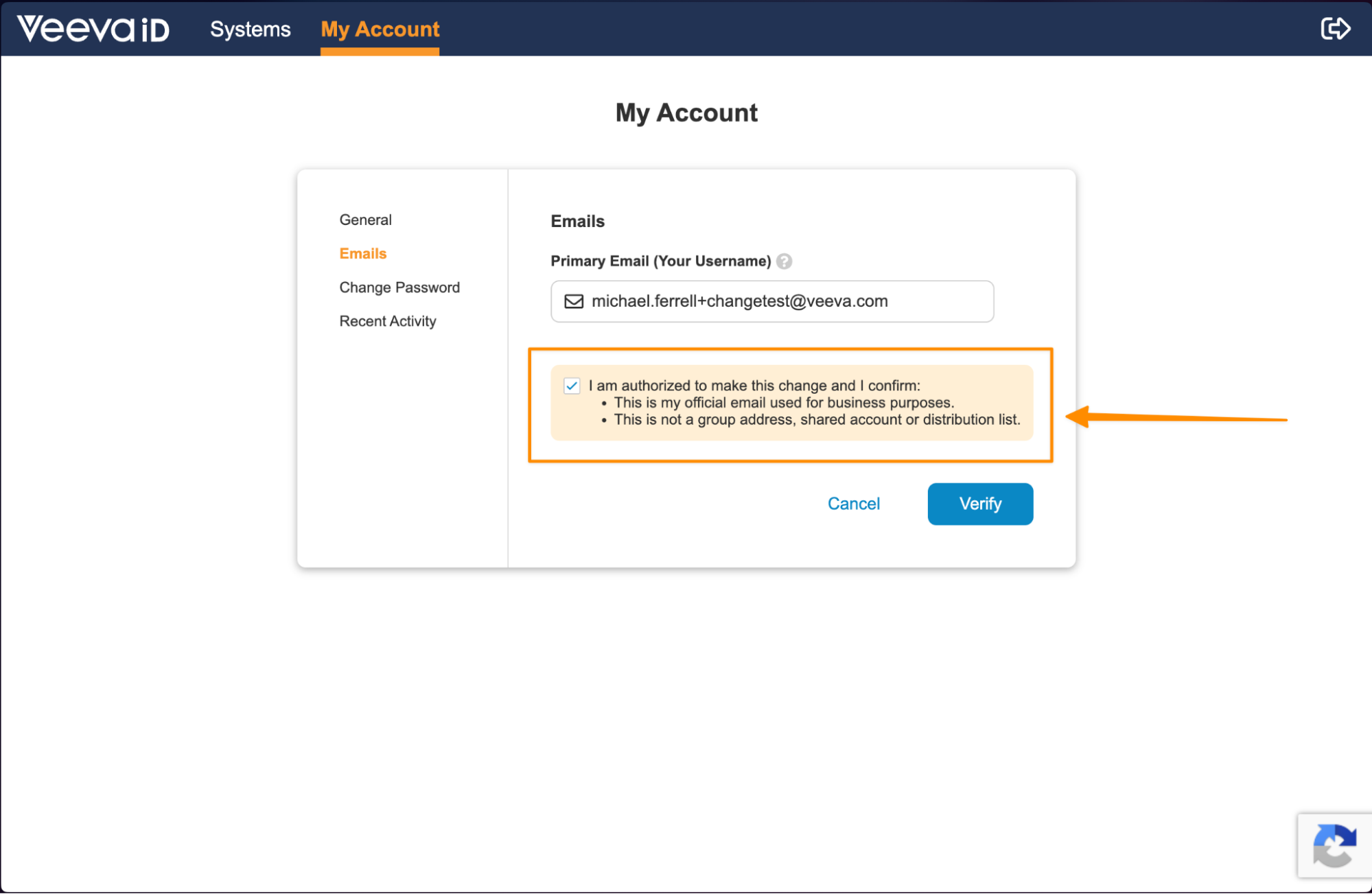
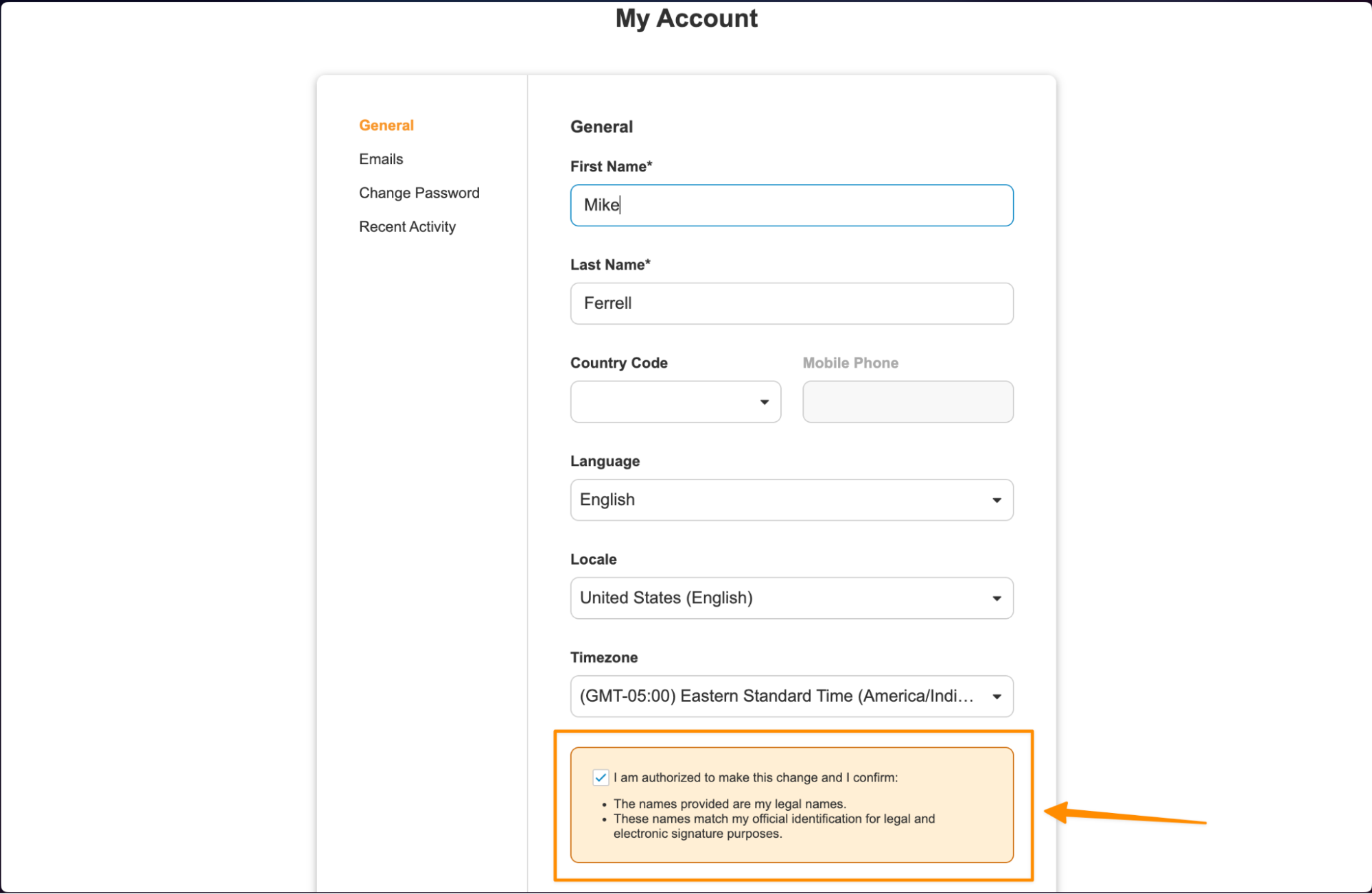
VeevaID Contextual Help for Personal Detail UpdatesAuto-on26R1.3
When VeevaID users update their personal details in the My Account tab, Vault now requires them to provide acknowledgement that the changes they are making are correct. This enhancement helps increase user awareness around potential downstream impacts of incorrect updates.
If the user is updating their email, they will acknowledge that they are providing their official business email, and it is not a shared email address:
When users update their other personal details, they will acknowledge the changes, and specifically that they are providing legal name information that matches official identification documentation:
Learn more about About VeevaID.
Enforce Atomic Security in Audit LogsAuto-on26R1.2
When a user is restricted from seeing certain fields via Atomic Security, any updates to these fields are hidden when the user views an individual object record audit trail. Prior to 26R2, users could still see information through the audit trail about fields that were restricted elsewhere via Atomic Security, which often meant that Admins would need to remove permissions to the audit trail altogether for these users.
This enhancement ensures users can have the ability to see record audit trails while respecting any applicable field-level Atomic Security. This enhancement does not apply to the Object Record Audit History in Admin > Logs, and applies specifically to object records (as field-level Atomic Security is not available for documents).
Learn more about Configuring Atomic Security on Fields and Viewing Audit Trails.
Admin Experience
API Access TokensAuto-on26R1.3
This feature allows users to use a long-lived API token to authenticate to the Vault API. This enables integration with tools including MS Entra Id SCIM as well as allowing authentication sub-processes in integrations to be replaced.
Tokens do not provide Vault UI access, are valid for one specific Vault, and grant the same permissions of the user who generated the API token. Each user can have up to 25 active API tokens.
Only users who have API permission can create tokens. Maximum API token expiry duration is set in Security Policy.
Query ProfilerAuto-on26R1.2
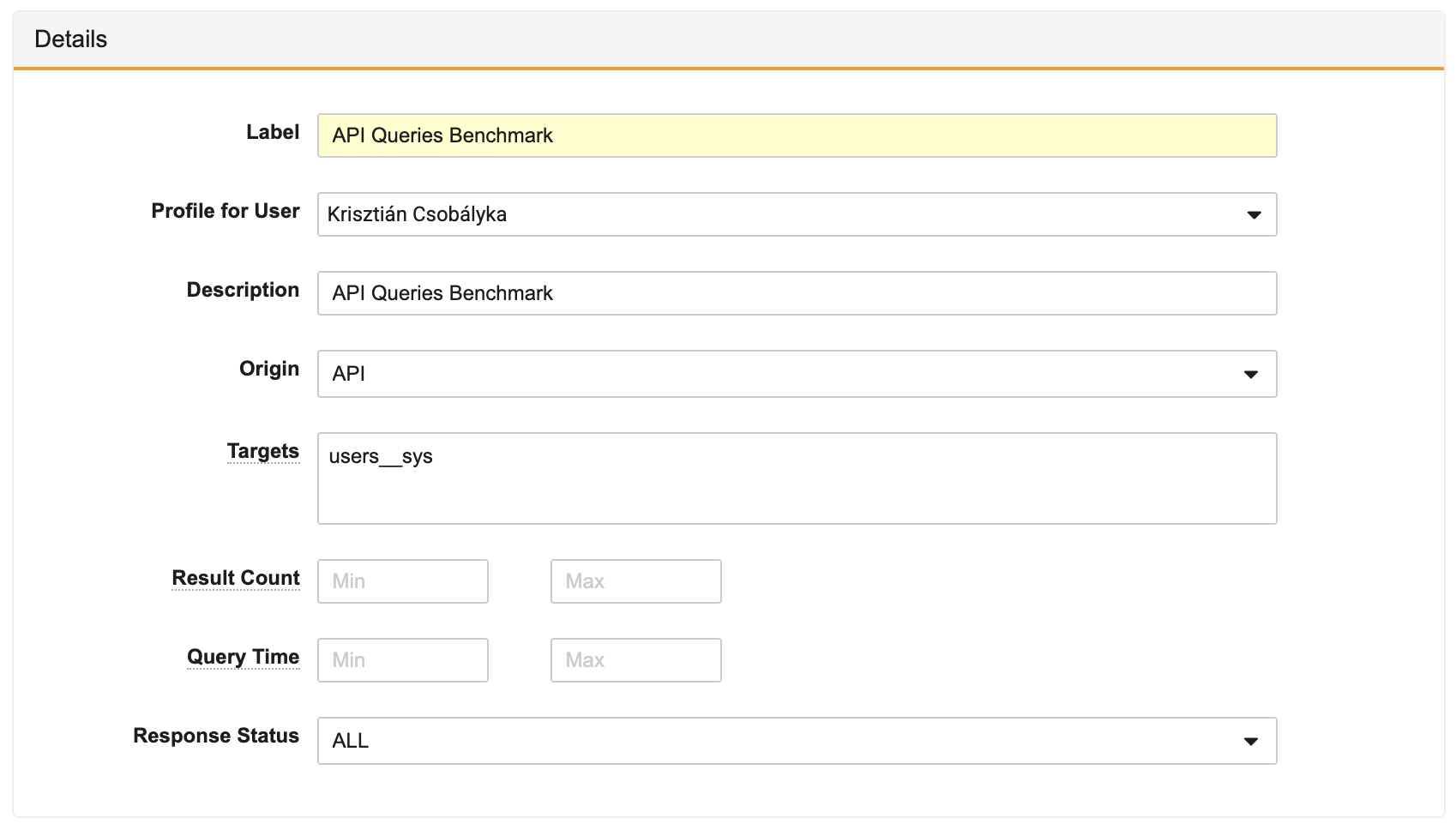
This feature allows Admins and Developers to monitor VQL query performance with the new query profiling tool. Analysing the profile of queries is a key part in monitoring their performance and overall health, but this process has been challenging in the past.
With Query Profiler, the results make it easy to analyze the queries running in Vault by looking at the data obtained during a profiling session. Users must start a profiling session where they are also able to set filters on what kind of VQL queries should be included in the results. These filters include:
- Origin: Filters where the VQL query originated from (API, SDK, or ALL).
- Targets: Filters the specific Query Targets of the VQL query (for example, restricting to queries on user__sys).
- Result Count: Filters queries to only be included if their result count is in a minimum or maximum range.
- Query Time: Filters queries to only be included if their execution time is in a minimum or maximum range.
- Response Status: Filters whether the query was a SUCCESS, FAILURE or if it received a WARNING.
Learn more about Viewing Admin Logs.
Record Migration Mode InheritanceAuto-on26R1.3
When records are created, updated, or deleted in Record Migration Mode, it can cause Vault to create, update, or delete other records within the same process. Vault treats these related record activities as part of the migration transaction, and thus the related records will inherit many of the same migration mode behaviors. In Object Record Audit History, Vaults displays “in migration mode” in the description for related records which inherited the migration mode.
Record Migration Mode for Delete OperationsAuto-on26R1.3
Vault now supports Record Migration Mode for delete operations and the No Triggers option. This feature allows users to delete migrated records without executing record triggers and Action Triggers. Blocking references will still prevent record delete operations even in migration mode.
This feature is supported for both API and Loader.
Layout Profile: Summary Dialog for Adding & Removing UsersAuto-on26R1.2
When adding or removing users from a Layout Profile, Vault now displays a summary dialog if any users were skipped due to an error. If all users were successfully added or removed, Vault continues to display a success banner instead.
Direct Data API: Additional Document Metadata in Workflow ExtractsAuto-on26R1.2
Workflow Item and Workflow Task Item extracts will include additional document metadata information. The field additions will be captured in the incremental metadata.csv (and correspondingly in the metadata_full.csv).
Workflow Item extract includes additional document fields to maintain the stamped document metadata for expired workflows:
document__sysdocument_version__sys
Workflow Task Item extract will include additional document fields to maintain the stamped document metadata for expired workflows:
document_id__sysverdict_document_major_version_number__sysverdict_document_minor_version_number__sysverdict_document_version_id__sys
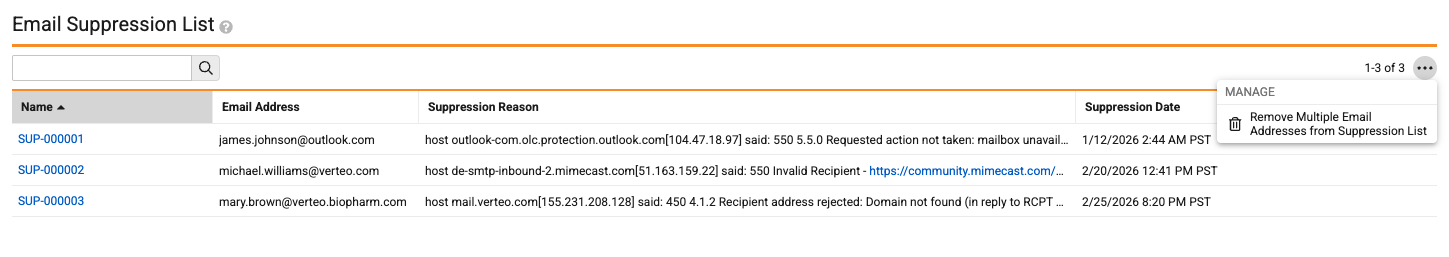
Notifications: Remove Multiple Email Addresses from Suppression ListAuto-on26R1.3
Admins can now remove multiple email addresses from the Email Suppression List, rather than initiating the action on each individual record.
While this makes the experience more efficient for Admins, it is important to note that removing an email address from the Email Suppression List does not prevent it from returning to the list if there are future email failures for that address.
Learn more About the Email Suppression List.
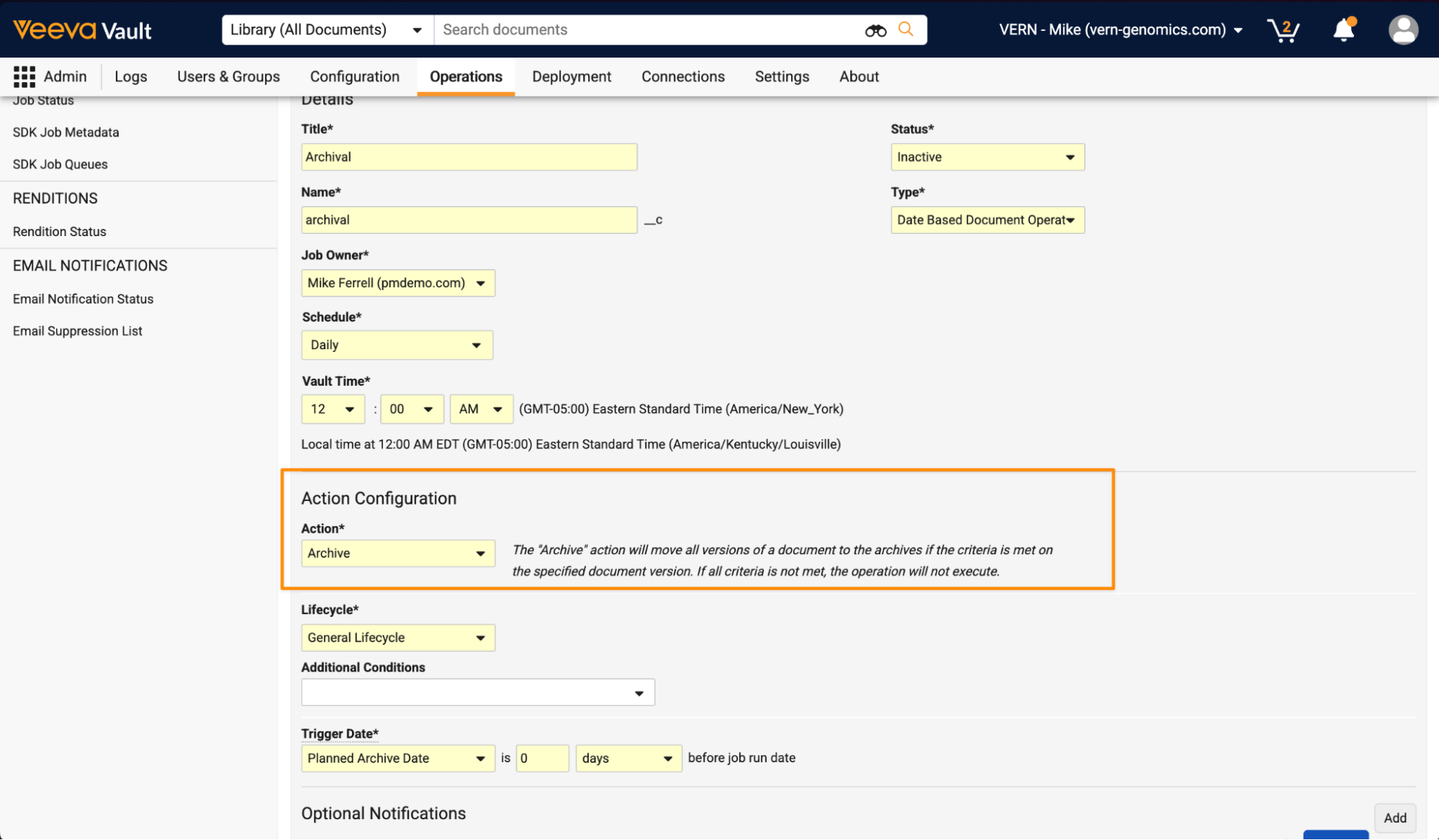
Document Archive JobAuto-on26R1.3
Admins can now configure jobs that automatically archive documents, supporting use cases where archival of documents should be automated based on document metadata.
When configuring a Date Based Document Operation job, a new Archive action is available:
This option is available on document jobs for Vaults where Document Archiving is enabled.
Learn more about How to Define Document Operation Jobs.
Packages Support for Record Attachments & Attachment FieldsAuto-on26R1.3
Vault configuration migration packages now support exporting and importing record attachments and attachment field files when exporting data.
After adding the Data Step (Dataset), you can edit the Dataset Item to enable Include Object Record Attachments to export record attachments. In the Columns sections of the Dataset Item, you can add attachment fields to include their attachments in the extract.
There is a 10MB per record attachment, and 100MB overall limit when exporting VPK.
Learn more about Using Configuration Migration Packages.
Connection Authorization EnhancementsAuto-on26R1.3
This feature enhances the Connection Authorization workflow, specifically focusing on the input methods for the API Key and Client Secret Auth Types.
- API Key Input Field Type and Length: The
api_key__sysfield is now a Long Text field with a 3600-character limit. - Single Input for API Key and Client Secret: This feature moves to a single-input model. Previously, Admins were forced to repeatedly input these encrypted fields while storing them in Vault.
- Updated visibility while providing input: A Vault Admin should be able to temporarily view the inputted value for all encrypted fields before saving. This is true for
api_key__sys,client_secret__sys, andpassword__sys.
Migration Package Validation EnhancementsAuto-on26R1.3
Migration Package Validation improves the reliability of configuration deployments by enforcing SDK code compilation checks and detecting missing sub-component dependencies during the validation phase.
Vault now compiles and validates SDK source code included in a Vault Package (VPK) during the validation step. If the code contains compilation errors, the validation process marks the specific code step with a Blocked status. This prevents the package from being deployed until the code issues are resolved.
Vault now performs enhanced dependency validation checks for missing sub-components. Even if a parent component (for example, an object) exists in the target Vault, the engine verifies that all referenced sub-components (for example, fields) are also present.
Performance & Availability
Bitmask FieldsAuto-on26R1.3
This feature introduces a Bitmask field type for objects, enabling Vault Applications to securely filter records efficiently without performance degradations at high volumes. This new field type will be used primarily for raw objects. Only Veeva can create Bitmask fields which also support a new bitwise operator in VQL for raw object records.
Vault Loader Performance EnhancementsAuto-on26R1.3
Vault Loader jobs now leverage the distributed job framework, which leads to faster load times for Vault datasets.
This behavior means there could be a change in the order in which the datasets are loaded as a later batch (for example, batch 3) may process faster than an earlier batch (for example, batch 1).
Processes leveraging the sequential loading of records via Vault Loader will need to process the batches of 500 records manually.
Minor Enhancements
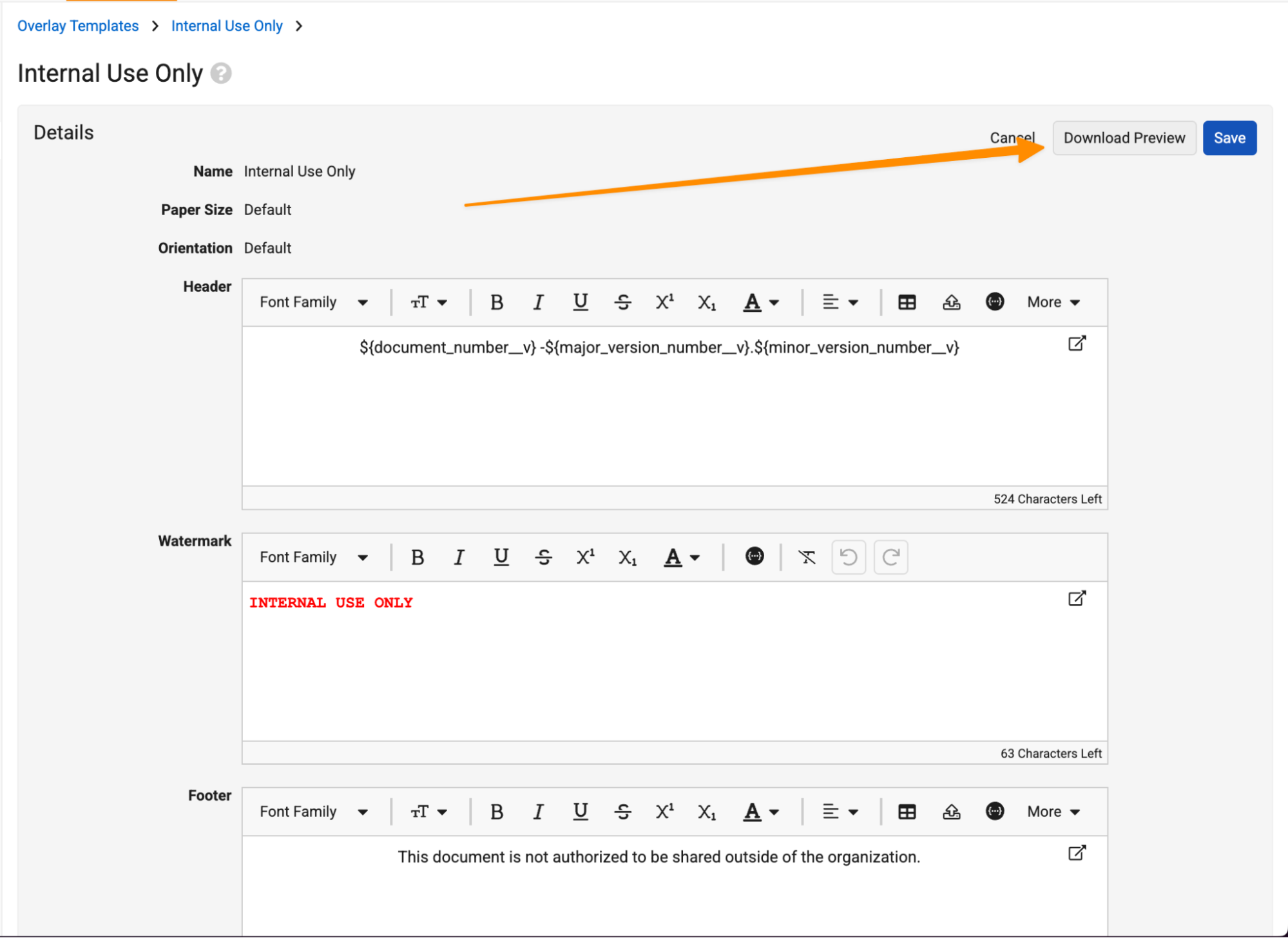
Preview for Overlay & Signature Page TemplatesAuto-on26R1.3
When editing overlays and signature page templates, Admins can now download a preview before saving their changes. This makes it easier for Admins to iterate on templates without committing those changes until all updates are generated as desired.
Learn more about Managing Overlays and Managing Signature Page Templates.
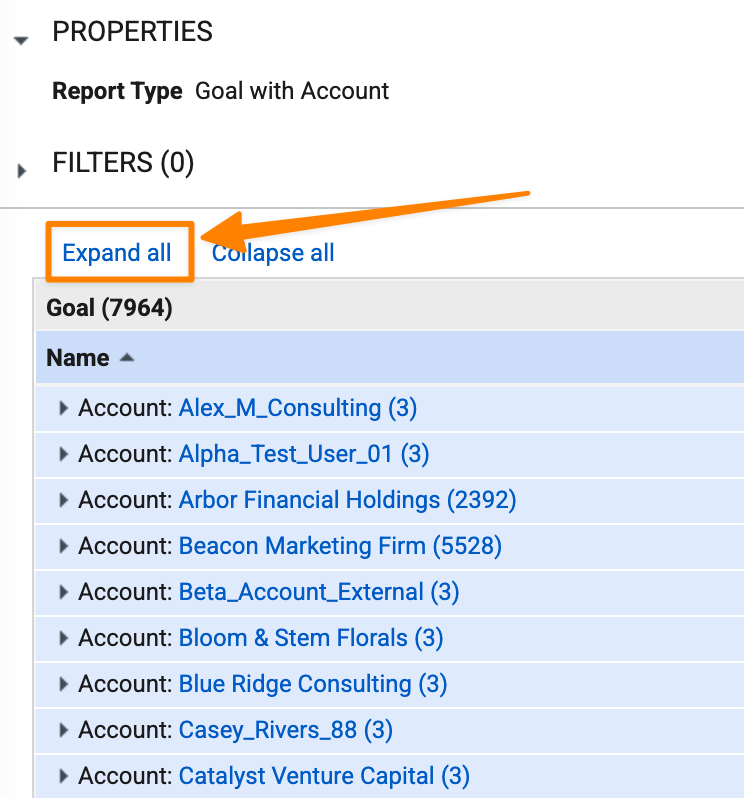
Enhanced Expand AllAuto-on26R1.3
The Expand all option is now available for users to expand all groups in reports with more than 2,000 records.
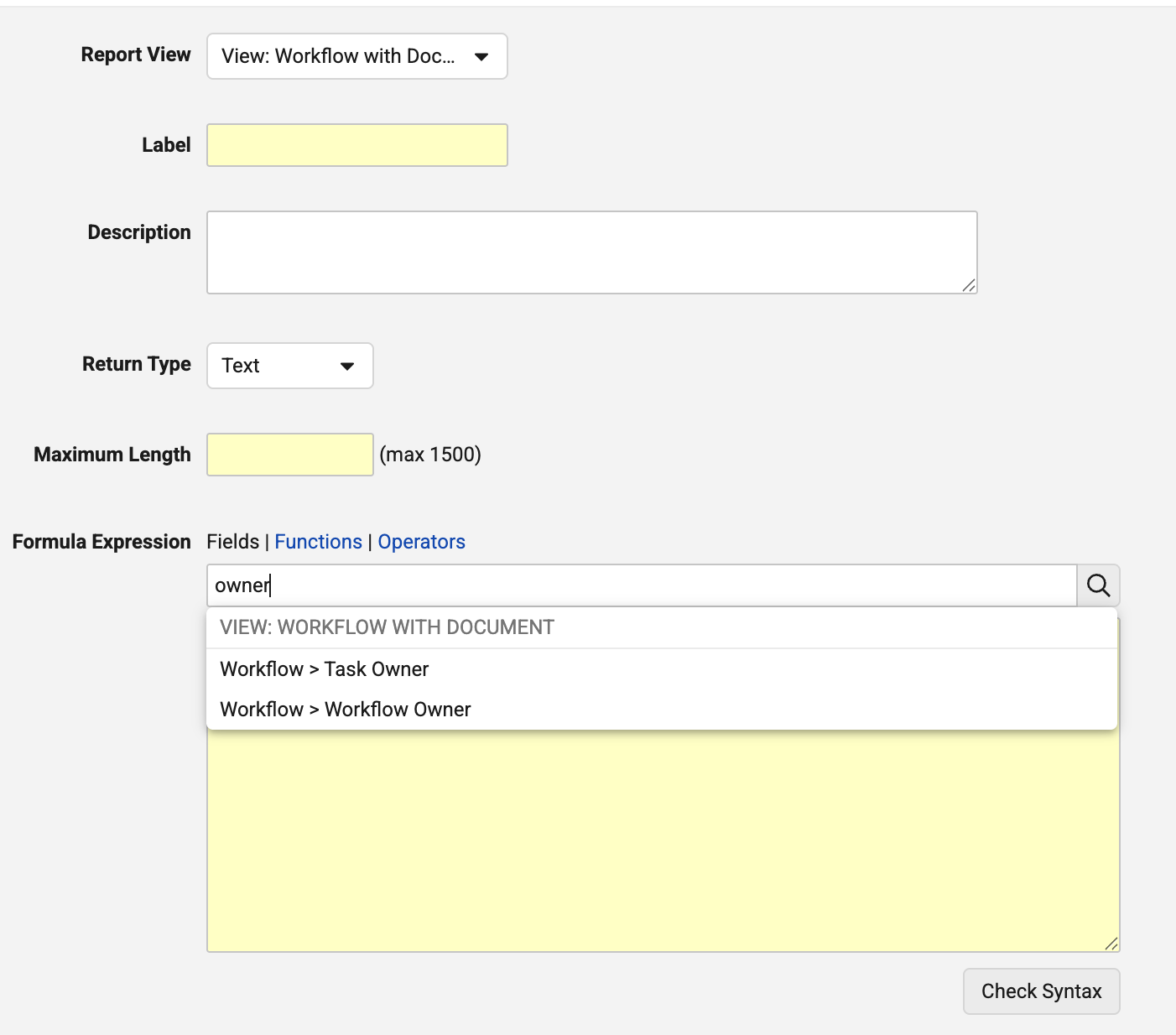
Support Workflow & Task Owner in Report Formula FieldsAuto-on26R1.3
When using formula fields on Multi-Pass reports that include Workflow with Document or Workflow with Object report views, users can now use Task Owner and Workflow Owner in formula fields:
This enhancement provides greater flexibility and ensures that report creators can use all workflow fields in formulas.
Learn more about Report Formula Fields.
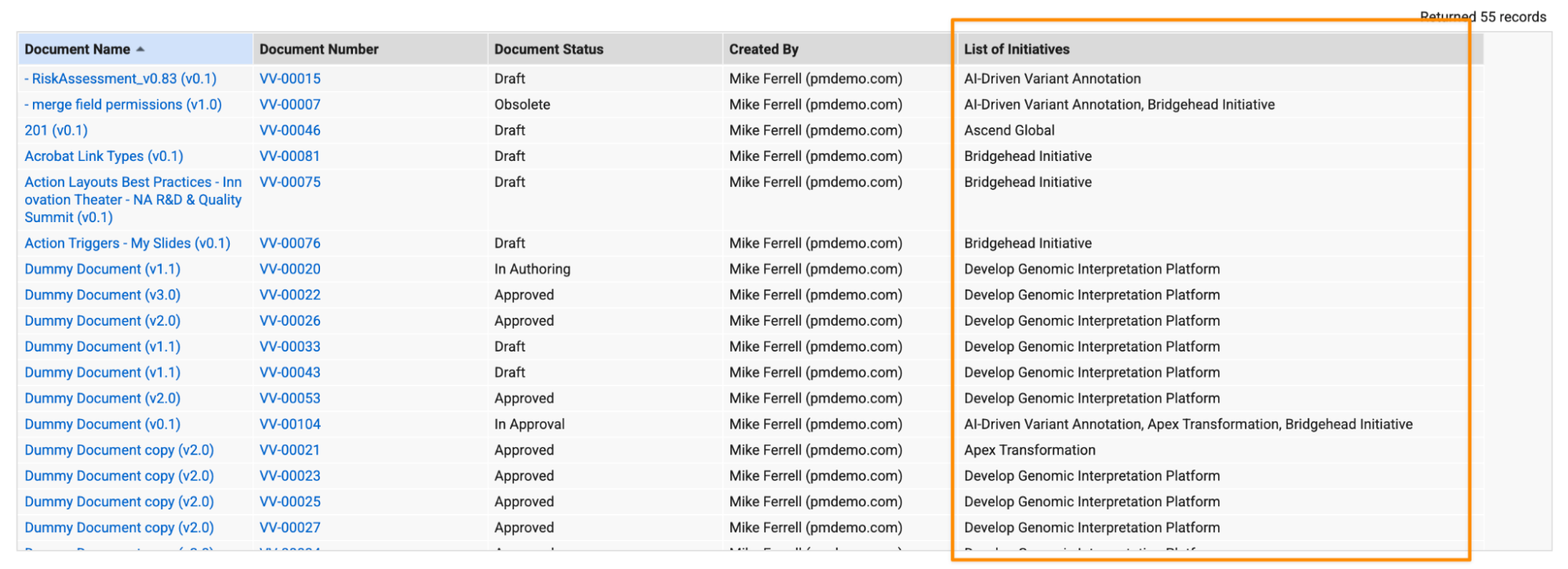
Support Multi-Object Reference Fields in Document Report FormulasAuto-on26R1.3
In Document reports, multi-object reference fields can now be used in formula fields with the Text() function. This converts the values in that field into a comma-separated list. This provides greater flexibility and reduces user confusion when creating and editing formula fields. Previously, only single-value object reference fields were available for selection in the formula editor.
Learn more about Report Formula Fields.
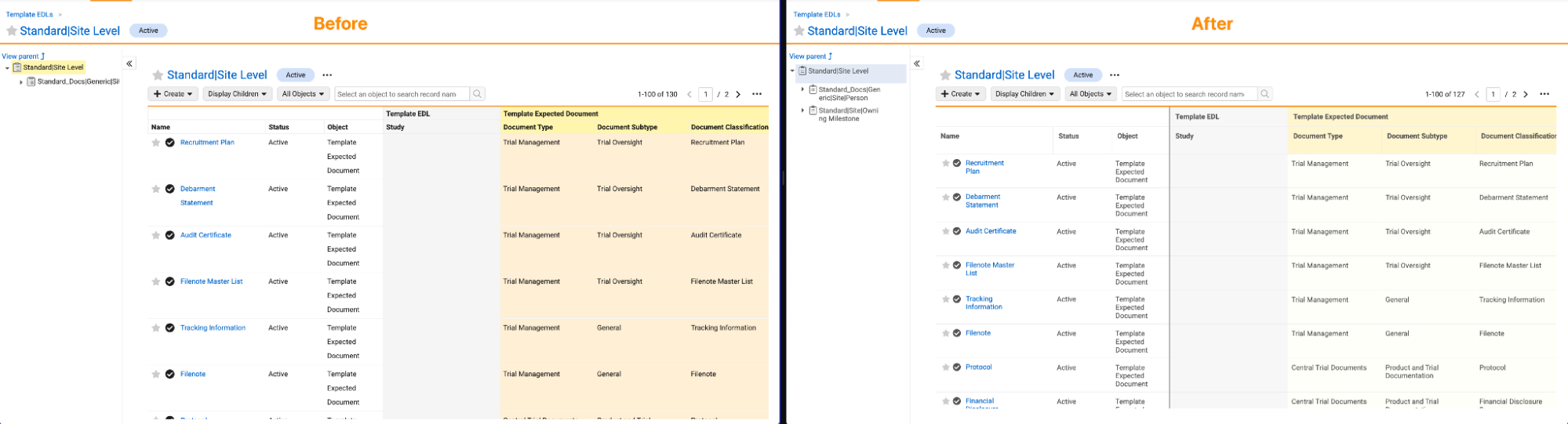
EDLs: Consistent Hierarchy ViewerAuto-on26R1.3
The EDL Hierarchy Viewer now provides a consistent user experience across Expected Document Lists (EDLs), EDL Items, EDL Templates, and EDL Item Templates. In many cases, a more modern viewer is already in use based on application-specific EDL functionality. This change ensures that the experience is consistent everywhere.
In Clinical Operations Vaults and RIM Vaults, this is applicable to the viewer when working with EDL Templates:
The changes include freezing columns and changes to the coloring.
When working with EDLs and EDL Items directly, there are no changes to the experience in Clinical Operations Vaults and RIM Vaults as these applications have already implemented the more modern viewer (such as the Content Plan Viewer in RIM Vaults).
Learn more about EDL Administration.
Download as PDF: Deduplicated Records in Related Join Object SectionsAuto-on26R1.2
When using the object record Download as PDF functionality, the generated PDF now consolidates duplicate records in the same manner as the Vault user interface.
This functionality applies when duplicate records are associated via simple join relationships or complex join relationships configured to display as simple.
Learn more about Download as PDF and About Object Relationships.
File Count Validation for Record AttachmentsAuto-on26R1.2
Vault now informs users that they cannot drag and drop more than 100 files in a record detail page. Attempts to do so triggers an error and cancels upload for all files.
Add API Name to Document Type Details ObjectAuto-on26R1.2
A new Document Type Name (document_type_name__v) field is added to the Document Type Details (doc_type_detail__v) object. This provides the public API name of the mapped document type, whereas previously only the document type’s label was available in the object record.
Vault AI
Bring Your Own Model: Google Gemini SupportAuto-on26R1.3
Starting in 26R1.3, Admins can configure custom Large Language Model (LLM) connections using Gemini 2.5 Flash and Gemini 2.5 Pro models hosted on Google Cloud. These connections can power custom agents in Vault AI.
To ensure safe Vault AI interactions, this integration fully supports Google Model Armor. Admins can define specific Model Armor templates for both prompt and response screening within Vault AI Settings.
Agent Configuration Enhancements 26R1.3
Vault 26R1.3 introduces several enhancements to agent configuration, providing Admins with greater control over agent accessibility and document context retrieval:
- API Access Control for Agent Actions: Admins can now use the new API Access setting to control access via REST APIs.
- Viewable Rendition Selection for Document Context: When configuring Document Data and Content or Related Document Data and Content agent context types, Admins can include the Viewable Rendition as part of the agent action’s context.
- Document Version Selection in Standard Agent Contexts: Admins now have more precise control over which document version is retrieved when executing an agent action. The Document Version and Source Document Version settings allow agent context configurations to specifically target either the selected document version, or a previous major version of the document.
Learn more about Configuring Custom Agents.
Vault AI MetadataAuto-on26R1.3
Vault AI Metadata is a new configuration layer that provides agents a semantic understanding of a Vault’s data model. Previously, agents reasoned primarily over physical component names, such as product__v and start_date__v, which lack the business context, synonyms, and descriptive detail needed for accurate query generation and natural-language interpretation.
Subject matter experts can now describe the meaning of objects, document types, fields, relationships, metrics, and picklists in business terms through a new set of MDL components: Vsmentity, Vsmfield, Vsmrelationship, Vsmmetric, Vsmpicklist, and Vsmvalue. Active components are automatically synchronized to a vector store and made available to agents.
An Admin can view and manage these component records in the new Vault AI Metadata section in Admin > Configuration > Vault AI Metadata. Customers can configure custom component records in addition to the standard records.
Document Version Compare AgentConfiguration26R1.3
The Document Version Compare Agent is a platform-level agent that helps users quickly grasp key changes between two document versions at a high level, and the overall significance of those changes. By comparing the selected version against the previous major version, it surfaces a concise, high-level summary of what has changed - providing users the context needed to make informed decisions about version updates. Users can execute this agent as a standalone agent via Vault AI Chat or directly from the document viewer when comparing versions.
Veeva Connections
Clinical Operations-EDC Connection
Clinical Operations-EDC Connection: Improved Error Handling for Protocol DeviationsAuto-on26R1.2
The Clinical Operations-EDC Connection now introduces granular error isolation for Protocol Deviations. Previously, a single Protocol Deviation containing an unmapped value could generate a Message Processing Error (MPE), causing the entire batch to fail across studies. Now, If an EDC Protocol Deviation contains a Category or Subcategory value that is not mapped in CTMS, Vault processes all other valid records in the batch successfully. Vault also generates a detailed User Exception Item (UEI) specifically for the affected record, allowing you to quickly identify and resolve mapping gaps.
Learn more about other new Clinical Operations features below.
Clinical Operations-EDC Connection: Restricted Subject Visit CreationAuto-on26R1.2
Previously, Clinical Operations users faced data gaps because Unblinded Subject Visit records in Clinical Operations Vailts were only created in case of discrepancies between the Restricted and the Unrestricted details of the Event in EDC (such as status, SDV, etc.), potentially missing key restricted events.
To resolve this, Vault now automatically creates Unblinded Subject Visit data every time a restricted event occurs in EDC, regardless of data differences.
By simplifying the logic, this feature improves data completeness and increases efficiency reducing the need for manual verification.
Learn more about other new Clinical Operations features below.
Clinical Operations-EDC Connection: Additive SDVAuto-on26R1.3
Clinical Research Associates (CRAs) often perform Source Data Verification (SDV) on additional data points beyond those identified in the Review Plan for a given subject, due to emerging risks, such as site staff attrition or frequent errors.
Previously, there was no automated way to report on this additional effort in Veeva CTMS, requiring manual tracking and leaving Study Managers without visibility into how often these reviews occurred.
To streamline this process, the Clinical Operations-EDC Connection now automatically transfers a summary of Additive Source Data Verification information at the Subject Visit level, specifically indicating whether the review was performed, the latest reason, and the date.
By eliminating the need for manual tracking or double-entry, this solution significantly increases data quality and consistency and operational efficiency for CRAs and ensures proper recognition for monitoring work while providing Study Managers with the transparency needed to easily identify site risks and monitoring trends.
Learn more about other new Clinical Operations features below.
Clinical Operations-Vault CRM Connection
New Clinical Operations-Vault CRM ConnectionConfiguration26R1.3
The new Clinical Operations-Vault CRM Connection bridges the gap between clinical study and commercial teams by automatically sharing Healthcare Professional (HCP) interactions between Veeva Clinical Operations and Vault CRM. This cross-functional transparency ensures a coordinated site experience, improves communication, and eliminates data silos.
The Clinical Operations-Vault CRM Connection includes the following features:
Bi-Directional Data Sharing
Clinical Operations to Vault CRM:
- Core study data (Study, Study Countries, and Study Sites) in Veeva Clinical are auto-created in Vault CRM, synchronizing data and eliminating manual entry.
- Vault CRM users can monitor high-level study statuses or follow hyperlinks to view study information (such as enrollment details or milestone dates) directly in Veeva Clinical.
- When clinical activities, discussions, and attendees are logged in Veeva Clinical by study team members, they automatically flow into Vault CRM as read-only Calls and Discussions. This gives MSLs immediate visibility into clinical interactions.
Vault CRM to Clinical Operations:
- When users log interactions or medical discussions in Vault CRM, this data is transferred to Clinical Operations as read-only Medical Activities, Discussions, and Attendees. This ensures study teams can view relevant field communications with their study site staff.
Intelligent Matching
The system uses customer-designated unique identifiers (such as VeevaID or OpenDataID) to accurately match Account records in Vault CRM with Person records in Veeva Clinical. Data flows only when a valid match is identified.
We’ve also included regional country mapping. To prevent spamming of region-specific CRM instances with irrelevant data, the connection uses a country connection mapping framework where interactions are filtered based on the HCP’s primary country.
Multiple CRM Instance Support
Since Vault CRMs are often country or region specific while Veeva Clinical is global, customers can configure and manage concurrent active connections between a single Clinical Operations Vault and multiple Vault CRM instances. Connection details can be easily copied to set up a new connection with another Vault CRM instance, reducing administrative burden.
Error Handling and User Exception Messages (UEMs)
To simplify troubleshooting, the system surfaces UEMs on the integration points in Clinical Operations and Vault CRM when an item fails to process. These UEMs include details specific to the error type, allowing administrators to identify more easily why a record failed. Any records that fail are automatically queued to retry during the next scheduled connection run.
Learn more about other new Clinical Operations features below.
Medical-CRM Connection
Medical-CRM Connection: CLM Content MetricsAuto-on26R1.3
This feature leverages the standard Vault to Vault connection to automatically pull CLM statistics from the CRM-connected Vaults into the source Medical Vault. By bringing this data full circle, Content Managers can view content performance directly alongside the medical content.
Learn more about other new Medical features below.
Medical-CRM Connection: Approved EmailConfiguration26R1.3
This feature automates the flow of Approved Email content from Medical Vaults to Vault CRM over a near real-time Vault to Vault connection. By utilizing Vault CRM’s document model, organizations can seamlessly synchronize Email Templates, Email Fragments, Template Fragments, and all supporting assets with their associated renditions and relationships left intact.
This feature removes the version lag and administrative overhead associated with legacy synchronization processes, ensuring that field teams always have immediate, compliant access to the latest materials for HCP engagement.
Learn more about other new Medical features below.
Medical-CRM Connection: Document TransferConfiguration26R1.3
This feature introduces the automated transfer of documents from Vault Medical to Vault CRM over the standard Vault to Vault connection. When a document in the Medical Vault is created or modified, the Document Transfer integration automatically creates or updates a corresponding CrossLink document in the connected Vault CRM. This near real-time integration ensures that Vault CRM always maintains immediate, seamless access to up-to-date, compliant content.
By replacing manual file management with an automated handoff, this feature significantly reduces administrative overhead and eliminates the risk of version lag for your field teams. It streamlines content management by automatically syncing your critical reference documents, templates, and medical content across platforms.
Learn more about other new Medical features below.
Medical-CRM Connection: Populate Inquiry's Preferred Contact Information on CaseConfiguration26R1.3
The Medical-CRM connection now automatically populates a new Preferred Contact Information field on Medical Vault Cases with the specific Person Information record mapped from the corresponding Vault CRM Medical Inquiry.
This integration eliminates manual guesswork, ensuring that Medical Information agents instantly know exactly which communication method to use for fulfillment even when a contact has multiple addresses or emails associated with it.
Learn more about other new Medical features below.
PromoMats-CRM Connection
PromoMats-CRM Connection: CLM Content MetricsAuto-on26R1.3
This feature leverages the standard Vault to Vault connection to automatically pull CLM statistics from the CRM-connected Vaults into the source PromoMats Vault. By bringing this data full circle, Content Managers and Marketers can view content performance directly alongside the creative assets.
Learn more about other new Commercial features below.
PromoMats-CRM Connection: Approved EmailConfiguration26R1.3
This feature automates the flow of Approved Email content from PromoMats Vaults to Vault CRM over a near real-time Vault to Vault connection. By utilizing Vault CRM’s document model, organizations can seamlessly synchronize Email Templates, Email Fragments, Template Fragments, and all supporting assets with their associated renditions and relationships left intact.
This feature removes the version lag and administrative overhead associated with legacy synchronization processes, ensuring that field teams always have immediate, compliant access to the latest materials for HCP engagement.
Learn more about other new Commercial features below.
PromoMats-CRM Connection: Document TransferConfiguration26R1.3
This feature introduces the automated transfer of documents from PromoMats Vault to Vault CRM over the standard Vault to Vault connection. When a document in a PromoMats Vault is created or modified, the Document Transfer integration automatically creates or updates a corresponding CrossLink document in the connected Vault CRM. This near real-time integration ensures that Vault CRM always maintains immediate, seamless access to up-to-date, compliant content.
By replacing manual file management with an automated handoff, this feature significantly reduces administrative overhead and eliminates the risk of version lag for your field teams. It streamlines content management by automatically syncing your critical reference documents, templates, and promotional content across platforms.
Learn more about other new Commercial features below.
Safety-EDC Connection
Safety-EDC Connection: Retain Primary Designation for Multi-Event CasesAuto-on26R1.2
Within the Safety-EDC Connection, the Safety Case Follow-Ups Honor Safety Decision feature is critical for reducing noise and sharing and tracking Safety decisions. However, Veeva EDC relies on a primary event to define its top-level structure, while Safety maintains a case-centric view. Previously, adding a new Adverse Event (AE) during a merge or follow-up triggered a change to the primary event. This automation often overrode the deliberate decisions of medical reviewers and caused EDC to inadvertently drop events in follow-up transmissions.
With this release, the Safety-EDC Connection introduces more stable logic for EDC-sourced Cases to ensure that primary event designations remain unchanged unless manually adjusted. By preventing automatic primary event updates, the connection maintains higher data integrity and respects the decisions of case processors.
Key updates include:
- Stable Merges: When merging an Inbox Item containing a new AE into an existing Case using the Merge to Current action, the new AE is no longer automatically set to primary. Instead, the Rank value is left blank.
- Consistent Follow-ups: When creating a new Case version using the Create Follow-up action, net-new AEs default to a non-primary status. This ensures the preservation of the existing case structure across versions.
- Explicit Safety Decisions: For both merge and follow-up actions, the resulting Safety decisions explicitly include an “add” decision for the new event to inform EDC of the update without disrupting the primary event hierarchy.
- Rank Requirement Cleanup: To eliminate data assumptions, any time an AE is received and not designated as primary (Rank value of 1), the Rank field remains blank.
Learn more about other new Safety features below.
Safety-EDC Connection: Respond to Form Sequence NumberAuto-on26R1.2
To improve the accuracy of safety case intake and eliminate ambiguity when processing multiple instances of the same reported event, Vault now captures the EDC Form Sequence Number as a dedicated field on Case Adverse Events. This makes it straightforward to distinguish between cases that share the same reported event. The field is visible on the Inbox Item detail view for cases sourced through the Safety-EDC Connection, and is included in the Inbox Item to Case Compare page to support accurate review and merge decisions. When users create a follow-up Case version, Vault automatically carries the Form Sequence Number forward.
This feature was released in 26R1.2 without documentation.
Learn more about other new Safety features below.
Safety-EDC Connection: Optimized Data ExchangeAuto-on26R1.3
To further streamline the exchange of clinical trial data and improve the efficiency of safety intake, Veeva introduces a series of enhancements to the Safety-EDC Connection. These updates focus on automating manual data entry, improving record traceability, and ensuring higher data quality during the transition from EDC to Safety. By refining how Vault handles complex clinical scenarios, organizations can reduce the risk of data discrepancies and accelerate the processing of trial-related Serious Adverse Events (SAEs).
Key capabilities include:
- Automated Follow-up Intake Management: For Cases marked for follow-up, Vault links to the new Inbox Item and copies the New Info Date, ensuring seamless continuity of trial data.
- Standardized Technical Traceability: Ensures clinical data integrity by retaining the system link (
link__sys) across follow-ups and preventing accidental changes during Case compare. - Localized Error Communication: Adds translated error messages for EDC-specific failures, allowing local safety teams to troubleshoot connection issues in their native language.
Learn more about other new Safety features below.
Safety-EDC Connection: Apply Cause of Death Details Only to Death-Related SAEsConfiguration26R1.3
To improve the precision of clinical trial data exchange and reduce unnecessary Inbox Item updates, Veeva Safety now supports an optional EDC configuration that restricts cause of death details to Cases explicitly identified as death-related. Previously, Vault applied death information to all Cases, regardless of seriousness classification. With this update, EDC can signal whether a given Case should include death information via the new Include Death Information field on the CDMS Subject Links object. When the field value is Yes, Vault applies cause of death details to the corresponding adverse event. When the field value is No, Vault withholds death information from the Case, reducing unnecessary trickle updates and keeping Case records appropriately scoped. If no death-related SAE exists for a patient but cause of death details are present, Vault retains those details on the CDMS subject data objects without applying it directly to a Case.
Learn more about other new Safety features below.
Safety-EDC Connection: Transmission Profile per ConnectionConfiguration26R1.3
To better streamline the exchange of clinical trial data and improve the efficiency of safety case intake, Veeva Safety now creates a dedicated Transmission Profile each time a Safety-EDC Connection is copied using the Copy to new action. This supports organizations such as CROs that manage multiple EDC Vaults (one per customer) where each connection requires its own distinct configuration, such as a different Organization. When an Admin copies a connection, the new Transmission Profile inherits the settings of the source profile and is assigned an incremented label (Safety-EDC Connection [n]) allowing Admins to independently configure each connection without affecting others. If the source connection has no linked Transmission Profile, Vault falls back to the base Safety-EDC Connection profile as the default for copying.
Learn more about other new Safety features below.
Safety-EDC Connection: Combine Reporter & Sender Comments for Multi-Event Follow-Up CasesAuto-on26R1.3
Veeva Safety now combines Reporter Comments and Sender Comments across multiple adverse events when Vault receives a multi-event follow-up Inbox Item from EDC. When a follow-up Inbox Item contains more than one adverse event, Vault combines the Reporter Comments and Sender Comments from each EDC event into a single Case-level field, ordered by sequence number with the primary event first. Each event’s comments are clearly labeled with the Event Reported term and Form Sequence Number, so reviewers can trace the source of each comment entry. If only one event is present, Vault does not add a delimiter.
This concatenation applies to subsequent multi-event follow-up Inbox Items only. The Merge to Current and Create Follow-Up actions do not trigger concatenation.
Learn more about other new Safety features below.
Safety-RIM Connection
Safety-RIM Connection: Data Transfer EnhancementsConfiguration26R1.3
To reduce manual data enrichment and improve the user experience of the Safety-RIM Connection, Veeva Safety delivers a series of data transfer enhancements covering additional field mapping, improved data transformations, and new Safety use cases.
Key enhancements include:
- Structured Drug Strength Data: Safety now supports granular substance strength tracking at the Product Variant level, enabling users to accurately track multi-substance products with differing strengths during case processing with strength data sourced from RIM.
- Multiple Trade Names per Registration: Safety now supports multiple trade names per Registration (an IDMP use case previously supported only in RIM), with trade name data sourced from RIM Registered Trade Names. The Safety product coding browser reflects the new data model.
- Safety Product Type Field: The Safety Product Type field on the Product object allows RIM users to classify products more precisely, with values synced to Safety to support page configuration and reporting.
- Registered As Field: The Registered As field on Registrations aligns Safety with RIM, synching the regulatory classification of each registration across both Vaults.
- Application and Registration Types: The Application and Registration Type values in Safety align with RIM, including the addition of a dedicated License Category (PMDA) field split from the existing Registration Type field.
- Substance Alias Data Transfer: Substance alias data syncs from RIM to Safety’s existing Substance Alias object, aligning both Vaults on substance data points.
- US and China Registration Number Transformation: Vault automatically transforms registration numbers for US and China submissions to meet local regulatory requirements. Vault prepends US numbers with the Registration Type and prepends China numbers with the required Mandarin characters.
- Primary Product Constituent for Combination Products: A new Primary field on Product Constituent records in both Safety and RIM Vaults identifies the primary component for complex and co-packaged products, with the value synced from RIM to Safety.
Learn more about other new Safety and Regulatory features below.
Safety-RIM Connection: New IDMP MPID Field MappingAuto-on26R1.3
With this enhancement, Vault now maps the IDMP Medicinal Product Identifier (MPID) field value from RIM directly to the dedicated IDMP MPID field on the Product Registration in Safety, ensuring IDMP-compliant product identifiers are stored in the correct field. Previously, the connection mapped the MPID value to a general-purpose field not intended for IDMP data. That field rule remains active but is now labeled MPID (Deprecated) to signal the transition. The new field rule is added to all applicable integration rules, with no disruption to existing data flows.
Learn more about other new Safety and Regulatory features below.
Quality-RIM Connection
Quality-RIM Connection: Enhanced Change Control for MaterialsConfiguration26R1.2
This feature enhances the existing Quality-RIM Connection by enabling users to apply change controls against Material records in addition to Products. Materials include packaging, active substances, and inactive ingredients. This level of detail facilitates Batch Release by supporting changes at the granular Material level where Quality Materials relate to RIM Packaging, Container, Active Substance, or Inactive Ingredient records. Tracking changes at the material level helps organizations make informed decisions about releasing batches.
What’s New?
The Enhanced Change Control (ECC) integration now supports defining changes at the Material level. Vault includes the following updates to the RIM and QMS data models and connection data flows.
Data Model Changes
- RIM: Vault now includes four new object types for the Change Item and Activity Change Item objects: Packaging Change, Container Change, Active Substance Change, and Inactive Ingredient Change. Each type includes a new ID field that references the related record, such as the
packaging__vfield on the Packaging Change object. - QMS: The Regulatory Change Item and Regulatory Activity Item objects now include the Material object type (introduced in 26R1).
Connection Data Flows
Vault syncs the following items from QMS to RIM:
- Packaging Change Items: When you create Regulatory Change Item records in QMS where the type is Material and the related material is a “Finished Material” (with the RIM Source Object of “Packaging” subtype), Vault syncs the record to RIM as a Change Item of the Packaging Change type.
- Active Substance Change Items: Regulatory Change Item records created in QMS where the type is Material and the related materials are “Raw Materials” (with the RIM Source Object of “Active Substance”) sync to RIM as an Active Substance Change.
- Inactive Ingredient Change Items: Regulatory Change Item records created in QMS where the type is Material and the related materials are “Raw Materials” (with the RIM Source Object of “Inactive Ingredient”) sync to RIM as an Inactive Ingredient Change.
- Container Change Items: Regulatory Change Item records created in QMS where the type is Material and the related materials are “Raw Materials” (with the RIM Source Object of “Container”) sync to RIM as a Container Change.
Key Benefits
- Granular Control: Quality users can create change controls against detailed material-level data from RIM.
- Streamlined Impact Assessment: RIM users can assess the regulatory impact of material changes and feed those results back to Quality automatically.
- Improved Batch Release: Facilitates a more detailed Batch Release process by ensuring all material-level changes are tracked across both Vaults.
Additional Considerations
- Material Alignment: Material records must be aligned between RIM and Quality through the Quality-RIM Connection’s Product Data integration.
- Material Selection: Any Materials that weren’t sourced from RIM (where the Link field is not populated), will not be synced to RIM as a Regulatory Change Item. Likewise, any Materials sourced from RIM with the RIM Source Object of either “Active Substance Alias” or “Inactive Ingredient Alias”, will not be synced to RIM as a Regulatory Change Item either, as these are purely used to search for the related Active Substance or Inactive Ingredient records.
- Existing Integration: The existing integration point for Activity Change Items (RIM) and Regulatory Activity Items (QMS) is reused, as the type is associated with the related item.
Learn more about the Quality-RIM Connection.
Learn more about other new Quality and Regulatory features below.
Quality-RIM Connection: Document Creation Triggers Outbound JobAuto-on26R1.3
With this enhancement, any document or document version created directly in its steady state now triggers the Quality-RIM Document Integration, creating the CrossLink in the target Vault. Previously, all source documents generated directly in the steady state would not trigger the integration.
Learn more about other new Quality and Regulatory features below.
Quality-RIM Connection: Enhanced Change Control Integration Deleted RCI EnhancementAuto-on26R1.3
This enhancement to the Enhanced Change Control integration between Veeva Quality and Veeva RIM ensures the connection does not automatically create any QMS Regulatory Change Item (RCI) records. Prior to this enhancement, if a QMS RCI record had been deleted after being sent to Veeva RIM, when receiving an update from the linked RIM Change Item record, the connection would attempt to recreate the respective QMS RCI record.
Learn more about other new Quality and Regulatory features below.
Quality-Safety Connection
Quality-Safety Connection: Promotion StatusConfiguration26R1.3
To strengthen traceability between quality and safety workflows, Vault enhances the Quality-Safety Connection to share promotion status updates in both directions, giving teams on each side visibility into how transferred records are progressing. Previously, source records in either Vault had no way to confirm whether a transferred record had advanced through the receiving Vault’s workflow. This update closes that gap in the following ways:
- When a user closes a Safety Inbox Item created from a QMS Complaint, Vault updates the associated Complaint with the Safety Inbox Item Promotion Status field, indicating whether the Inbox Item has been promoted to a Case. For Inbox Items promoted to Cases in the Safety Vault, the QMS Complaint includes the Safety Case State and Safety Case ID values from the Safety Vault.
- When a Complaint Intake record created via the PQC integration reaches the Assessment Complete state, Vault populates the associated Safety Case with the linked Complaint Names, Complaint IDs, and Complaint Intake Promotion Status, indicating whether the Complaint Intake has been promoted to a Complaint.
Together, these updates enable teams to track record promotion across systems without manual follow-up or cross-Vault lookups.
Learn more about other new Quality and Safety features below.
RIM-Clinical Operations Connection
RIM-Clinical Operations Connection: Submission Tracking EnhancementsConfiguration26R1.2
What’s New?
This release expands the Submission Tracking Integration (released in 25R2) to track additional clinical trial application outcomes, specifically Withdrawn and Study May Proceed. When a Regulatory Objective related to a clinical trial application enters one of these new state types, the RIM-Clinical Operations Connection transfers the data from RIM to Clinical.
Key updates include:
- New State Types: New Withdrawn and Implicitly Approved state types and Submission Verdicts are provisioned in RIM to support the connection.
- Mapping Logic
- To ensure cross-system alignment, the connection translates RIM verdicts into specific fields on Clinical Milestones, where the RIM Withdrawn verdict maps to the Clinical Withdrawn submission decision value.
- RIM’s Study May Proceed verdict maps to the Clinical Approved submission decision value.
- Automated Verdicts
- Non-EU procedures: The Regulatory Objective lifecycle entry action on the Withdrawn and Implicitly Approved states updates the related CDDs’ lifecycle states in RIM to Withdrawn/Study May Proceed, which then automatically populates the HA Application and HA Approval milestones in Clinical.
- EU CTR procedures: Users can manually set a verdict (for example, “Withdrawn” or “Study May Proceed”) on a specific Country Decision Detail (CDD) record whenever that specific country provides a decision. Every time a verdict field is updated on a CDD, the connection immediately transfers that data to the specific Clinical Milestone for that country. Once the overall regulatory process is complete or a global decision is made (like withdrawing the whole application), the user moves the Regulatory Objective (RO) to a final state (for example, “Withdrawn” or “Implicit Approval”). This RO state change triggers an automated cascade (via entry action) that updates all remaining related CDDs to that same state or verdict. These cascaded updates trigger the connection to push the final status to all relevant Clinical Milestones across all countries involved in the procedure.
- Date Field Handling
- Withdrawn Applications: When a verdict is set to Withdrawn, the
actual_finish_date__vis cleared (made empty) for EU CTR Part I/II, EC IRB Approval, and HA Application/Approval milestones. - Implicit Approvals: When set to Study May Proceed, the
actual_finish_date__von Clinical milestones is automatically populated based on thesubmission_decision_date__vfrom RIM. - Milestone Re-use: When a resubmission is required and a new Regulatory Objective is created for an Initial Application, the connection automatically populates relevant Study and Country Milestones in Clinical. Vault ignores whether the
link__sysfield is already populated, eliminating previous User Exception Messages. - Updated Unlinking Behavior: When a CDD record is made inactive in RIM, the connection now triggers an automated unlinking process for corresponding milestones:
- Unlink & Retain Data: For Archived studies or Completed/Inactive milestones, the
link__sysis cleared but existing field data remains. - Unlink & Clear Data: For Active studies where the milestone is not Completed or Inactive, both the
link__sysand all mapped fields are cleared.
Key Benefits
- Improved Tracking Accuracy: Provides a more comprehensive view of clinical trial application statuses by including “Withdrawn” and “Study May Proceed”.
- Increased Automation: Reduces manual data entry by automatically triggering field updates and data transfers across the connection based on lifecycle state changes.
- Streamlined Resubmissions: Simplifies the resubmission process by allowing the automatic re-use of existing Clinical milestones for new regulatory objectives.
Additional Considerations
- Milestones in the Complete lifecycle state are still ignored and will not have their
link__syspopulated.
Learn more about other new Regulatory and Clinical Operations features below.
RIM-Clinical Operations Connection: RIM Connection StatsAuto-on26R1.3
What’s New?
RIM Vaults using the RIM-Clinical Operations Connection will leverage Vault Platform’s Connection Stats object to track transactions completed from Clinical Operations to RIM.
The following fields are added to the Connection Stats object in RIM:
- Clinical Studies Created
- Clinical Sites Created
- Product Clinical Studies Created
- Unique Crosslinks Created
- Crosslink Versions Created
- User Exception Messages
- User Exception Items
- Clinical Studies Updated
- Clinical Sites Updated
- Document Metadata Updates
The RIM Vault’s inbound connection jobs update the Connection Stats, populating each with the corresponding counts currently written to the Veeva-internal Customer Activity Log.
Key Benefits
Vault Admins now have visibility into the daily number of automated transactions completed by the RIM-Clinical Operations Connection into RIM via the Connection Stats object. Stats are viewable within the connection record, or under Business Admin > Objects > Connection Stats.
The primary benefit of connection statistics is the ability to monitor the connection’s daily performance and usage metrics within a connected Vault. These statistics offer valuable insights at both the individual connection level and the system-wide level and can be used to build reports and dashboards to monitor trends and overall system usage.
Additional considerations
- Feature enablement: Auto-on
- Not in scope: Connection Stats for the other RIM connections, as well as Clinical Operations stats for inbound processes for this connection, will be implemented as separate features.
Learn more about other new Regulatory and Clinical Operations features below.
Study Training-Clinical Operations Connection
Study Training-Clinical Operations Connection: Support for Cross-Domain UsersAuto-on26R1.3
Cross-domain users are now automatically created in Study Training, following the exact same rules as standard domain users for security profiles, license types, and activation rules. When Security Profile Mapping records are in place, the corresponding security profile is assigned in Study Training; otherwise, the connection applies the default Training User Security Profile. This eliminates manual data entry, allowing Admins to seamlessly manage and transfer cross-domain users. For full visibility and easy tracking, all automated creations and updates are documented directly in the Clinical User Management job log.
Learn more about other new Clinial Operations: Study Training features below.
Clinical Operations
Features in the Veeva Connections section also affect the Clinical Operations application family.
All Clinical Operations Applications
Support Localized Number Formatting for App PagesAuto-on26R1.3
Currently, Clinical Operations applications utilize a period-only system for displaying and entering decimals. Forcing this specific format on all users regardless of their regional standards leads to potential confusion during data review and inefficiencies during entry, particularly in regions where commas are the standard decimal separator. This lack of localization currently affects all numeric field types, including Number, Currency, and Percent fields.
To resolve this, we have updated specific Clinical Operations application pages to use the International Components for Unicode (ICU) standard for all numeric fields. Vault now automatically determines the correct decimal and grouping separators for a user’s locale and dynamically formats the display while handling user inputs based on that specific locale. This update applies to Number, Currency, and Percent fields including Veeva CTMS, Payments, Disclosures, SiteConnect, and the ClinOps Homepages (eTMF & SSU).
The system ensures that symbols, abbreviations, and separators for Percent and Currency fields are positioned and displayed correctly according to local standards. The system also intelligently handles copy-and-paste actions by automatically identifying and removing digit grouping separators, such as thousands-separator periods or spaces, to ensure clean numeric entry. Throughout this process, the Vault backend maintains data integrity by continuing to store all values using standard periods, while the UI components handle the real-time conversion for the user.
This enhancement provides a seamless, localized user experience that allows global Clinical Operations teams to work more efficiently within their native formats. By aligning the interface with regional numeric standards across the entire Clinical Operations suite, Vault reduces the friction of manual data entry and decreases the likelihood of errors in critical financial and clinical tracking fields.
Prevent Creation of New Adobe Formatted OutputsAuto-on26R1.3
All new formatted output templates now leverage the Word functionality and no new Adobe templates can be created. Existing Adobe templates can continue to be edited.
EDL Override Support for Site ConnectAuto-on26R1.3
EDL Template Overrides now update Site Connect fields on Expected Documents, such as Send using Site Connect, Auto-request from Site, and Sponsor/CRO Comments. This enables country-specific tailoring of Site Connect processes.
Note: Customers with Site Connect and EDL Template Overrides should ensure these fields have the correct values on their EDL Template Overrides.
CTMS
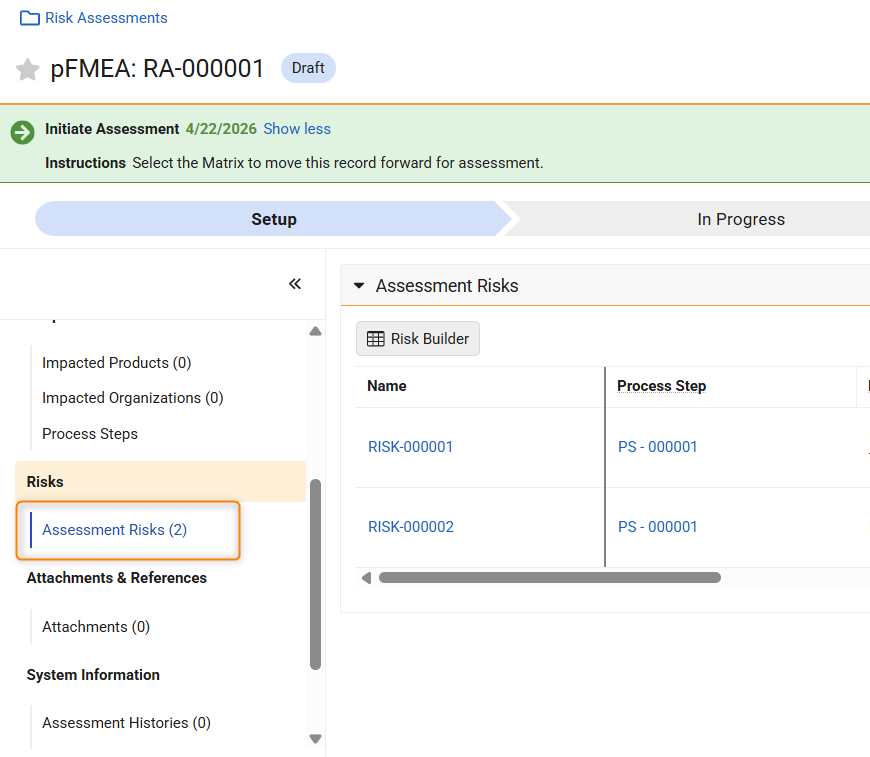
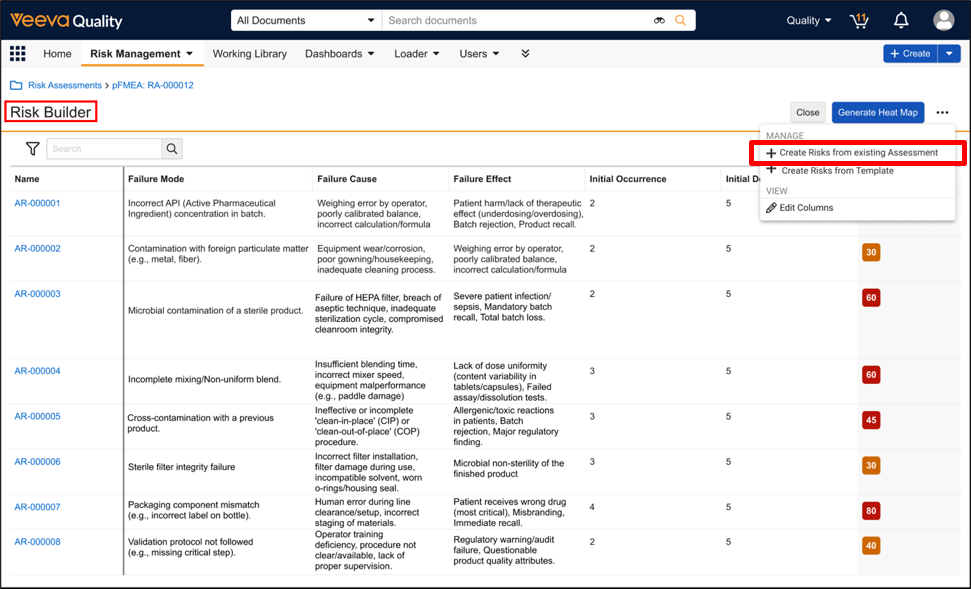
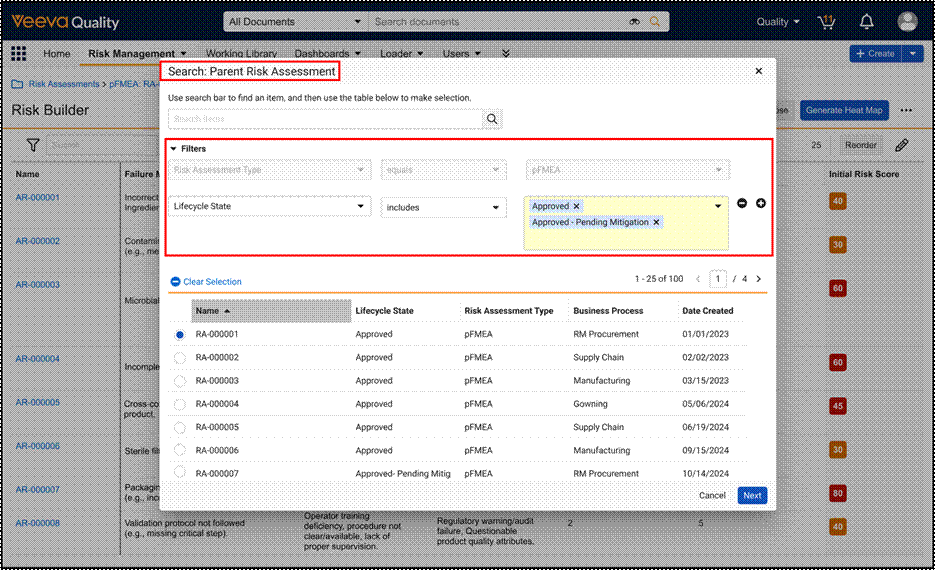
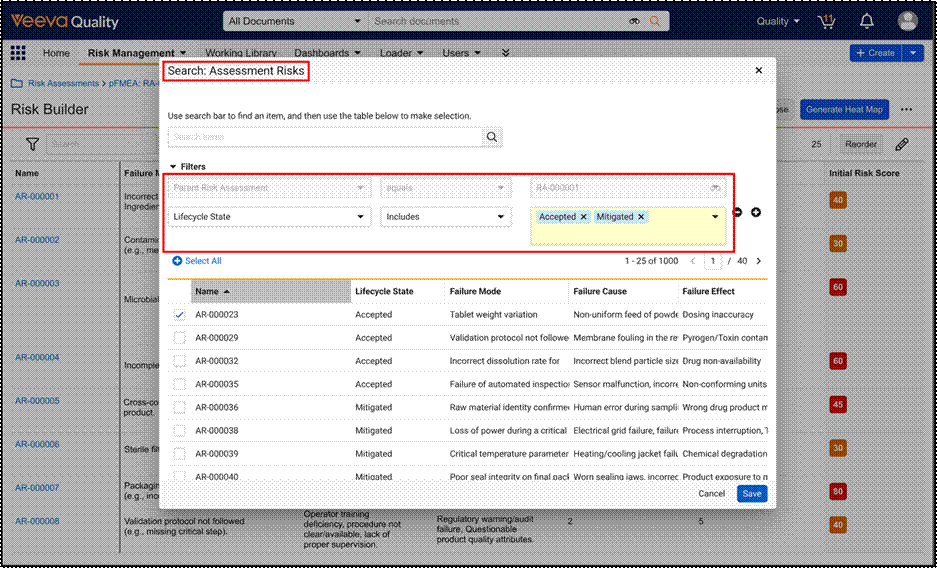
Remove Reliance on Risk Library for Study Specific Risk AssessmentsAuto-on26R1.2
During Risk-Based Quality Management (RBQM) in clinical trials, study teams often identify risks, critical processes, or mitigations that are unique to a specific protocol. Prior to this feature, Veeva CTMS required these specific elements to exist in a Global Risk Library before they could be utilized in a Study Risk Assessment. This dependency created inefficiencies for customers who were forced to navigate the administrative process of updating a global library, which delayed the finalization of risk assessments.
This release introduces an expansion of RBQM capabilities, formerly named Risk-Based Study Management (RBSM), that removes the strict reliance on Risk, Risk Mitigation, and Critical Process library records. Users now have the flexibility to:
- Create Study-Specific Risks: Manually enter unique Short Description for risks, mitigations, and critical processes directly on the record without a library reference.
- Copy from Another Study: Use the enhanced Generate Risk Assessment action to pull study-specific risks, critical processes, and critical data directly from an existing Study into a new one.
- Reference the Risk Library: Continue to link to Global Library records when standardization across studies is preferred.
This update empowers Sponsors and CROs to move beyond the constraints of one-size-fits-all libraries, enabling a truly agile risk management approach that significantly accelerates the overall risk assessment process.
Critical to Quality (CtQ) Factors Support for RBQMConfiguration26R1.2
Critical to Quality (CtQ) factors are fundamental parameters of a clinical trial guaranteeing subject safety and the reliability of study results (such as primary endpoints, recruitment targets, or eligibility criteria). In Risk-Based Quality Management (RBQM), these factors serve as the foundation for identifying which risks truly matter to the success of a study. Prior to this feature, Veeva CTMS did not have a dedicated structure to track these CtQ factors This often forced study teams to manage the CtQ factors outside the system, often in spreadsheets, which led to a disconnected risk assessment process.
This release includes data model enhancements to integrate CtQ factors into the RBQM workflow. Users can now:
- Track & manage CtQ factors in Study Risk Assessments.
- Link CtQ to Critical Processes and Data for full traceability.
- Automate setup via the updated Generate Risk Assessment action, which pulls CtQ factors from Risk Templates or existing Study Risk Assessments.
This enhancement directly promotes compliance with Quality by Design principles by anchoring CtQ factors within the entire Study Risk Assessment process. By centralizing this strategy within Vault, Sponsors and CROs can significantly improve the documentation of their study strategy, providing a transparent, audit-ready record of how strategic quality factors drive operational oversight and patient safety.
Monitoring Event Follow Up Item Seeding Logic UpdatesAuto-on26R1.2
This feature updates the logic for the Seed Monitoring Follow-Up Items action on Monitoring Events. The system now considers the Follow Up Item’s Opened Date when seeding Closed Follow Up Items into Monitoring Events.
If a Follow Up Item’s Opened Date is before the current Monitoring Event’s Actual Visit Start Date and the Follow Up Item is resolved, Vault seeds the Follow Up Item as a Closed Follow Up Item considering Completion Date values up to today’s date.
If a Follow Up Item’s Opened Date is after the current Monitoring Event’s Actual Visit Start Date and the Follow Up Item is resolved, Vault seeds the Follow Up Item as a Closed Follow Up Item considering Completion Date values up to the current Monitoring Event’s Actual Visit End Date.
Learn more about Monitoring Event Follow Up Items.
CTMS Transfer: Study Site AddressesAuto-on26R1.3
In clinical research, sponsors frequently outsource Clinical Operations to Clinical Research Organizations (CROs). However, investigational drug shipments can remain the direct responsibility of the sponsor. Even when these activities are managed externally, enabling robust sponsor oversight is critical to ensure trial integrity and regulatory compliance. To successfully drive these shipments or maintain necessary oversight, sponsors depend heavily on having access to accurate, up-to-date Study Site Addresses captured by the CRO. Prior to this feature, Study Site Addresses were not included in the automated CTMS Transfer scope, forcing study teams to rely on manual data tracking, offline communication, or duplicate data entry across disparate systems. This manual workaround was inefficient, time-consuming, and prone to administrative errors. Furthermore, the lack of a streamlined, automated solution carried a significant risk of logistical disruptions, such as drug shipment delays or misrouting due to outdated or uncoordinated site address records.
This new feature introduces the automated transfer of Study Site Address data with CTMS Transfer, establishing a seamless synchronization of location data from a source (CRO) vault to a target (Sponsor) vault. In-scope Study Site Address records are automatically transferred from the source and linked to the appropriate Study, Study Country, Study Site, and Country records in the target vault. Transferred records are secured against unauthorized edits or deletions by non-Vault Owners, and modifications in the source vault are synchronized to the target vault.
This enhancement reinforces CTMS Transfer to completely eliminate the inefficiency and risk of manual address tracking between Sponsor and CRO partners. It guarantees that sponsors possess the immediate, accurate, and granular Site details required to securely drive drug shipments and maintain vital operational oversight.
Monitoring Event Question Status BadgesAuto-on26R1.3
Vault now displays visual status badges on question section headers within Monitoring Events to indicate the completion status of questions with required responses within respective sections. This feature supports CRAs in identifying outstanding required questions while a Monitoring Event is in progress.
When a question section contains questions with required responses, Vault displays one of three dynamic indicators next to the section header text depending on the state of completion:
- Red Empty Circle: Appears next to the section header if all questions with required responses within that section remain unanswered or empty.
- Orange Half-Filled Circle: Appears when at least one required response has been provided, but other questions with required responses within the section remain incomplete.
- Green Circle with Checkmark: Appears once all questions with required responses within that section are successfully populated.
Monitoring Event Seeding Respects Previous Monitoring Event of Same Content TypeAuto-on26R1.3
This feature refines how the system identifies the Previous Monitoring Event to establish the date cut-off for seeding for the Monitoring Event. These updates ensure seeding actions - including Issues, Follow-Up Items, and Monitored Enrollment - use the correct date and seed the most accurate data.
Key enhancements:
- Content-Sensitive Prioritization: For Vaults with CTMS Content Field Defaulting enabled, the system now prioritizes events of the same content type, such as a Blinded Monitoring Event referencing the Previous Blinded Monitoring Event field. If no match is found, the system falls back to the general Previous Monitoring Event field.
- Default Blinding for Risk Actions: For Vaults with CTMS Content Field Defaulting enabled, Risk Mitigation Action and Risk Remediation records now default to Blinded regardless of the user’s role.
These auto-on updates are beneficial for customers with CTMS Content Field Defaulting enabled, allowing Blinded Monitoring Events to capture a complete history of activities since the previous visit without being influenced by the timing of Unblinded events. They also ensure that only valid records are referenced as the Previous Monitoring Event.
Additive SDV Supported in Monitoring Event Subject Visit Seeding LogicAuto-on26R1.3
During clinical trials, Clinical Research Associates (CRAs) often perform Source Data Verification (SDV) on subjects or visit items that fall outside the Clinical Monitoring Plan, which is known as Additive SDV. While Additive SDV is supported and tracked in Veeva EDC, these verification efforts were historically not reflected within Veeva CTMS on Subject Visits or Monitored Subject Visits. This data gap meant CRAs had no easy way to record within Monitoring Events that Additive SDV had occurred, pushing them to rely on manual, inefficient, and error-prone workarounds. Ultimately, it prevented the complete operational oversight and visibility necessary for robust trial monitoring.
This new feature addresses these challenges. Subject Visits, Unblinded Subject Visits, and Monitored Subject Visits now have dedicated fields to capture Additive SDV details, including whether it was performed, the date it occurred, and the reason. Furthermore, the system has been enhanced to automatically pull in these values into the Monitoring Events during the seeding process. Specifically, the updated seeding logic automatically evaluates the presence of Actual Visit End Dates across current and previous Monitoring Events to dynamically determine the correct timeframe for pulling records based on their SDV Complete Date or Additive SDV Performed Date.
This enhancement removes the inefficiencies of manual tracking by automating the ingestion of Additive SDV details directly into your Veeva CTMS monitoring workflows. By ensuring CRAs can seamlessly maintain an accurate and complete record of their extra verification efforts, this feature guarantees the necessary granularity and visibility for precise data quality management and comprehensive trial oversight.
Protocol Deviation Subject Visit Context ImprovementAuto-on26R1.3
To improve context, the new Visit (visit__v) lookup field on Issue (pdv__ctms) records displays users-friendly Subject Visit Definitions, such as Week 4 or Screening, instead of system record names. This enhancement makes it easier for users to identify visits at a glance.
Disclosures
Print Disclosures & Save to LibraryConfiguration26R1.3
Users can now generate a PDF or printable version of a Disclosure for review, eTMF storage, or cross-linking into RIM. For example, users can generate a PDF of an EU Initial Application that can be set up to cross-link into a RIM Content Plan for an EU Clinical Trial Application Submission.
Disclosures Person & Organization Search & FillAuto-on26R1.3
Vault now includes a Global Directory search component to allow users to easily fill Organization and Contact information in the Disclosure Form. This feature is available on US and EU forms.
Disclosures US Results: Add Notes & NA ExplanationsAuto-on26R1.3
Users can now annotate and enter ‘NA’ in US Results tables. This includes:
- NA Explanations: Users can mark a number field as ‘NA’ and provide an explanation on the Participant Flow Milestone, Baseline Measures, and Outcome Measures data tables.
- Notes/Comments: Users now also have the ability to Add Notes/Comments to the Participant Flow Milestone table.
- Units: Users can now provide units for the Participant Flow Milestone Table.
Disclosures US Results: Outcome Measure EnhancementsAuto-on26R1.3
Users can now define different Arms per Outcome Measure and re-order Outcome Measures as needed. Users can also select Results not ready and add anticipated posting dates for delayed data.
Validate Action on US Registrations using APIAuto-on26R1.3
Users can now validate their US Registrations Disclosure data (such as their US Results) against the ClinicalTrials.gov API directly within Vault. A new Validate action provides on-screen validation messages, allowing users to correct errors before final submission. This pre-validation step streamlines the submission process, reduces the risk of rejection, and helps ensure that Disclosures are accurate and compliant.
Disclosures: Site List EnhancementsConfiguration26R1.3
The US Registration Site List now skips validation when the Study Type is Observational or Expanded Access. This update allows users to change the Patient Registry Flag or Expanded Access Type on Disclosures. Expanded Access Disclosures can now be triggered automatically if Study Design Type is Expanded Access. Additionally, Vault now displays an indicator on sections with Open Comments if any subsections contain comments.
CTMS, Disclosures
Move CTN Setting back to CTMS SettingsConfiguration26R1.3
Admins can now enable the CTN Setting in the CTMS section of the Admin Settings page.
Learn more about Configuring Japanese Clinical Trial Notifications (CTN).
eTMF
TMF Quality ReviewConfiguration26R1.2
Maintaining a healthy Trial Master File (TMF) requires more than just verifying individual document integrity at the point of submission. While tactical document quality checks are essential for ensuring correct metadata, formatting, and signatures on an isolated basis, they often fail to capture the “relational” health of the TMF, specifically whether the collective body of documents provides a coherent and complete narrative of the trial expected per Good Clinical Practice (GCP) and global regulatory requirements. Historically, most organizations performed these quality reviews manually using external spreadsheets or reports. However, this disconnected approach makes it difficult to track review progress, manage sampling consistently, and provide a clear audit trail of sponsor oversight, ultimately increasing the risk of inspection findings or late-identified documentation gaps.
The new TMF Quality Review feature introduces a centralized framework to manage these periodic and strategic TMF reviews directly within Veeva eTMF. This solution streamlines the process through five key pillars:
-
Standardized Strategy: TMF Admins define Quality Review Templates to standardize which document types require review, the specific guidance reviewers should follow, and the periodic frequency of these checks. Study Managers can then leverage these templates when creating Quality Reviews for specific Studies.
-
Coordinated Execution: Once the Quality Review owner is identified and the review schedule is confirmed, Quality Review Cycles can be triggered automatically by Study milestones or initiated manually. Quality Review Owners can then precisely define the scope of the review by selecting specific Review Cycle Items, which can represent the Study Level, specific Study Countries, or individual Study Sites. Specific reviewers are assigned to each selected country or site to ensure comprehensive trial coverage.
-
Intelligent Sampling: Instead of manual selection, the system identifies the review scope using Fixed Sampling Percentage or Risk-Based logic based on Document Types and Subartifacts. This provides teams the flexibility to tailor their sampling strategy based on various factors.
-
Guided Review Interface: Reviewers are guided through their assigned scope starting from the Review Cycle Item, which displays the full list of documents sampled for their specific area. A dedicated Quality Review panel on each document provides specific Review Guidance to ensure every team member evaluates coherence and completeness against the same objective standard.
-
Integrated Remediation: When a gap in the trial narrative is found, reviewers raise Quality Issues or Clinical User Tasks directly from the document. These findings are automatically linked to the review cycle, providing a transparent record of identification and resolution
This enhancement transforms TMF quality management into a proactive, system-driven strategy. It helps organizations ensure strict adherence to their internal TMF Review Plans and facilitate the early identification of deficiencies such as gaps, errors, or late filings. Ultimately, this framework maintains the coherent narrative regulators expect, keeping the trial ready for inspection at any time to maximize inspection success.
Prevent Modifications on Archive Snapshot DocumentsAuto-on26R1.3
During Study archival, documents associated with multiple Studies are automatically stored as a static snapshot copy. To protect the integrity of these historical records, the system now prevents modifications to these documents:
- No Unarchiving: Users cannot unarchive document snapshots. The system blocks both the Unarchive Document user action and the Update Multiple Documents API endpoint.
- No Copying: Users cannot duplicate these snapshot records. The Make a Copy user action is disabled to ensure archived snapshots remain linked to the original source document.
- Clear Error Messaging: If a user attempts to unarchive or copy a snapshot document, Vault displays clear error messages explaining that the action is not allowed.
CTMS, Disclosures, eTMF
Added More Standard Fields for Study PhaseConfiguration26R1.3
Vault now includes additional CDISC standard values as inactive options in the Study Phase picklist. Admins must activate these values to enable triggers, prepopulation, and rules functionality for Disclosures. Additionally, Vault includes Expanded Access as an inactive option in the Study Design Type picklist.
OpenData Clinical
Display Preferred Affiliation in OpenData Clinical DetailsAuto-on26R1.2
This release provides increased Investigator affiliation flexibility by allowing users to designate a primary or preferred Contact Information record within a Vault. To ensure data integrity, Vault enforces a primary constraint for contact records. The OpenData Clinical Details section dynamically displays the address and contact details of the chosen primary affiliation. This enhancement deprecates the manual Change Primary Affiliation action in favor of a standard Is Primary field on Contact Information records, making it easier for users to manage preferred affiliations.
Primary Country for OpenData Clinical InvestigatorsAuto-on26R1.2
Primary Country is now automatically populated for OpenData Clinical Investigators. Vault leverages curated global directory data to set the value based on the Investigator’s primary site affiliation. This enhancement supports usage of the Clinical Operations-Vault CRM Connection when OpenData Clinical is enabled. OpenData Clinical provides the primary country for Investigators, which Vault needs to route activities to regional CRM instances.
Support for Investigators without Site AffiliationsAuto-on26R1.2
This feature enables OpenData Clinical to deliver records for Investigators found in clinical trial registries, even when a specific affiliation to an OpenData Clinical Site cannot be confirmed. Previously, Vault only delivered Investigators with at least one confirmed site affiliation.
Global Directory to OpenData Clinical MatchingAuto-on26R1.3
OpenData Clinical’s enhanced matching process streamlines how customers align existing Global Directory records to the OpenData Clinical dataset. This process automatically generates bulk Data Change Requests (DCRs) within the Vault to manage and track matches. Using a dedicated Match Vault Record DCR type, the system routes requests to Veeva data matching stewards. Once matched, OpenData IDs return to the Vault, where users can apply them to records.
Local Language Name & Address Details in OpenData ClinicalAuto-on26R1.3
This feature enables OpenData Clinical to deliver select Investigator and Institution/Site details in their localized language. This supports operational workflows, such as drug delivery logistics, clinical trial notifications, and regional site communications, by providing localized language representations for countries with non-Latin alphabets or character-based writing systems, as well as sites with non-English names.
For Investigators within applicable countries, OpenData Clinical delivers Local First Name, Local Middle Name, Local Last Name, and Local Full Name, alongside an associated Local Language picklist. For Institutions/Sites (Organization object), OpenData Clinical delivers Local Name with an associated Local Language picklist, while the Location object captures geographical references including Local Name, Local Street Address 1, Local Street Address 2, and Local City with an associated Local Language picklist.
When OpenData Clinical is enabled, Vault blocks manual edits to these fields, requiring a Data Change Request (DCR) for updates. The DCR dialog includes a section for local fields to propose changes to names and addresses. Within the DCR interface, this section automatically collapses when all local field values are null, defaults to open when at least one local field contains a non-null value, and is hidden for out-of-scope countries where Vault does not capture local fields. For excluded records, users can maintain manual edit capabilities within the specialized OpenData Clinical application section.
Additionally, this feature delivers a Full Name field to track the full concatenated Site Name in English and Prior objects (Prior Person, Prior Organization, and Prior Location) now track all Local fields and Full Name updates.
Set OpenData ID in Record View ModeAuto-on26R1.3
For Investigator or Institution records that are not mapped to OpenData Clinical, users with appropriate permissions can now select the Set OpenData ID action directly from the record’s View mode. Previously, users had to view the record in Edit mode to select Set OpenData ID.
Prior Contact Information Relationship OptimizationAuto-on26R1.3
This feature ensures data consistency for Vaults with OpenData Clinical enabled as Veeva transitions to a new relationship between Prior Contact Information records and Contact Information records.
Payments
Fee Schedule Template BuilderConfiguration26R1.2
The Fee Schedule Template Builder is a powerful new workspace designed to simplify the planning, calculating, and managing of study costs. This feature provides a dedicated app layout for building Fee Schedule Templates that display information in a dynamic and intuitive grid layout.
The Fee Schedule Template Builder includes:
- Interactive Fee Management Grid: A spreadsheet-like interface that allows you to manage procedures, visits, and costs in a single view. You can quickly toggle between different views for Procedures, additional Subject Costs, and Site Fees.
- Flexible Reimbursement Categories: You can designate Procedures as:
- Included: The amount is added to the total visit cost.
- Invoiced/Contingent: Items not included in the Visit Costs but included as separate paid items when applicable. These costs are included in maximum cost calculations.
- Standard of Care: Procedures performed as standard medical practice. Not included in Visit Cost or Maximum Cost Calculations.
- Comprehensive Budget Overviews: Calculated visit costs, broken down to visualize overhead, tax, and total overall costs. Additional calculations for total costs per subject and maximum site costs based on planned enrollment help visualize total financial commitments.
- Real-Time Cost Summaries: As you enter amounts, total and maximum projected costs are automatically calculated, giving you visibility into how individual line-item changes impact the overall budget.
- Improved Navigation: The grid supports keyboard navigation, allowing you to move through cells and update costs without relying on your mouse.
This feature transforms a complex, manual process into a live, integrated workspace, reducing the risk of errors, streamlining processes, and providing improved visibility into overall study costs.
Payments:Alternate Currency RoundingAuto-on26R1.2
Veeva updated how Vault handles currency rounding to better support global clinical trials. Previously, when rounding for some currencies using different formats than two decimal places, the system would not round the corresponding Amount value based on the currency. Now, Payments functionality handles the local currency for any Vault currency, such as zero-decimal currencies as well as those requiring three decimal places, saving the value with the same minor units as the currency selected.
Tax Rates: Calculating Tax Rate Total ValueAuto-on26R1.2
The system now automatically calculates and validates the Tax Rate (Total) on Tax Rate records based on the values entered for Country, Secondary, and Tertiary Tax Rates. This ensures data integrity and prevents Tax Rate miscalculations.
When creating or updating Tax Rate records that are not in use, the Tax Rate (Total) is automatically calculated as the sum of the three individual rates. If a specific rate is left blank, the system treats it as zero for the total calculation. For Tax Rates that are in use, to prevent unintended data changes, users can only update individual tax rates if the new sum matches the existing Tax Rate (Total). If there is a mismatch, the system displays an error message.
Fee Schedule Builder: Site LevelConfiguration26R1.3
Building on the foundation of the Fee Schedule Template Builder, the new Fee Schedule Builder extends these powerful cost management capabilities to the Study Site level. This feature provides payment teams with a dedicated, site-specific workspace to define and refine site budgets.
The Fee Schedule Builder includes:
- Site-Specific Updates: While the Template provides initial budget information, the site level Fee Schedule Builder allows users to check out and update specific cost details based on site negotiations.
- Template-to-Site Automation: Users can utilize the Create Fees from Template action to populate a Site’s Fee Schedule Builder with Procedures, Visits, and Cost data from the selected template.
- Dynamic Cost Calculations: The workspace automatically calculates total and maximum projected costs, providing real time visibility into total estimated site costs based on projected enrollment.
- Streamlined Fee Creation: A workflow step can be used to generate and update the actual Fee records based on the data defined within the Fee Schedule Builder, ensuring information remains in sync.
This extension transforms the high-level planning on the Template Builder into site-specific reimbursement data, streamlining the path from study design to site payment.
Fee Schedule Templates: Cost AutomationConfiguration26R1.3
Building on the efficiency of the new Fee Schedule Template Builder, this feature streamlines the initial budgeting process by eliminating the manual entry of starting costs for procedures and administrative site fees. By leveraging reference cost libraries, Vault can automatically populate the Unit Cost and Upper Limit values for these records.
This feature includes:
- Cost Reference Libraries: We’re introducing two new objects, Procedure Reference Cost and Site Fee Reference Cost, to store baseline costs by country and currency.
- Smart Query Logic: When a user adds a Procedure or Site Fee to a template, the system automatically queries the reference cost libraries for a matching record:
- The system looks for matching reference cost records based on the Global Procedure or Site Fee Definition and the Study Country.
- If multiple cost records exist, the system automatically selects the entry with the most recent Reference Date.
- To prevent financial errors, the system verifies that the Reference Cost currency exactly matches the Fee Schedule Template currency.
- Automated Cost Population: When a matching record is found, Vault automatically populates the Unit Cost and Upper Limit. If no matching record is found or if currencies do not match, these fields remain blank for manual entry.
Automated values remain fully editable by users, allowing payment teams to adjust costs for localized study needs. This feature streamlines template setup by reducing manual data entry, minimizing potential errors, and providing immediate insight into standard regional costs.
Payments: Site Contract TrackingConfiguration26R1.3
To streamline Clinical Trial Agreement (CTA) management, the new Site Contract Tracking feature introduces a dedicated framework for managing contract data and documentation, directly linking Contract records to their corresponding documents and Site Fee Schedules.
To support this, a standard Contract object is now available to track CTAs and other legal agreements between sites and sponsors. Each Contract record can be related to one or more Fee Schedules, ensuring integrated tracking of financial terms and status. A standard contract lifecycle is provisioned to track the Contract status from Drafting through Approval, with subsequent Contract Amendments tracked via separate Contract records.
Users can generate contract documents directly from templates stored in Vault, automatically pulling in important data such as Study, Country, Site and Product information. This functionality reduces administrative burden and improves compliance by filing the documents to the correct, secured location in Vault.
We are also introducing new document type groups to safeguard sensitive financial and legal data. These groups allow for granular security controls to ensure that only authorized users have access to contract and payment documents. New standard application and Study Team Roles specifically tailored for payment, contract, and legal users are now available, making it easy to assign Personnel.
By bringing together legal contractual agreements with operational fee schedules, this feature provides a standardized way to manage contracts, reducing administrative bottlenecks, protecting sensitive data, and accelerating the site initiation process.
Site Connect
Document Action FilteringConfiguration26R1.2
Administrators can now control the distribution actions available to users by specifying either Revise & Return or None in the new Available Actions field when configuring the Send Document to SiteVault action on document lifecycle states.
By selecting a specific action—or allowing both—organizations can ensure that users only select the distribution action that aligns with the intended business process for documents in that state.
Study Plan in ProductionAuto-on26R1.3
The Study Plan feature provides a collaborative workspace within Vault that allows Sponsors/CROs and Sites to establish, align on, and measure against standardized metrics and milestones. By introducing a unified tracking space directly in the Site Connect interface, sites gain visibility into operational benchmarks—such as enrollment targets and key activation dates.
A Study Site Plan can be created by either a Sponsor/CRO or a Site User. Site Users can generate a plan directly through the new Study Plan section in the Site Home UI by clicking the + Create button. Alternatively, Sponsors can trigger plan creation through a new configurable action, Create Study Site Plan.
Upon creation, the plan will be in a Draft state where both parties can collaboratively populate the plan values of items associated with Open metrics and milestones. During this phase, Site Users enter values directly via the Site Home UI, while Sponsors and CROs populate data through the underlying Study Site Plan Item records. If a Study is not utilizing the connection to Veeva EDC or Payments at the time of plan creation, those specific line items are automatically grayed out in the UI and unable to be populated.
Once the plan is populated, either the Sponsor/CRO or Site can route it for review. The flow of the review process depends on which party initiates the review.
- Site-Initiated Review: A task is immediately generated to the Sponsor/CRO and the plan enters the In Review state. Once the Sponsor/CRO completes their review the plan enters In Site Review. At this point, the Site can choose to Approve and finalize the plan or Make Changes and send the plan back to Draft.
- Sponsor-Initiated Review: The plan immediately enters the In Site Review state. The Site can choose to Approve and finalize the plan or Make Changes and send the plan back to Draft.
If the plan circles back to the Draft state, either stakeholder can again make changes to the Plan and restart the review cycle.
Once the site chooses to Approve the plan, the plan moves into the Final state, and the planned values are no longer editable by either party. In this locked state, the agreed-upon plan values are copied to the item forecast fields and sync across the Open associated milestones and metrics. If using the standard metrics and milestones, the forecast and actuals from the linked milestones and metrics flow seamlessly into the Site Connect UI and as milestones complete, this will be reflected in the status. If a Sponsor/CRO is not utilizing the standard milestone types the values can be entered manually directly on the individual plan item records to preserve site visibility.
To provide a quick, intuitive view of ongoing site progress, the UI features color-coded status indicators in the difference column once actual values populate. A green indicator signifies that the agreed-upon plan is successfully met or ahead, while an orange indicator provides an immediate visual warning that the actual date or metric value is later than or less than the plan.
Invoicing Sponsor in Site ConnectAuto-on26R1.3
Site Invoicing will enhance the payments module within Site Connect by autocreating Payment Requests when Sites upload Invoices.
Site Users with the Manage Site Reimbursements responsibility will see a new +Add Invoice button within the Reimbursements tab of the Payment Information section. Once a document has been uploaded, a Payment Request record will automatically be created in the corresponding Sponsor/CROs Vault. The record will have a Source value of Site Invoice, link to the uploaded invoice document within the Site Invoice field, and be associated with the applicable site.
Additionally, Rejected Payment Requests will now display within the Reimbursements tab.
Secure Payments DocumentsAuto-on26R1.3
Sponsors/CROs can now control payment document visibility among individual site users in Site Connect by managing the assignment of the Manage Site Reimbursements Study Person Responsibility.
For site users with the Manage Site Reimbursements responsibility, a new Documents tab is available directly inside the Payment Information section. This tailored workspace organizes payment-related files into dedicated All, Sent, and Received sub-tabs, mirroring the familiar grid layout and metadata columns of the Document Exchange section.
When a site user lacks an active Manage Site Reimbursement responsibility, they will be restricted from viewing payment documents within both the Document Exchange and Payments interfaces. This secure handling applies specifically to four VCD Artifacts: Site Invoices (vdc_206), Site Budgets (vdc_302), Contractual Agreements (vdc_106), and Payment Letters (vdc_201). Additionally, unauthorized site users are now unable to upload Site Documents of type Budget or Invoice.
Document Exchange communications also dynamically account for this security layer. Only Site Personnel with the active Study Person Responsibility will receive targeted email and in-app notifications pertaining to payment document exchanges and tasks.
Daily Document Exchange EmailsAuto-on26R1.3
To streamline site communications and eliminate fragmented document exchange notifications, sites now receive one daily document exchange email.
Previously, manually sent documents triggered separate emails based on when they were sent, grouping only the documents exchanged with that Site within a certain time frame. However, documents sent to the Site through auto-document exchange would all be grouped together in a daily email sent to the Site.
With this update, Site Connect consolidates document exchanges—whether sent manually, through packages, or via auto-exchange—into a single, comprehensive daily digest per sponsor. This ensures clinical research sites receive a predictable, organized summary of the day’s document exchange on a sponsor by sponsor basis. Included in this summary is also document recall notifications.
Optional Safety Distribution AcknowledgmentAuto-on26R1.3
Sponsors/CROs can now distribute Safety Documents that do not require formal Site acknowledgment.
This feature introduces a new Acknowledgement Waived field on the Safety Distribution Default object. When this field is enabled on a default record, it establishes that any subsequent site distributions generated under those criteria do not require formal site acknowledgment. Upon distribution to a site, the system automatically creates a distribution task with an action of Acknowledgment Waived and marks it as complete, ensuring no action is required from the site.
For Site Users, these waived documents are visible within the All Documents tab of the Safety Distribution section and in the All and Received grids of the Document Exchange section. In the Safety Distribution UI grid, the Acknowledged By and Acknowledged Date columns will display Acknowledgement Waived by Sponsor/CRO.
These waived documents will still be included in the daily safety email to ensure awareness, but they are excluded from weekly summary emails so there is no outstanding task for the site to complete.
Download Acknowledgment DetailsAuto-on26R1.3
This feature adds two new columns in the All Documents tab of the Safety Distribution section:
Acknowledged By and Acknowledged Date. If the Safety Letter was acknowledged via email, outside of the UI, the Acknowledged by field will read Completed via Email.
Additionally, users can now download the acknowledgment history directly from the All Documents tab of the Safety Distribution section. The downloaded file is saved locally as [sitename]_safety_distribution_details.csv and mirrors the layout and columns found within the user interface grid.
Site Connect UI EnhancementsAuto-on26R1.3
This feature updates the UI hover text for the connection icon on the Study Site object. If a Study Site is connected to SiteVault the text will now read “This Study Site is connected to SiteVault”, when hovering over the icon.
In addition, the user interface in Site Home is enhanced so that when a user compresses their screen, certain key components dynamically scale to fit the smaller view, including the search bar, record count, and buttons such as Send for eSignature.
Update Email Details after Document ViewAuto-on26R1.3
If a Distribution Task Recipient record has an Email Status of Failed and the recipient subsequently views the document in the UI, which updates the Email Status field to Viewed, the Email Detail field will now clear.
Download Document EnhancementsAuto-on26R1.3
This enhancement will improve the consistency of the naming convention of zip files when documents are downloaded from a safety email, ensuring the same format whether or not authenticated document links are included.
Study Startup
Milestone Planning View EnhancementsAuto-on26R1.3
This release introduces a more dynamic visualization and editing experience to the Milestone Planning View, streamlining how Clinical Operations teams manage trial timelines. We are committed to refining this experience and will continue to deliver further planning enhancements in upcoming releases.
Building upon our existing feature set, we’ve introduced automated projections based on dependency date offsets and ghost icons to compare current plans to new projections. When date offsets are leveraged and manual changes are made within the Milestone Planning View, the system will skip running the Update Dependencies workflow after saving as to not override timeline adjustments that have been made by an end user. In the case where a Milestone is manually edited in the Planning View and its Downstream Milestone is not displayed on the page, the system will still run the Update Dependencies workflow to ensure alignment with your timeline changes. The Update Dependencies workflow will also run for any workflow or state change actions initiated from the Planning View.
The page also introduces new filtering including the ability to adjust the timeline with preset zoom options and a date range selector, a Today button which allows for quick navigation to the current day, and a new Milestone Category filter to further narrow down milestone results. The Apply Dependencies toggle has also been relabeled to Show Dependencies and when enabled, will now show a Level column with a clickable link to the related Study, Study Country, or Study Site record.
The feature also includes some refinements to error support, including locking the cell when a user does not have access to edit and providing more error details in the compiled csv. The planning view also introduces new visual indicator treatment for Site level milestones shared via Site Connect’s Study Plan feature and when milestone roll-up dependencies are used to automatically update milestones to avoid unneeded data entry.
Finally, a few user experience changes have been included to help teams managing milestones more easily navigate the Milestone Planning View. These changes include a system action to launch the Milestone Workspace from the Milestone’s Action menu, a pencil icon on the date cells, timeline hand dragging support, and ability to adjust the width of the data columns.
Milestone Study Country & Site HardeningAuto-on26R1.3
With this release, we are enhancing the Multi-Country and Site IRB & EC Submissions Multi-Country and Site IRB & EC Submissions feature. Previously, managing dependencies between Country-level applications and Site-level approvals required manual tracking, which often led to redundant data entries. To streamline this process, Vault now automatically creates finish-to-finish dependencies with a Date Offset of zero between Country-level Applications and Site-level Approvals when the Select Specific Country and Site field is set to true on the Country Submission Application Milestone and a Milestone Study Site is created. The automatic creation of a finish to finish dependency is supported between the following milestone types:
- EC/IRB Application (
ec_irb_submission__v) and Site EC/IRB Approval (site_ec_irb_approval__v) - Other Country Application (
other_country_application__v) and Other Site Approval (other_site_approval__v)
This enhancement allows study teams to clearly identify where an approval originated from, while concurrently improving visualization in the Milestone Planning view.
Additionally, Vault only creates Milestone Items for the specific Milestone the Country or Site was added to, rather than creating Items for all incomplete Milestones of that type, effectively only linking Expected Documents to the relevant Submission. For example, if you add a Milestone Study Site to an initial EC/IRB Country Application but not the resubmission, Vault only links the relevant expected documents to the initial application.
This targeted linking prevents the creation of unnecessary records during frequent resubmissions, such as protocol amendments, so that your submission tracking remains clean and reliable.
CTMS, eTMF, Study Startup
Early-Finish OffsetsAuto-on26R1.2
Vault now automatically pulls in downstream Milestone dates when an upstream Milestone is Completed or Planned earlier than originally expected. Previously, dependencies only pushed dates further out. Now, the system applies the defined Date Offset to ensure timelines stay accurate in both directions. This enhancement works in tandem with other dependency features including Weekday Adjusted Offsets, 26R1 Milestone Dependency Enhancements, and Actual Date Offsets.
Learn more about Date Offsets.
New Standard Milestone Types: First IP Received, Operational Closeouts, & Sample TestingConfiguration26R1.2
This feature introduces three new standard values to the Milestone Type picklist to enable more granular tracking of Site Startup and Study Closeout, allowing for a clear distinction between operational activities and system-level state changes.
- First IP Received at Site (
first_ip_received_at_site__v): Tracks the initial Investigational Product delivery at the site level, separate from the Site Activated milestone. - Country Operational Closeout (
country_operational_closeout__v): Tracks the completion of operational activities at the country level while keeping the country open for final submission processing. - Study Operational Closeout (
study_operational_closeout__v): Tracks the completion of operational activities at the study level (e.g., staff roll-off) prior to the formal system close out and archival.
Learn more about Standard Milestone Types.
Milestone Document HardeningAuto-on26R1.2
This feature optimizes Milestone Document creation by reducing the previous five-minute delay, ensuring records are generated in near real-time. This enhancement provides more accurate data for dependent features like the Milestone Workspace, Milestone Document Panel, and Seed Package Documents action. Additionally, when clicking the Unapproved Documents widget on the TMF Homepage, the view has been updated with a new custom Documents page for unapproved items, further removing the system’s reliance on the Milestone Document field.
Learn more about TMF Homepage.
Study Milestones in Process MonitorAuto-on26R1.3
Process Monitor now supports the tracking and visualization of Study, Study Country, and Study Site progression using clinical milestones. Previously, Process Monitor generated analytics and graphs based strictly on the duration records spent in object lifecycle states. This did not allow for processes tracked across multiple related records, like Milestones, to be used in Process Monitor.
Users can now measure progress, visualize graphs, and evaluate key performance indicators (KPIs) based on Milestone Actual Finish Dates. When creating a Process View, the Process drop-down includes three new standard options:
- Study Milestones
- Study Country Milestones
- Study Site Milestones
These processes display Study, Study Country, and Study Site progression using milestone completion as the individual process steps. Once a milestone process is selected, users can select active Milestone Type values within the Cycle Time drop-down. This allows quick and easy visualization of milestone-based cycle times for Studies, Study Countries, and Study Sites, unlocking comprehensive bottleneck analysis and KPI reporting.
Block Milestone Rollup DatesAuto-on26R1.3
A new Block Rollup Dates multi-select picklist on Template Milestone and Milestone objects allows admins to indicate whether rollup logic should be blocked from auto-populating the Baseline Start Date, Baseline Finish Date, Planned Start Date, and/or Planned Finish Date. This provides more granular control over which individual date fields receive values from rollup dependencies, allowing users to manually maintain some milestone dates while still permitting other dates, like Actuals, to roll up as intended.
eTMF, Study Startup
Include Current Day in the Study Document Audit ExportAuto-on26R1.3
When a user exports the full Study Document Audit Trail and includes the current day in their date selection, the system now includes audit entries for that day in the generated export.
Previously, a gap existed for audit entries generated between the last daily job run and the exact time of the export. Now, when the selected date range includes today, the system pulls all audit entries up to the current day and includes them in the export.
Study Training
In addition to the features described below, the Study Training application also received enhancements from the following features described in the Quality: Training section:
- Optional Due Dates for Matrix Training
- Curriculum Due Dates
- Pre-Training Quiz
- Completion of E-Learning Completes Training Assignment
- Quiz Dynamic Sections
- Training & Study Training Enhancements
- Curriculum Completion Percentage Update
Additionally, Display Application Configurations by Enabled Application (Quality) and Study Training-Clinical Operations Connection: Support for Cross-Domain Users apply to Study Training Vaults.
My Study Team: Enable Individual Learner Page ViewAuto-on26R1.2
Clinical Research Associates and Principal Investigators require a direct way to view detailed training history for their study staff without relying on reports, which PIs cannot access. Vault now provides a dedicated Individual Learner Page, accessible by clicking a Learner’s card on the My Study Team page, that displays comprehensive views of the Learner’s open, completed, and all Training Assignments.
By accessing this information at the point of need, training managers can now quickly determine whether a Learner is qualified to perform specific tasks or if they require a “nudge” to complete outstanding requirements so that they can ensure study compliance and verify that staff are qualified for specific tasks without waiting for manual reports.
Update Training Assignments Job Performance ImprovementsAuto-on26R1.2
Occasional large-scale events such as migrations can lead to slow background processing and system timeouts due to the massive volume of configuration permutations and logging overhead triggered during deployments.
Vault now introduces infrastructure optimizations for the Update Training Assignments job in Study Training. By focusing on relevant matching paths and utilizing bulk memory filtering for data fetching for assignments, these updates reduce job runtimes by up to 40% and significantly decrease log file sizes.
These enhancements ensure a more stable, scalable environment with faster status updates, even as your study data grows.
Geographic Assignment RolloutAuto-on26R1.3
Managing document version training in global studies is often complex because site readiness and local regulatory approval vary by region. To facilitate this process, Study Training now creates a unique Training Requirement for every version of a connected Clinical Operations document, allowing you to manage each version independently, for example allowing learners in Japan to continue with version 1.0, while learners in France move to version 2.0.
Key updates include:
- Partial Disablement of Training Requirement Impact Assessment (TRIA): TRIA will no longer be issued for connected documents. Instead, the Study Training Matrix Builder user interface is updated to allow for management of these updates directly. When a Training Admin removes a requirement from the matrix, Vault prompts them to choose between cancelling open Training Assignments or requiring that Learners complete them.
- TRIA will continue to operate as it does today for any non-connected Training Requirements which are created directly in Study Training and not automatically via the connection.
- Ineligible Lifecycle State: A new Ineligible lifecycle state is introduced for the Curriculum-Training Requirement object, which allows existing Learners to finish training on the current version while ensuring new hires start on the latest version. In this case, existing Training Assignments remain open for completion, but Vault does not issue new assignments to Learners.
- Recognize Prior Completion: This new Training Requirement field allows Training Admins to excuse Learners who have already completed a previous version of a training for connected requirements.
These changes provide granular control over regional training rollouts and prevent the need for manual workarounds, ensuring Learners are always trained on the content appropriate for their specific location without disrupting global compliance oversight.
Study Training Matrix SnapshotConfiguration26R1.3
Study Managers often need to provide a record of their Training Matrix to auditors or store a version-controlled copy in their eTMF to document what was live at a specific point in time.
Vault now automatically generates a Training Matrix Snapshot each time a Training Admin clicks Publish to take a matrix update live. This feature saves time by creating a permanent, uneditable PDF document that captures all records represented on the live matrix and storing it directly as a study document, with the naming convention {Study Number} - {Publish DateTime} - Training Matrix Snapshot.
This ensures compliance records are always accurate and easily accessible, with subsequent updates automatically creating new major versions of the document so you can maintain a clear audit trail of your training history. Each snapshot includes a cover sheet with the study number and publication details for a professional and readable format.
Admins must activate the Training Matrix Snapshot document type and configure the appropriate lifecycle and permission sets to enable this automation. This may include updating existing entry actions within the Draft to Approved document lifecycle’s Approved state: When Vault creates a related Training Requirement, this should exclude the entry actions to exclude the Training Matrix Snapshot document type when creating a related Training Requirement.
Study Training Matrix Extract & LoadConfiguration26R1.3
Setting up and maintaining your study training requirements is now more efficient with the introduction of Excel-based configuration for the Training Matrix: this feature allows Training Administrators to perform mass updates and automate initial study setup.
You can now export a Training Matrix to a formatted Excel file to add or remove Learner Roles or Training Requirements and update checkbox mappings in a familiar grid interface, then import the file back to a draft matrix to apply changes in bulk.
Study Training Matrix Builder Edit Menu ChangeAuto-on26R1.3
Administrators currently use the Study Training Matrix Builder to manage study-specific training requirements, however the primary Edit and Delete buttons occupy valuable screen space needed for the new Study Training Matrix Extract & Load functionality.
We moved these buttons into the Actions menu so that the interface remains clean and accessible as we add more powerful data management tools, allowing you to manage complex training matrices more efficiently.
To use these actions, simply click the Actions menu and select the desired task.
Replace User AssociationsAuto-on26R1.3
Manual, repetitive work is often required to merge duplicate users, as Training Admins must perform extensive data extracts and re-uploads to resolve these collisions, which may originate, for example, from users changing their email.
This release introduces a new Replace User action on the Person object, which associates the new User with the target Person. Once launched, Vault automatically updates sharing settings on completed assignments and manages user status so that you can merge persons quickly and securely while maintaining data integrity.
eCOA
Studio
Compliance Schedule View Auto-On 26R1.3
This feature enables Sponsor/CRO Staff and Site Staff to view a participant’s survey compliance status in the scope of the study’s schedule of assessments. It enables users to quickly identify completed, missed, or pending surveys at specific protocol events.
Customizable End Status Labels Auto-On 26R1.3
This feature enables sponsor/CRO staff to customize the labels for the Study Withdrawn and Study Complete terminal events in Studio to match study protocols. Site staff see these updated labels when managing participant events.
eCOA Custom Site Roles Configuration 26R1.3
This feature enables eCOA Vault Owners and System Administrators to create custom site user roles with defined permissions in eCOA Vault. The eCOA application automatically adjusts the user interface for Site Staff based on their assigned role and permissions. This ensures site users only access the specific data and actions required for their clinical study responsibilities.
eCOA Assigned Access Permissions Auto-On 26R1.3
This feature enables Sponsor/CRO staff to require and assign a survey-specific permission to complete an eClinRO Assessment. Site Staff who do not have the required permission will be unable to complete the assessment, transcribe results, or change data.
Survey Schedule and Compliance Enhancements Available for Use 26R1.3
This feature enables Study Builders to configure survey schedules with the following enhancements:
- Availability window between specific times of day across multiple days
- Dynamic day-of-the-week token in the survey display labels
- Expected completion windows to distinguish between surveys submitted during the preferred period and those submitted late.
Enhanced Diary UI for Required Surveys Available for Use 26R1.3
This feature enables Study Builders to configure diary surveys with “for” or “between” type schedules in addition to as-needed schedules. MyVeeva Users can then access these required tasks through an enhanced interface that displays available and in-progress tasks alongside past completed entries.
Participant ID Instructions Auto-On 26R1.3
This feature enables Study Builders to define site-facing instructions on how to populate the Participant ID field. Site staff can view these instructions when creating or editing a participant record to ensure data accuracy.
Reusable Survey Translation Export and Import Auto-On 26R1.3
This feature enables Sponsor/CRO Staff to export and import translations on a per-survey basis to facilitate the reuse of localized content across different studies or in Library Manager. Users can download individual JSON files and upload to a specific survey so that the system matches the survey based on translation keys and source strings.
Reusable Survey Translation Export and Import Auto-On 26R1.3
This feature enables Sponsor/CRO Staff to export and import translations on a per-survey basis to facilitate the reuse of localized content across different studies or in Library Manager. Users can download individual JSON files and upload to a specific survey so that the system matches the survey based on translation keys and source strings.
Collection Document Enhancements Auto-On 26R1.3
This feature adds several enhancements to eCOA screenshot reports, including the ability for Sponsor/CRO Staff to generate reports for individual surveys. The reports also now include translation file details.
Bookmarks and Hyperlinks in EOSM and SDS Auto-On 26R1.3
This feature adds bookmarks and hyperlinks to the End of Study Media Participant Record and the Study Design Specification. Sponsor/CRO Staff and Site Staff can use these navigation tools to move between survey responses and corresponding audit records or browse the End of Study Media Participant Record by event and survey groupings. Sponsor/CRO Staff can use the bookmarks in the Study Design Specification to easily jump to specific sections of the document.
Ul Improvements for Multi-Part and Composite Surveys Auto-On 26R1.3
This feature adds the following usability enhancements for multi-part and composite surveys:
- Part Display Labels are displayed to Site Staff for multi-part and composite survey items in the Event Surveys List.
- Study Builders can view a Site Staff respondent chip in the Studio Surveys table for composite surveys with eClinRO parts.
- Sponsor/CRO Staff can view survey data grouped by part and parent survey in the correct sequence.
Summary of Change Character Validation Auto-On 26R1.3
This feature restricts Summary of Change field entries to 1,000 characters or fewer in Studio. This ensures that Study Builders do not encounter internal server errors when upversioning a study.
26R2 Feature Event Audits Auto-On 26R1.3
This feature ensures that relevant user actions associated with new features are recorded as audit events in the MyVeeva audit trail. This provides Sponsor/CRO Staff and Site Staff with a comprehensive record of system activity for compliance purposes.
Rule Instance Ranges and Custom Functions Auto-On26R1.2
Study Builders can reference a range or list of survey instances in rule expressions. The expression engine now includes four new custom functions to complete the following tasks:
- Get all matches
- Check all match
- Check any match
- Determine whether a date or time occurs in a defined time window
This reduces complex syntax, allowing users to build rules faster and more accurately.
Visual Analog Scale (VAS) Enhancements Auto-On26R1.2
Study Builders can configure Visual Analog Scales to include negative numbers, custom labels, answer images, percentages, and custom increments. This flexibility helps surveys meet specific survey design goals and supports more complex configurations.
Library Manager
Replaceable Tokens for Library Surveys Available for Use 26R1.3
This feature enables Sponsor/CRO Staff to create and use custom tokens as placeholders in library surveys. Sponsor/CRO Staff can update these token values when pulling a survey into a study collection to allow for study-specific customization of locked surveys.
Study Home
Compliance Dashboard Auto-On 26R1.3
This feature adds a dynamic compliance dashboard to Study Home, enabling Sponsor/CRO Staff to monitor study, country, site, and participant adherence in near-real-time. Users can configure a study-wide compliance threshold and apply multi-dimensional filters to identify performance risks and trends.
Handling Removed Sites Auto-On 26R1.3
This feature ensures that sites and countries that are deleted in eCOA Vault are automatically removed from Study Home and are not available to select when adding participants in eCOA (Sites). Sponsor/CRO Staff can generate accurate reports and site numbers can be reused for new locations without causing inaccurate records.
Populate QSCATID for Multi-Part and Composite Survey Scores Auto-On 26R1.3
This feature ensures that for multi-part and composite survey scores, the QSCATID column in the Survey Data report is populated with the parent user survey ID instead of null. This change aligns with single-part survey scores, which already populate the column with the user survey ID.
Clearer Reporting Outage InformationAuto-On26R1.2
During system outages, the reporting section in Study Home displays a specific unavailable message. Sponsor/CRO Staff can refresh the connection without disrupting their access to other tasks in Study Home.
eCOA API
Authenticated Discovery Endpoint for Inbound API Auto-On 26R1.3
his feature adds an authenticated discovery endpoint for the Inbound Participant API. Sponsor/CRO Staff can use existing credentials to direct sites to the proper region for authorization, ensuring a more secure and scalable integration.
eCOA (Sites)
Support for Multiple Caregivers Auto-On 26R1.3
This feature enables Site Staff to assign up to five caregivers to a participant and enables all assigned caregivers to receive survey tasks. When a caregiver starts a survey, they are given the option to claim the task, which then becomes unavailable to other caregivers to prevent duplicate entries. When starting surveys in-person, site staff must select which caregiver will be completing the caregiver surveys. Reports are updated to separate the Caregiver role and detailed Relationship information into different columns.
Handling Removed Sites Auto-On 26R1.3
This feature ensures that sites and countries that are deleted in eCOA Vault are automatically removed from Study Home and are not available to select when adding participants in eCOA (Sites). Sponsor/CRO Staff can generate accurate reports and site numbers can be reused for new locations without causing inaccurate records.
System Accessibility Enhancements Auto-On 26R1.3
This feature ensures system-wide accessibility by standardizing colors, icons, and primary action buttons throughout eCOA and MyVeeva for Patients to align with Web Content Accessibility Guidelines (WCAG). It updates the primary action button color from orange to blue and introduces consistent hover states to improve visibility and navigation for all users.
Clearer Reporting Outage InformationAuto-On26R1.2
During system outages, the reporting section in the Study Details tab displays a specific unavailable message. Site Staff can refresh the connection without disrupting their access to other tasks on the page as well as continue to complete other tasks in the site experience.
MyVeeva for Patients
MyVeeva for Patients
System Accessibility Enhancements Auto-On 26R1.3
This feature ensures system-wide accessibility by standardizing colors, icons, and primary action buttons throughout eCOA and MyVeeva for Patients to align with Web Content Accessibility Guidelines (WCAG). It updates the primary action button color from orange to blue and introduces consistent hover states to improve visibility and navigation for all users.
iOS 26 Support Auto-On 26R1.3
This feature ensures a consistent visual experience for users on Apple devices running iOS 26 or iPad OS 26. The system updates the application interface to align with built-in iOS styles.
Claiming Unlinked AccountsAuto-On26R1.2
MyVeeva users who have an account created during an in-person workflow will automatically have the system link the new study to their existing MyVeeva account. This provides users with a single account experience and removes the need to manage multiple logins when joining new studies.
Improved MyVeeva Access During Regional OutagesAuto-On26R1.2
MyVeeva users can access MyVeeva for Patients more reliably during regional system outages, because the system now processes authentication requests through more than one region if the primary region is experiencing an outage. Additionally, mobile users with biometric or PIN authentication enabled can access the app in an offline state automatically during the regional outage.
Commercial
Features in the Veeva Connections section also affect the Commercial application family.
PromoMats
Claims: Auto-Linking in Readiness PanelAuto-on26R1.2
Auto-Linking status is now available in the Document Readiness Panel, providing real-time insight into the progress of Text Asset Auto-Linking when it is initiated on an eligible document. This feature helps provide a clear and consistent way to assess the status of Auto-Linking on a document and to confirm when the process is complete. Learn more about the Document Readiness panel.
Claims: Entry Criteria to Validate References Are in Steady StateConfiguration26R1.2
A new entry criteria, All References are Approved, has been introduced to help ensure references linked to Text Assets are in a steady state. With this entry criteria configured as part of the Text Asset Lifecycle, the presence of a non-steady state reference linked to a Text Asset would prevent the Text Asset from moving to its steady state. Customers currently using the Text Asset Target Relationship Exists entry criteria can transition to using this to validate that all associated references are approved, rather than simply confirming that references have been linked.
eCTD: Auto-On Redline AnnotationsAuto-on26R1.2
The Redline Annotations feature for eCTD Compliance Packages has been enabled in all PromoMats Vaults. With this enhancement, all eCTD Compliance Packages with the Include Linked References setting set to Yes will be generated with redline annotations, rather than the legacy blue links. As part of this feature, the Add Annotations from Vault Links Application setting has been removed.
Claims: Remove Auto-Link on Initial Upload FlagConfiguration26R1.2
The Auto-Link on Initial Upload Admin checkbox has been fully removed, as detailed in this announcement. Customers can now use the Text Asset Auto-Linking Settings page to configure the specific document lifecycle states that trigger the Auto-Linking process following a document upload.
Claims: Deep DeleteAuto-on26R1.2
Deleting a Text Asset record now automatically deletes all related child records. Previously, if a Text Asset had any related child records, users were required to manually delete each of those before the parent record could be deleted. If a custom child object has been added to a Text Asset, its deletion rules must be set to Cascade delete children records to enable this functionality.
Claims: New References SectionConfiguration26R1.3
This release introduces a new References section that incorporates Platform features such as customizable column actions (edit and freeze columns) and the ability for users to comment. The new References section also includes more details on each reference, such as the anchor title, document lifecycle state, and document type. To use this new References section, Admins must manually replace the existing References section in the default Text Asset layouts with this version.
Claims: Text Asset Auto-Linking Settings EnhancementsAuto-on26R1.3
The current Text Asset Auto-Linking Settings page introduced in 26R1 is receiving an enhancement to allow Auto-Linking to be triggered by document lifecycle state changes. Previously, Auto-Linking was only triggered when documents were uploaded, but the enhancement now also triggers Auto-Linking when documents transition to configured lifecycle states.
This enhancement also removes the need to rely on the existing Suggest Links entry action. We recommend that users remove this entry action if it’s currently configured. This feature only applies to users that have Text Asset Auto-Linking Settings in use and is auto-enabled on release, requiring no additional configuration.
Note: As a reminder, customers must adopt these new settings to continue to trigger Auto-Linking upon initial document upload as the legacy Auto-Link on Initial Upload checkbox has been removed.
Rights ManagerAdmin Checkbox26R1.3
The new Rights Manager feature standardizes content licensing and consent tracking by elevating License and Consent Files. It is designed to assist DAM Librarians with renewals and Brand Managers with content reuse verification, establishing a solid foundation for compliance-driven decision-making.
This centralizes previously fragmented metadata across components into a record tied to the Rights document to easily validate and track expiration impact. This feature includes one new document type (Rights) with two subtypes (License and Consent), as well as a dedicated Rights Documents Lifecycle (Draft, In Review, Revise and Resubmit, Active, Expired or Revoked, and Superseded). There is also a Rights panel on the Doc Info page with color-coded status cards, and an Expiring Rights Report for items expiring within 90 days to reduce compliance risks.
This feature requires Admin configuration and does not automatically migrate existing custom rights implementations. We recommend that customers enable the 26R1 Standard Component Document Relationship type (components__v) for easier adoption of future feature enhancements. This feature provides visibility to the status of a Rights document’s relationship across components but has no automated enforcement or workflow actions against those components.
Metrics in PortalsAuto-on26R1.3
Metrics in Portals surfaces CLM engagement metrics directly within Portal to help brand teams make informed reuse decisions and evaluate performance of the material in the field. The metrics display a limited view within the Quick Look viewer side panel, only showing Document Views, Remote Documents Sent, Remote Document Views, Remote Document Downloads, and a Click-through Rate Percentage. The Click-through Rate Percentage is calculated by the number of Remote Presentation Views divided by the number of Remote Presentations Sent within the selected time window.
In order to configure this feature, the PromoMats-CRM Connection and CLM Content Metrics integration point must be enabled first. Metrics visibility is available to users with Read Reports and Dashboards as well as Read Document permissions. Learn more about the PromoMats-CRM Connection.
MLR: Reviewer QueueAuto-on26R1.3
This feature introduces a new Homepage view that prioritizes MLR reviewer tasks. Tasks are prioritized based on the Date of First Use (dissemination_publication_date__v), Task Due Date, and Content Type via four new Priority Document Type Groups.
Users can also designate documents as Expedited to have them bypass prioritization and appear at the top of the Reviewer Queue.
MLR: Improved Similarity ScoresAuto-on26R1.3
PromoMats now calculates the text portion of Content Similarity Scores by directly comparing two documents. Previously, the text portion of the similarity score was calculated using the relative similarity of keywords. This improved scoring method is more precise and ensures that scores are reproducible, independent of the broader data set.
MLR: Document CompareAuto-on26R1.3
PromoMats can now provide a visual comparison between two similar documents, allowing users to see both documents side-by-side with differences highlighted. This draws the reviewer’s attention to new or changed elements of a material that require more thorough review, improving the efficiency of the review process. This feature is available for documents with a content similarity score between 80-99%.
Multichannel
PromoMats: CLM PublishingAdmin Checkbox26R1.3
This feature introduces a more efficient way to publish CLM presentations for Vault CRM. It generates presentations as well as their individual slides out of PPT, PDF, and Word documents. This allows users to make edits to individual slides when needed while still benefiting from automation.
When a new version of a source presentation is uploaded, Vault automatically detects which slides have been added, removed, or updated, and it leverages the new CLM Slide Comparison UI to display a side-by-side view of the current and new versions for comparison.
Within minutes of up-versioning or initial creation, presentations are sent to Vault CRM over the standard Veeva Connection. This single automated process simplifies the content supply chain and eliminates the need for multiple publishing methods.
PromoMats: CLM Slide Comparison User InterfaceAuto-on26R1.3
The CLM Slide Comparison UI feature helps content creators and owners effectively review differences between current and new versions of CLM presentations. As part of our updated CLM Publishing, the system now automatically identifies revisions, including new, modified, or deleted slides, after the content creator uploads a new version. Users can see both the new and current presentations side-by-side, giving them a clear view of all the changes. This allows users to quickly approve the new version or make any necessary modifications before it’s synchronized to Vault CRM.
Medical
Features in the Veeva Connections section also affect the Medical application family.
MedComms
Scientific Statements: Auto-Linking in Readiness PanelAuto-on26R1.2
Auto-Linking status is now available in the Document Readiness Panel, providing real-time insight into the progress of Scientific Statement Auto-Linking when it is initiated on an eligible document. This feature helps provide a clear and consistent way to assess the status of Auto-Linking on a document and to confirm when the process is complete. Learn more about the Document Readiness panel.
References in SCP UIAuto-on26R1.2
References for Scientific Statements can now be managed directly from the Scientific Communication Platform (SCP) UI. A new dialog enables users to add or remove references and anchors for individual Scientific Statements.
Scientific Statements: Entry Criteria to Validate References Are in Steady StateConfiguration26R1.2
A new entry criteria, All References are Approved, has been introduced to help ensure references linked to Scientific Statements are in a steady state. With this entry criteria configured as part of the Scientific Statement Lifecycle, the presence of a non-steady state reference linked to a Statement would prevent the Statement from moving to its steady state. Customers currently using the Statement Link Target Relationship Exists entry criteria can transition to using this to validate that all associated references are approved, rather than simply confirming that references have been linked.
Enable Scientific Communication Platform UI in All MedComms VaultsAuto-on26R1.2
The Scientific Communication Platform (SCP) UI is now enabled automatically in all MedComms Vaults. This enhancement provides an improved experience for SCP users in MedComms, with all Communication Platform records using the application-specific interface by default. With this feature, the Enable Communication Platform UI Application setting has been removed, as detailed in this announcement.
Scientific Statements: Deep DeleteAuto-on26R1.2
Deleting a Scientific Statement record now automatically deletes all related child records. Previously, if a Scientific Statement had any related child records, users were required to manually delete each of those before the parent record could be deleted. If a custom child object has been added to a Scientific Statement, its deletion rules must be set to Cascade delete children records to enable this functionality.
Medical: Rights ManagerAdmin Checkbox26R1.3
The new Rights Manager feature standardizes content licensing and consent tracking by elevating License and Consent Files. It is designed to assist DAM Librarians with renewals and Brand Managers with content reuse verification, establishing a solid foundation for compliance-driven decision-making.
This centralizes previously fragmented metadata across components into a record tied to the Rights document to easily validate and track expiration impact. This feature includes one new document type (Rights) with two subtypes (License and Consent), as well as a dedicated Rights Documents Lifecycle (Draft, In Review, Revise and Resubmit, Active, Expired or Revoked, and Superseded). There is also a Rights panel on the Doc Info page with color-coded status cards, and an Expiring Rights Report for items expiring within 90 days to reduce compliance risks.
This feature requires Admin configuration and does not automatically migrate existing custom rights implementations. We recommend that customers enable the 26R1 Standard Component Document Relationship type (components__v) for easier adoption of future feature enhancements. This feature provides visibility to the status of a Rights document’s relationship across components but has no automated enforcement or workflow actions against those components.
Medical: Metrics in PortalsAuto-on26R1.3
Metrics in Portals surfaces CLM engagement metrics directly within Portal to help brand teams make informed reuse decisions and evaluate performance of the material in the field. The metrics display a limited view within the Quick Look viewer side panel, only showing Document Views, Remote Documents Sent, Remote Document Views, Remote Document Downloads, and a Click-through Rate Percentage. The Click-through Rate Percentage is calculated by the number of Remote Presentation Views divided by the number of Remote Presentations Sent within the selected time window.
In order to configure this feature, the Medical-CRM Connection and CLM Content Metrics integration point must be enabled first. Metrics visibility is available to users with Read Reports and Dashboards as well as Read Document permissions. Learn more about the Medical-CRM Connection.
MedInquiry
Note: With this release, the document viewer is changing with the Improved Document Viewer Platform feature, which affects what recipients see when they open a response package.
New Case Management User InterfaceConfiguration26R1.2
This release introduces the new Case Management UI, a new app page that helps users efficiently manage Medical Information cases with all Case and Case-related records available on one page. Admins can now configure the layout and fields displayed within this interface, ensuring that teams have a streamlined experience when logging cases, selecting contacts, and assembling and sending responses to HCPs.
Editable Standard Formatted Output TemplatesAuto-on26R1.2
This release includes a Platform enhancement that allows the Label, Output Format, and template file to be editable for standard Formatted Output templates, which were previously not editable. In MedInquiry, this applies to the Case Report for Attachment (case_report_for_attachment__v) template.
Create Inbound Interactions from Ingested EmailsConfiguration26R1.3
This feature introduces a new Inbound Interaction object in MedInquiry to capture incoming communications across email, phone, and chat channels. Once configured, when an email is ingested into MedInquiry, it automatically creates an Inbound Interaction record of type Email rather than immediately generating a Case Request. These interactions are visibly integrated into the Case Management UI timeline panel, further supporting streamlined Case processing.
Enhanced Logging for MedInquiry Email IntakeAuto-on26R1.3
When an email is ingested and the email processor fails to create any of the records (Case Request or Inbound Interaction, Case, Case Contact), Vault immediately notifies the Email Processor User about the issue. Admins can edit the notification template.
Add/Remove Responses Dialog EnhancementsAuto-on26R1.3
The Add/Remove Responses dialog is updated to display Responses across all lifecycle states. To provide better visibility and reduce user confusion, the dialog now explicitly shows the lifecycle state of each Response.
While only Responses in the Ready for Fulfillment state can be actively selected, users can now easily locate their prepared Responses and understand what steps are needed to make them available.
Case Management UI: Timeline PanelConfiguration26R1.3
A new configurable timeline panel in the Case Management UI displays inbound and outbound interactions, such as emails, phone calls, and chats. Users can now track communication histories with HCPs while simultaneously managing records like Case Requests and Adverse Events in the center panel. Admins can select which interaction types are displayed within the panel and which ones can be created.
Update Component Security on Name Field on User Role Setup (Case) ObjectAuto-on26R1.3
The User Role Setup object now supports system-managed naming. The Name field can be set to automatically generate record names using a standardized sequential format (for example, URS-MI-######).
Multichannel
Medical: CLM PublishingAdmin Checkbox26R1.3
This feature introduces a more efficient way to publish CLM presentations for Vault CRM. It generates presentations as well as their individual slides out of PPT, PDF, and Word documents. This allows users to make edits to individual slides when needed while still benefiting from automation.
When a new version of a source presentation is uploaded, Vault automatically detects which slides have been added, removed, or updated, and it leverages the new CLM Slide Comparison UI to display a side-by-side view of the current and new versions for comparison.
Within minutes of up-versioning or initial creation, presentations are sent to Vault CRM over the standard Veeva Connection. This single, automated process simplifies the content supply chain and eliminates the need for multiple publishing methods.
Medical: CLM Slide Comparison User InterfaceAuto-on26R1.3
The CLM Slide Comparison UI feature helps content creators and owners effectively review differences between current and new versions of CLM presentations. As part of our updated CLM Publishing, the system now automatically identifies revisions, including new, modified, or deleted slides, after the content creator uploads a new version. Users can see both the new and current presentations side-by-side, giving them a clear view of all the changes. This allows users to quickly approve the new version or make any necessary modifications before it’s synchronized to Vault CRM.
Vault AI for Medical
MedInquiry: Case Processing AgentConfiguration26R1.3
The MedInquiry Case Processing Agent is an AI-powered tool that automates the triaging and processing of incoming medical inquiries. By extracting key information from processed emails, the agent automatically generates the necessary Case Requests, Adverse Events, and Product Quality Complaint records. This agent reduces manual data entry for Medical Information specialists, allowing them to focus on resolving complex cases while expediting the overall response time.
Medical Product AgentConfiguration26R1.3
The Medical Product Agent for Vault Medical applications infers product details from unstructured text and provides the best matched records for standard Product data model objects, such as Product Family, Local Product, Product Form, and Product Variant. By providing a matching solution that can be called by the Case Processing Agent, this agent reduces the need for manual record selection, enabling automation and increasing efficiency.
Quality
Features in the Veeva Connections section also affect the Quality application family.
Batch Release
Configurable By ExceptionConfiguration26R1.2
With this release, when a Check Requirement’s Including Genealogy is set to By Exception, users can select Disposition Decision values in the Release Exception Decision field to determine when Vault should skip a related record. For example, prior to this feature, Vault skipped an item only when the Disposition Decision was Accepted. Now, this can be set to Accepted and Conditionally Accepted, or any other custom decision. When this field is null, Accepted will be skipped for backwards compatibility with existing behavior.
Item Creation Status BannerAuto-on26R1.3
A warning banner is now displayed on the Batch Disposition Execution Page when Batch Disposition Item creation is still in process or has failed. When item creation is complete, the banner will update with a link to refresh the page and view the completed items.
Qualification Check for Market-Ship DecisionsConfiguration26R1.3
Materials used to make a Finished Product are often qualified for use in specific finished material products for specific markets. This feature relates Qualifications to either Finished Material batches or Markets (Countries). To enable this, there are two new sections on Supplier Qualifications: Qualified Markets and Qualified Finished Materials. When these new sections are enabled, they can be evaluated when dispositioning a batch.
Market Specific Document CheckConfiguration26R1.3
Certificates are typically market specific and need to be created when a market-ship decision is being made for the selected markets. This feature enables document check requirements to be market specific and item requirements to be related to a market.
Disposition Plan Autonumber TitleAuto-on26R1.3
This feature adds a Title field to the Disposition Autonumber object so that it can be viewed when populating the Parent Plan for a Disposition Plan. This field is auto-populated with the latest version of the related Batch Disposition Plan Title.
Collapse Disposition HeaderAuto-on26R1.3
With this release, users can open and close the Disposition details section on the Batch Disposition Execution Page.
QMS
Qualification Support for LIMS & Batch ReleaseAuto-on26R1.2
This release introduces an enhanced qualification data model that expands support for LIMS and Batch Release. The data model changes will enable qualification approvals for any type of Material (Raw, Intermediate, Active Substance, and Drug Product) to be tracked against finished products and markets. These enhancements help organizations make informed decisions about releasing batches and defining material test requirements.
Audit Room: Configurable Lifecycle StatesConfiguration26R1.2
With this release, Audit Room now supports modifying state labels and adding additional state lanes. To improve organizational consistency and clarity, Admins can now relabel standard lifecycle states and state types. Additionally, the Audit Room now supports custom lifecycle states and state types, removing previous validations that limited the Inspection Request Lifecycle to standard states and state types.
A new Inspection Audit Room Owner application role enables designated users to configure the Audit Room experience. These users can adjust the display and order of Inspection Request Lifecycle states displayed in the Audit Room through a new configuration interface.
Audit Room Owners can now tailor the visual board to reflect their specific business process and include up to ten lifecycle state lanes, allowing Audit Room Owners to prescribe the exact order of state lanes in the Audit Room user interface.
This feature enables alignment with organizations’ internal terminology, allowing for more complex business processes and improved clarity, consistency, and overall management of inspection processes.
Learn more about configuring Audit Room.
Audit Room: Publish Inspection Request from Any Lifecycle StateAuto-on26R1.2
This feature removes the validation that requires users to move an Inspection Request into the Fulfilled state prior to moving it into the Published state. Users can now publish Inspection Requests from any lifecycle state. This allows customers to tailor the Audit Room to meet their needs and reflect their specific business process.
Audit Room: Multiple Scribe NotesAuto-on26R1.2
The Audit Room now supports multiple Scribe Notes, expanding beyond the previous single-note limitation. This enhancement allows audit teams to segment documentation by day, inspector, or specific area of scope, ensuring every detail of an inspection is captured with precision.
A new standard document classification, Scribe Notes (scribe_notes__v), is now available to allow users to attach multiple Scribe Note documents to a single Inspection record through the Audit-Document join object, in addition to the existing standard Scribe Notes field on the Audit object.
This feature also introduces a dedicated dialog in the Audit Room that users can open to view a list of up to 100 Scribe Note documents associated with the record, with pagination and scrolling for easy navigation. Selecting a document opens it in a new window for quick review.
Learn more about creating and contributing to Scribe Notes, and configuring Audit Room.
Display Record Count for QMS SectionsAuto-on26R1.2
To improve data visibility, Vault now displays real-time record counts for several QMS-specific sections. This allows users to immediately see the volume of associated data without needing to manually scroll or expand the section. The record count is now available for the following sections:
- Action Steps
- Notification Recipients
- Reason for Change History
- Record History
- Related Event
- Risk Builder
By aligning the behavior of these QMS sections with other Vault areas that already utilize record counts, this update streamlines the user interface and ensures a predictable and intuitive navigation experience across the entire platform.
This feature is automatically enabled for all Vaults.
Create Related Record: Copy Custom Fields for Referenced RecordsAuto-on26R1.2
With this release, the Create Related Record functionality is enhanced to ensure a more seamless data transfer during record creation. For Related Record Configurations that include reference records, the Create Related Record action now accurately copies all existing custom field values from the source-reference join records to the target-reference join records, prioritizing the existing data over system-defined default values. Prior to this release, custom fields on target-reference records would populate with default values, even if the source-reference record contained different data.
Learn more about Related Record Configurations.
Audit Room: Auto-Add Assignee to Fulfiller Role on Inspection RequestAuto-on26R1.2
This feature enhances the Audit Room feature by automatically adding the user selected as the Assignee on an Inspection Request record to the Fulfiller role within that record’s Sharing Settings, eliminating the need to manually add the user to the role.
QRM: Create Risks from Existing Risk AssessmentConfiguration26R1.3
In global life sciences, Quality Risk managers must often manually re-enter the same risk data for repetitive processes such as manufacturing identical products across different geographic sites. While Risk Templates allow organizations to add reusable risks into Assessments, this release introduces another option: The ability to copy existing Assessment Risks from approved Assessments.
Quality Risk managers can now run the new Create Risks from Existing Assessments user action from an Assessment’s record details page or from the Risk Builder (shown below).
After initiating the action, users can search for and select a single source Assessment. Vault only displays Assessment records in the lifecycle states defined by the user action configuration, and that match the parent Assessment’s object type (for instance, pFMEA). Users can add more filters and use search to further refine the list of available source Assessments.
After selecting an Assessment, users can select one or more Assessment Risk records in the selected source Assessment that match the lifecycle states specified by your Admin’s configuration.
After users click Save, Vault creates new Assessment Risk records by copying the selected Assessment Risk records and associating them with the target Assessment. Vault copies standard and custom fields, excluding fields related to the Risk Matrix selected on the Assessment, such as Risk Scores (RPN). Vault does not copy any related Mitigation Action records. If the source Assessment Risks have related Process Steps, Vault searches for Process Steps with matching names in the target Assessment and associates them with the newly created risks. Vault automatically creates Process Steps if no matching Process Step exists. Vault copies related Hazard information using similar logic. The new Assessment Risk records maintain traceability through an automatically populated Source Risk field.
When the background process is complete, Vaults sends a notification to the initiating user with a CSV file containing details of successes and any failures.
Adopting this configurable feature helps organizations optimize Risk Assessment creation for identical processes across sites. It reduces variability of risk definition and saves time by eliminating repetitive manual data entry, allowing Risk Managers to focus on risk evaluation.
QRM: Manage Related Documents in Risk BuilderAuto-on26R1.3
Risk management often requires documentation to support assessments, mitigation strategies, and regulatory compliance. Risks are typically associated with documents like SOPs, validation reports, or mitigation evidence.
Organizations use a variety of configuration approaches to establish the relationship between Assessment Risk records and supporting documentation using error-prone and time-consuming custom workarounds.
This feature standardizes the relationship between Assessment Risks and their supporting documents and simplifies the process of linking documents to risks by allowing users to add, remove, and view an Assessment Risk’s supporting documentation without leaving the Risk Builder interface.
A new Assessment Risk Documents column is now available in Risk Builder. Admins can add this column to the Assessment Risk section of an Assessment layout so it appears by default, and users with the appropriate permissions can add and adjust the placement of the column themselves within the Risk Builder.
When the Risk Builder is in view mode, cells in the Assessment Risk Documents column display View Assessment Risk Documents if an Assessment Risk is related to at least one document, as shown below.
When a user clicks View Assessment Risk Documents, a dialog opens displaying the related documents.
Users can click on a document’s name to open it in a mini browser window for quick reference.
When the Risk Builder is in edit mode, users can hover over the related documents cell of an Assessment Risk without associated documents to display an Add Document (+) icon.
Clicking the icon opens a dialog that allows users to select documents to relate to the Assessment Risk. Up to ten documents can be associated with a single Assessment Risk. The list of available documents consists of the latest version of all documents the user has permission to view. Selecting a document links its latest version to the Assessment Risk record.
When the Risk Builder is in edit mode, related document cells for Assessment Risks that have at least one document display Edit Assessment Risk Documents, as shown above. When a user clicks the text, Vault displays a dialog that allows users to add or remove related documents.
Users can filter Assessment Risks in the Risk Builder grid to show only rows for risks that have or do not have associated documents.
If your Vault uses the Periodic Risk Review feature to automatically create Assessment Risk History records for each risk when an Assessment is approved, Vault now captures the specific version of each risk’s linked documents at the time the Assessment’s history is generated in the Assessment Risk History records.
This feature applies to any type of Risk Assessment that utilizes Risk Builder. It helps organizations optimize the way they link documents and risks by eliminating the need to navigate between different Vault tabs, keeping users focused on the risk assessment. It also ensures that the specific versions of supporting evidence used during an assessment are captured in the history record.
Complaint ContactsConfiguration26R1.3
With this release, Veeva QMS updates the method for capturing and managing contacts in complaints processing from static Person reference fields into a scalable, transactional Complaint Contact object and data model. Copying Person reference fields as part of promoting a Complaint Intake to Complaints can lead to Person information becoming out-of-sync on the related Complaints, potentially introducing compliance and audit risks.
This feature introduces a data model that integrates the new Complaint Contact object across Complaint Intakes, Complaints, and Complaint and Medtech Complaint Quality Events, and supports External Notifications. For details on External Notifications enhancements related to Complaint Contacts, see the following related 26R2 features: External Notifications: Complaint Enhancements and External Notifications: Support for Complaint Intake.
To achieve this flexible architecture, the following enhancements are available:
- Complaint Contact object: A child object of the Complaint Intake object, this object lets users create contacts specific to a Complaint Intake. Users can add one Initial Reporter type Complaint Contact, and multiple Complainant type records. Custom object types are supported.
- Master Complaint Contact object: Stores reusable data for Complaint Contacts. Admins can configure Action Triggers to populate a Complaint Contact with values from a selected Master Complaint Contact record, significantly reducing manual data entry errors.
- Complaint Contacts application section: While Complaint Intakes use a standard related object section to display Complaint Contacts, Complaints and Quality Events now have a dedicated Complaint Contacts application section. This section displays all contacts linked to the source Complaint Intake record. For users with appropriate permissions, this view supports direct record creation, action ellipsis navigation, and instant inline editing of contact records directly from the target Complaint or Quality Event records. In order to use this section, Complaints and Quality Event Complaints must have the Complaint Intake reference field populated.
For offline documentation and reporting, the standard Download as PDF action has been enhanced to support the Complaint Contacts section. The generated PDF fully respects user sorting preferences and field view permissions.
To configure this feature, Admins must add the Complaint Contacts related object section to a Complaint Intake object layout, and the Complaint Contacts application section to the existing page layouts of the Complaint or Quality Event (Complaint) object.
Note: Complaint Contacts can be used for one Complaint object type (for instance, Medtech Complaint) while other object types can continue using Persons.
External Notifications: Support for Email Document AttachmentsConfiguration26R1.3
This feature enhances the External Notifications feature by allowing users to send documents by email as attachments rather than only as links, allowing organizations to securely share critical files directly with external stakeholders. This enhancement is available for all objects supported for External Notifications. Admins can automate standard attachment rules when configuring the Send External Notification action as an entry action or event action, and grant users the ability to choose the best delivery method for notifications triggered via user action.
To configure this feature, Admins must first enable the global Enable Document Attachments on External Notifications setting by navigating to Admin > Settings > Application Settings > QMS. This two-way flag exposes the Send Documents as Attachment configuration option for the Send External Notification user, entry, and event actions.
For event and entry actions, this option determines whether documents are included in an external notification as links or attachments. If this option is enabled for user actions, after selecting a document, users can choose between sending it as Attachments to Email or as a Link in Email (the current default).
No additional permissions are required to send documents as attachments.
Note: Vault limits each email to a maximum of five attachments and a total file size of 15,360 KB.
External Notifications: Support for Complaint IntakeConfiguration26R1.3
This feature enhances the External Notifications feature by introducing support for Complaint Intake records. Admins can now configure External Notifications to send automated email acknowledgements directly from Complaint Intake records to the associated individuals outside your organization, such as the reporter and complainant.
Using External Notifications for the Complaint Intake object requires use of the new Complaint Contact object to manage information about recipients.
External Notifications for Complaint Intakes are supported only for standard Complaint Intake object types. External Notifications can only be sent to individuals identified as Complaint Contacts on Complaint Intake records.
External Notifications: Complaint EnhancementsAuto-on26R1.3
To support the Complaint Contacts feature, External Notifications now support the use of Complaint Contacts on Quality External Notification Templates for the Complaint and Quality Event objects (Complaint and MedTech Complaint object types). For these objects only, an Admin must select an option from the new Recipients Option drop-down. For existing templates, the option defaults to blank (which Vault interprets as the pre-existing Persons functionality), and no changes are required.
However, if using the features introduced in 26R2 that require Complaint Contacts, Admins must select the Complaint Contacts option. Admins can select up to ten active Complaint Contact object types as the default recipients for outbound communications.
Additionally, the corresponding Send External Notification action (user, event, and entry actions) has been enhanced to utilize the default Complaint Contact Types defined in the template and enforce automated data validations. For event and entry actions, Vault skips contacts if the Complaint Contact is inactive, missing an email address, or has Consent to Contact set to No or blank, and continues processing all eligible contacts. For the user action, users must correct validation failures manually.
External Notifications: Alternate Communication LanguagesConfiguration26R1.3
This feature introduces the ability to automatically send external email notifications in the language specified on Complaint Intakes, Complaints, and Quality Events (restricted to the Complaint and Medtech Complaint object types). This functionality significantly improves global stakeholder management and regulatory alignment by ensuring that critical compliance and follow-up communications are seamlessly delivered in the recipient’s preferred language.
A new Communication Language (communications_language__v) field is now available for activation on the Complaint Intake, Complaint, and Quality Event objects. It allows users to capture a preferred communication language when creating a record.
Admins can enable translation of external notifications across user, entry, and event actions by selecting Use Communications Language to translate notifications within the Send External Notification action’s configuration.
When sending an external notification, Vault evaluates the record’s Communication Language field and applies the translated notification template that corresponds to the language selected on the record. If a translated template is unavailable, Vault utilizes the default notification template to ensure an uninterrupted process flow.
Organizations can efficiently scale and manage their localized notification templates using the standard bulk translation import capabilities under Settings > Language & Region Settings.
Additionally, this feature works together with the 26R2 External Notifications: Preview Email feature, allowing users to preview the outbound email in the alternative language of the selected template.
External Notifications: Preview EmailConfiguration26R1.3
With this release, the External Notifications feature is enhanced to allow users to review complete renderings of external correspondence directly within the user action dialog prior to sending an external notification. This functionality is available for all objects supported for External Notifications.
Admins can configure this feature by enabling the new Allow user to preview email? checkbox on the Send External Notification user action configuration.
With this checkbox selected, upon running the Send External Notification user action, the resulting dialog displays the complete HTML version of the email body and subject line. The Preview Email panel populates all the tokens defined in the notification template and dynamically refreshes if the user updates the notification template selection. Note that some tokens including links may not display as expected.
Preview Email integrates with the new External Notifications: Alternate Communication Languages feature for Complaint Intakes, Complaints, and Quality Event (restricted to the Complaint and MedTech Complaint object types), automatically applying the translated notification template to the preview according to the communication language defined on supported records.
This feature mitigates the risks associated with unverified emails by allowing users to visually confirm correct notification template selection, accurate token-merging, and multilingual translations before correspondence reaches external recipients. This proactive visual review directly prevents data errors, reduces compliance risk, and ensures professional communications with external email recipients.
Complaint Intake Follow-Up: Manual ProcessingConfiguration26R1.3
This feature introduces a standardized approach to manage outbound follow-up requests for information about complaints from a Complaint Intake record, eliminating the need for custom solutions.
This feature introduces the Complaint Intake Follow-Up object. Each time additional information is needed about a Complaint Intake, users can create a Complaint Intake Follow-Up record. Vault maintains a relationship between each Complaint Intake Follow-Up record and its source Complaint Intake record.
A new Complaint Intake Follow-Ups section is available for Complaint Intake records that shows all related Complaint Intake Follow-Ups, providing a consolidated view of all outbound requests for more information. Users can also create Complaint Intake Follow-Ups from this section.
A new Follow-Up Attachments section is also available for Complaint Intake records, shown above. Users managing Complaint Intake Follow-Ups often upload files as attachments (for instance, pictures or emails) to these records to capture information sent and received from reporters, complainants, or any Complaint Contact. The Follow-Up Attachments section shows attachments across all Complaint Intake Follow-Up records tied to the related Complaint Intake record, eliminating the need to click into each Complaint Intake Follow-Up record to view its attachments.
The Complaint Intake Follow-Ups and Follow-Up Attachments sections are also available for standalone Complaints, Quality Event Complaints, and Quality Event Medtech Complaints. In order to use the new sections on Complaints or Quality Event Complaints, the Complaint must have the Complaint Intake reference field populated.
A Complaint record’s Complaint Intake Follow-Ups section shows all of the Complaint Intake Follow-Ups related to the source Complaint Intake record. Users can create Complaint Intake Follow-Ups from this section and update existing follow-ups.
The Follow-Up Attachments section shows all attachments across all Complaint Intake Follow-Up records that are related to the Complaint ‘s source Complaint Intake record, providing a view of all attachments associated with a Complaint’s follow-ups in one location.
The new sections described above are included in the generated PDF when a user executes the Download as PDF action.
Complaint Email Ingestion: Support for MedTech Complaint Intake Object TypeConfiguration26R1.3
Life sciences organizations receive complaints from consumers and healthcare providers. These complaints are often captured through a website or call center where they are logged in a Customer Relationship Management (CRM) system. When a potential complaint requires quality assurance attention, Veeva QMS customers can create and triage Complaint Intake records to determine if a Complaint record is necessary.
There are multiple methods of creating Complaint Intake records in a Quality Vault, including manually, through a custom integration with an external IT system, or based on an email sent to a configured Vault inbox using the QMS Complaint Email Ingestion feature.
This feature enhances the Complaint Email Ingestion method by adding support for the MedTech Complaint Intake object type of the Complaint Intake object. Admins can now select the MedTech Complaint Intake object type when configuring an Inbound Email Address Configuration, as shown below.
Promote to Complaint Enhancement: Ignore Inactive FieldsAuto-on26R1.3
The QMS Promote to Complaint action now ignores any inactive matching fields when copying field values from source Complaint Intake and Reported Product records to any generated Complaint records. Vault now strictly maps only to active fields.
Previously, if an active custom field shared a similar name with an inactive standard field, Vault prioritized the inactive field, potentially leading to data omission and mapping conflicts.
Related Batch Application SectionConfiguration26R1.3
Quality processes such as Deviations and Complaints frequently impact multiple manufacturing batches. Efficiently tracking these relationships is vital for accurate impact assessments. Prior to this release, adding batches to process records required users to perform multiple clicks to find a batch by name. Admins can now add a new Related Batches application section to a process record’s layout, and configure default filters for key Batch fields that users can populate to narrow their search when adding or removing Batch records.
The following screenshot shows a comparison of the dialog previously used to select a batch, and the new version of the dialog:
When adding Batches from the Related Batches section on a process record, the record selection dialog displays Admin-configured default filters and operators, such as starts with or is in the range. Users can add, remove, and change the filters and operators as needed. Each time a user opens the new batch record selection dialog, the configured filters display by default.
The dialog displays up to 50 batch records per page, and can include up to 20 pages for a total of 1,000 records. Batch records already associated to the process record are not available for selection when adding Batches. Users can add, remove, or reorder the columns in the dialog, and Vault automatically saves the changes as the user’s column preferences for each type of process. Batch records are shown in ascending order by name, and users can change the sort order.
As a user enters values in the filters, Vault automatically applies them to the list of Batch records shown at the bottom of the dialog. Users can now select multiple batches to assign to the process record in a single operation. The dialog includes an option to select or unselect all records that appear in the filtered or unfiltered list. Clicking OK links the selected batches to the process record by creating a join record between the Batch and process record and returns the user to the Related Batches section, where the linked batches are shown. The following screenshot shows a comparison of the Batches related object section, and the new Related Batches application section.
Users can use the new section to perform inline edits on the related Batch records’ fields directly from the process record. If a modified field uses the Reason for Change feature, Vault prompts for a reason.
When a user executes the Download as PDF action on a process record that contains the Related Batches section, the resulting PDF displays up to 1,000 associated Batch records.
This feature is available for use on any process record that uses a standard or custom join object to associate a process record to a Batch record. It minimizes clicks and streamlines the process of Batch record association.
External Collaboration Checklists: Display Follow-Up Interface on Completed ChecklistsAuto-on26R1.3
The Supplier Questionnaires: Follow-Up & Invite Respondents feature is now enhanced to ensure a consistent visual experience and better data accessibility throughout the record lifecycle.
With this release, the Follow-Up Questions and Responses interface displays in read-only mode after a checklist reaches the Completed state. Prior to this release, follow-ups were unavailable once a checklist was finalized.
External Response Collaboration: Inspection RequestsConfiguration26R1.3
Previous Veeva QMS releases introduced the External Response Collaboration feature to provide business partners with temporary, short-term, streamlined access to certain records for the purpose of completing specific tasks within quality business processes. This release expands the External Response Collaboration feature’s supported quality business processes to include Inspection Requests.
This enhancement streamlines the inspection process by allowing external collaborators from a business partner to securely access and contribute information to inspection requests. Enabling suppliers to work directly within your organization’s Quality Vault eliminates delays caused by email-driven communication, reduces the administrative burden on internal teams, and ensures all parties operate from a single source of truth.
This feature requires Admin configuration.
Audit Room: Expanded Inspection Request CardConfiguration26R1.3
The Audit Room feature delivered in the Veeva QMS 24R3 release introduced a streamlined user experience for managing Inspection Requests. Audit Room now enables organizations to display additional key fields on each Inspection Request card. This helps users quickly assess important information without opening the Inspection Request record.
The following screenshot shows the Audit Room page with a new Configure Fields to Display menu option, available under the Audit Room Owner menu. This menu is only available to users placed into the Audit Room Owner application role on Audit and Inspection Request records.
Selecting the new menu option opens the dialog shown below. This dialog presents the Audit Room Owner with a list on the left of available Inspection Request fields. Moving fields to the list on the right side will make them visible on Inspection Request cards. Audit Room Owners can add up to five fields to display.
Users can show and hide the fields selected by an Audit Room Owner by clicking on Show fields or Hide Fields at the bottom of Inspection Request cards. The screenshot below shows several Inspection Request cards with their fields displayed.
Users can click Hide All Fields at the top of the Audit Room page to hide fields on all Inspection Request cards. This text changes to Show all Fields when none of the Inspection Request cards are displaying fields.
This feature requires an Admin to add the Audit Room Owner application role to the Audit and Inspection Request lifecycles and grant the role permission on the appropriate lifecycle states. It also requires an Admin to configure a method of adding a user into the Audit Room Owner application role for each Audit and Inspection Request record.
Reason for Change: Support for Test Plan & Effectiveness CheckConfiguration26R1.3
Introduced in 24R3, the Reason for Change feature allows organizations to capture the rationale behind updates to data fields on completed records. To provide more robust and unified audit capabilities across quality processes, this release expands the list of supported QMS standard objects to include the Test Plan (test_plan__v) and Effectiveness Check (effectiveness_check__qdm) objects.
Create Related Record: Validation UpdatesAuto-on26R1.3
This release introduces two new validations that Admins should be aware of to prevent the Create Related Record feature from encountering errors.
Configuration Validation
With this release, Admins cannot select a Vault system object (objects with a __sys suffix) in the Source Object field when creating a Related Record Configuration.
Runtime Validation
Using pre-existing functionality, Admins can configure a Related Record Configuration to automatically include the source record’s related reference records in the newly created record, as shown below.
When an Admin creates or edits a Related Record Configuration, it is not possible to select an inactive join object as the Source-Reference Record. However, an Admin could inactivate the join object while it is still referenced in the Source-Reference Record field of a Related Record Configuration. With this release, users performing an operation that executes the Create Related Record action as a user or entry action now receive a notification displaying an error message. The error message indicates which source join object is inactive, and Vault does not create a related record.
Both validations are automatically enabled in this release.
Vault AI for Quality
Complaint Agent & Deviation Agent: User Experience ImprovementsAuto-on26R1.2
This feature enhances the user experience for Investigation and CAPA Plan summary generation to allow users to navigate away from the record during narrative summary generation. When users navigate back to the record after summary generation is complete, they can review and save the generated summary. This feature also improves the readability of generated summaries.
Learn more about using the Complaint Agent and Deviation Agent.
Complaint Agent: Medtech ComplaintsConfiguration26R1.2
This feature expands the Generate Investigation Summary and Ask Questions capabilities to Medtech Complaints. With this enhancement, Vault can now provide on demand, narrative-style summarizations of the Investigations related to Medtech Complaints, providing the user with an opportunity to review, modify, and save the summarized content to the record. Beyond summarization, the Complaint Agent can now also answer questions about a Medtech Complaint via the Veeva AI Chat interface.
Learn more about configuring the Complaint Agent.
All Quality Applications
Display Application Configurations by Enabled ApplicationAuto-on26R1.3
Previously, the Application Configurations section on the Admin > Configuration page displayed the majority of the configuration options for the Quality Suite of applications in a list. Every Quality application’s configuration option was displayed, even if the Vault did not utilize the application.
In the 26R2 release, the Admin > Configuration page now displays configuration options by application, and only displays configuration options for applications that are enabled in your Vault. Configurations that are common across all Quality applications display in a common section. The only exceptions are the following, which remain in the Platform Configurations > Business Logic section:
- Quality Teams
- Quality External User Templates
- Quality Record Checks
- Quality Relationship Automation
The screenshot below shows a before and after comparison when all Quality Suite applications are enabled in a Vault.
The reorganized display still respects user permissions.
Here are some examples of how the new Admin > Configuration screen will determine when a Quality application’s section appears.
- If a customer does not have the Validation Management application enabled in their Vault, the Admin > Configuration screen will not display the Validation Management section or the associated links to that application’s configuration options to any Admin user.
- If an Admin has the Validation Management application enabled in their Vault, but does not have permission to access the application’s configuration options, the Admin > Configuration screen will not display the Validation Management section or configuration options.
This feature provides an improved user experience with application-specific context for Quality Suite application configuration options. It is automatically enabled for Admins in the 26R2 release.
Surveillance
VPS: Generate AER SnapshotConfiguration26R1.2
Vault Product Surveillance now generates a point-in-time snapshot of relevant data when submission documents are created, ensuring that the information leveraged in the Vault UI and PDF and XML submissions are synchronized and immutable post-approval. This allows users to view historical versions of submitted AERs in a read-only view directly within Vault, rather than needing to pull down prior versions of generated PDFs or XMLs to view or compare changes over time.
The VPS: Generate Submission Documents action is at the heart of this update, as it now additionally captures relevant data at the time of generation so that it can be stored and used in the future when this version of the AER is viewed within Vault. Best practice flows will ensure that once these submission documents are generated, the UI of the AER will be read-only in Vault unless the AER itself is returned to a Drafting state.
VPS: Concomitant Products and Related AER ImprovementsAuto-on26R1.2
This release introduces several smaller changes to add support for a Related Adverse Event Reports data grid in H10 and AER management regarding Concomitant Products.
Section H10 will now provide the ability to include a data grid referencing either related AER numbers (if the AERs are present in the system) or a text field for a manually captured AER Report Number if the reference exists outside of the Vault.
The concomitant product enhancements include adding a new field to capture concomitant product text for PMDA, EU/MIR, and eMDR, as well as updated logic to intelligently source information either directly from the system if it exists (if Concomitant Product Available in System is true) or from this new dedicated field, if not.
VPS: Configurable Data MappingConfiguration26R1.3
This release introduces a major enhancement to Vault Product Surveillance’s (VPS) Adverse Event Report (AER) management engine, allowing organizations to provide configurable mapping overrides for data within their AER and Health Authority forms to the source objects and fields configured within their Vault.
Today, the Veeva Product Surveillance application currently generates Adverse Event Reports dynamically using a runtime lookup model. Data is aggregated from various source objects (Complaint, Product, Organization, etc.) and mapped to Health Authority (HA) specific XML and PDF templates.
With this feature, the new Configuration Mapping Overrides component, available to Admins, provides a clear in-application toolset to define where data should be sourced within your organization’s Vault for the preparation of various HA forms. Core data for these forms can be sourced from anywhere within the supported object hierarchy (AER, AER > Complaint, AER > Complaint > Product | Product Variant | Product Family) and mapping overrides are available for text/long text, date, number, picklist, and boolean fields.
Mappings are leveraged when the AER’s forms are generated, so data at rest is not impacted immediately with this feature. Admins will see new mapping tools available, but no functional changes to AER generation will be implemented unless configuration to those components is performed.
VPS: Product Attributes ManagementConfiguration26R1.3
This feature enables users to manage and build variable product master data directly from the Adverse Event Report (AER) user interface, generating more clear and specific AERs with the most relevant labels and terms, sourced directly from your Vault. This enables organizations to support AERs in scenarios where a product such as “Cholecap” may have a local name like “Chole-J” in some regions due to trademarks, and which guides users to select the most appropriate terminology within their AER flows.
When populating product information during the AER flow, Vault now traverses your product hierarchy to find the most relevant regional or global attribute labels to present this to your users. Users can modify this suggested value if appropriate, or supplement it with additional information either sourced from your Master Data set or with net-new values provided by the user within the AER. When a net new local variation is presented by the user, Vault captures this information from the AER and pushes it back into the Master Data set for future reference and use.
Admins can enable the Smart Defaulting for Product Attributes setting to enable this functionality. After doing so, Vault will begin referencing the new Regional Product Attribute (RPA) data now available in your Vault, supported by Nomenclature Code information and Product Information / Categorization data, which Admins will want to review in order to set up initial Master Data sets.
VPS: Contact Information ImprovementsConfiguration26R1.3
This release evolves the process of managing contact information within Adverse Event Report (AER) flows, introducing a new user interface to streamline the entry of contact and address information for Adverse Event Reports. Vault will intelligently default known information from source records, allow selection from valid existing options, or permit manual selection and entry where needed. These improvements are available for all contact roles required on Health Authority (HA) forms, including Manufacturer, Authorized Representative, Importer, Manufacturing Site, Complainant, Reporter, Report Submitter, Reprocessor, and MDR Contact. These changes result in a reduction in the number of clicks required to enter contact information in most locations, and generally higher fidelity data across AERs wherever contact information is referenced.
HACCP
HACCP Flow Diagram: Display Risk Category Field as Clickable LinkAuto-on26R1.3
In the Risk Assessment section of the Information panel, the Risk Category field now displays as a clickable link. Users can click the link to open the Risk Category record in a small browser window.
HACCP Flow Diagram: Update to Label of Hazard Significance Filtering OptionAuto-on26R1.3
Currently, users can filter Process Hazard Analysis records in the Hazard Analyses section of the Information panel according to the value of the Is Significant Hazard? field. With this release, the label of the “Insignificant” filtering option is updated to “Non-significant”.
HACCP Flow Diagram: Intermediate State for ConnectionsAuto-on26R1.3
When creating, editing, or deleting a connection between two steps on the HACCP Flow Diagram, the connection’s line now changes to gray and users cannot make further edits to that connection while Vault saves the changes.
QualityDocs
Cascade DCC Roles to Documents on Task ReassignmentConfiguration26R1.3
This feature allows Vault to cascade document lifecycle role assignments down to the governed documents when users reassign a Document Change Control (DCC) workflow task. Previously, Vault did not cascade roles upon task reassignment. This meant that new assignees could be tasked with reviewing or approving a workflow without having the required permissions to view all associated documents.
With this update, Vault ensures that new participants automatically receive the appropriate permissions to view and interact with every document in the workflow, preventing blind approvals and saving time spent manually adjusting document access.
Document Change Control: Display Related DocumentsConfiguration26R1.3
Admins can now configure Vault to include the Add Related Documents button on Document Change Control (DCC) workflows to help you quickly pull associated files into your change control records. The Add Related Documents button is available in the Change Authorization, Documents to be made Effective with Workflows, and Documents to be made Obsolete with Workflows sections of a DCC record. The legacy sections Documents to be made Effective and Documents to be made Obsolete do not support this feature.
Previously, managing a large volume of DCC records presented challenges, as users could easily overlook bidirectional relationships or fail to manually associate critical related documents, such as translations.
With this update, clicking the button opens a dialog that displays all bidirectional related documents for files currently in that section. The existing section filters persist inside the dialog, allowing the user to easily find, select, and add multiple relevant documents to the current section at once.
When the user clicks the button from a supported DCC section, Vault dynamically displays the associated files. Users can then search for specific related documents, select the desired files, and add them directly to the DCC section.
Document Change Control: Unlink Document Change Request & Periodic ReviewAuto-on26R1.3
With this release, Vault now automatically unlinks associated Document Change Request (DCR) and Periodic Review (PR) records from a Document Change Control (DCC) record when the related target documents are removed from the Documents to be made Effective with Workflows and Documents to be made Obsolete with Workflows sections of the DCC. The audit trail for the DCC and the corresponding DCR and PR records show that the unlinking was performed by the System on behalf of the user who removed the target document. The legacy Documents to be made Effective and Documents to be made Obsolete sections do not support this enhancement.
Quality Relationships Panel Enhancement: Support for Archived DocumentsAuto-on26R1.3
The Document Relationships section of the Quality Relationships panel now displays archived documents with an associated icon. As with all related documents, archived documents are only visible to those with the appropriate permissions.
Process Navigator Enhancement: Improve Object Reference Field Value SelectionAuto-on26R1.3
This enhancement improves the performance and load time of object records when selecting values in Object Reference fields in the Dynamic Association Criteria feature of Process Navigator.
Document Control Homepage Enhancement: Improve Object Reference Field Value Selection in Filter CriteriaAuto-on26R1.3
This enhancement improves the performance and load time of object records when selecting values in Object Reference fields in a Document Control Homepage section filter.
Periodic Review: Reassignment of Inactive Document OwnerConfiguration26R1.3
Vault now prevents periodic review jobs from failing when they encounter an inactive document owner. This feature ensures continuous compliance by routing the document to a backup group for reassignment instead of causing silent errors.
Previously, if a document owner became inactive before or during a periodic review cycle, the scheduled job would fail for that document without creating the necessary periodic review record. Because Vault sent no notification, Document Control users remained unaware of the issue, creating compliance gaps.
With this update, Vault handles inactive owners dynamically based on when the user is inactivated, ensuring that your periodic review processes proceed smoothly and transparently.
The system manages inactive owners in two ways depending on when the user profile is deactivated:
- Inactivation Before the Periodic Review Job Runs: If a document owner is inactive when the job executes, the job no longer errors out. Instead, Vault initiates a standard object workflow and generates a Prompt for Fields workflow task. This task becomes available to users in a configured document role group (such as a quality assurance group). The user who accepts the task selects a new owner, which executes a system action to update the Document Owner and the Periodic Review Owner.
- Inactivation After the Periodic Review Job Runs: If the owner is deactivated after the periodic review record is already created, a new user action is available. Users in the configured group can manually execute the Reassign Owner on Document & Periodic Review action from the document. This single action allows the user to select a new document owner, automatically updates both the document and Periodic Review record owner fields. To support this behavior, Admins can navigate to Admin > Settings > Application Settings > QualityDocs to find a new configuration section. From there, you must select the specific document role user group that should receive the Prompt for Fields workflow task and gain access to execute the manual user action.
Admins must configure Dynamic Access Control on the Periodic Review object to enable this feature.
Process Navigator: Support Ten Dynamic Document Association Criteria RulesAuto-on26R1.3
This feature increases the number of Dynamic Document Association Criteria Rules that can be created for a given process from 3 to 10.
Process Navigator Enhancement: Support None Operator in Dynamic Document AssociationsAuto-on26R1.3
Vault now supports the Is none of the following operator in Dynamic Document Association Criteria Rules in Process Navigator. This operator enables users to select values to exclude from the dynamic matching. Previously, it was only possible to select all of the values to be included.
External Collaboration Enhancement: Data Model UpdateAuto-on26R1.3
With this release, the Display in Business Admin checkbox on the External Collaboration Reassign Tasks object configuration is now read-only and no longer editable.
External Collaboration Enhancement: Display Veeva Quality LogoAuto-on26R1.3
With this release, the “Veeva Quality” logo displays on the document viewer for the steady state document that is sent to external collaborators via a secure public access link after the external collaboration has ended.
Document Change Request: Add Target Document OwnerAuto-on26R1.3
With this release, Vault automatically adds the Document Owner of the target document associated with a Document Change Request record to the Target Document Owner role in the Sharing Settings of the corresponding Document Change Request record with both Read and Edit permissions.
If the target document for the DCR is changed, the new Document Owner is added to the Sharing Settings but the original Document Owner is not automatically removed. If the Document Owner on the target document changes, the existing DCR Sharing Settings are not automatically updated with the new owner.
QualityDocs, Station Manager
QualityDocs & Station Manager Objects: Add Key Document FieldsAuto-on26R1.3
This feature introduces standard, system-managed document fields on key QualityDocs and Station Manager objects. This update allows users to view critical metadata directly on object records that reference a target document, ensuring data visibility across your quality processes without manual tracking. Previously, users could not easily view document-level metadata like the document title or department from the object record detail or list views. This required navigating back and forth between the object record and the document itself.
With this feature, Vault uses object formula fields to dynamically display current document field values directly on the associated object records. This ensures users always see accurate, up-to-date document metadata from list views and detail pages, making it easier to filter, sort, and export relevant information. These fields are system managed and cannot be edited by a user.
The following standard fields are available on Controlled Copy Trace, Periodic Review, Station Document, Document Change Request, and Change Authorization objects:
- Document Number (
document_number__v): A text formula field that looks up the standard Document Number field. - Document Title (
document_title__v): A text formula field that looks up the standard document Title field. - Owning Department (
owning_department__v): A formula field that looks up the standard Owning Department field.
These fields are available in all Vault environments, but must be added to the appropriate object page layouts to be made visible to users.
Station Manager
Support Document Title in Station ManagerConfiguration26R1.3
Station Manager now supports displaying the Document Title instead of the default Document Name in the Library View, Document View, and Document Information pages for both iOS and Android applications. Previously, the app only displayed the Document Name, which is often less descriptive. This made it difficult for operators to quickly identify the correct files.
With this update, Admins can choose which field best suits their operators’ needs by updating the newly renamed Station Manager Configurations component (formerly Station Document Metadata). Simply select either Document Name or Document Title within the component. If you do not explicitly select the title option, Station Manager continues to display the Document Name by default. Once configured, the Sync API automatically passes this value to connected mobile devices so that operators can locate the correct documents easily.
Training
Some Training features additionally apply to Study Training. See Clinical Operations: Study Training.
Optional Due Dates for Matrix TrainingAuto-on26R1.2
Training Administrators can now assign training to Learners that must be completed before they perform specific tasks, but without requiring a fixed due date. This feature allows you to create “at-your-own-pace” Training Assignments, providing greater flexibility in how you manage Learner requirements. This feature benefits Training Administrators by simplifying the creation of optional training, and Learners benefit from accessing necessary materials without the pressure of an immediate deadline.
Previously, Training Assignments required a due date, calculated from the Training Requirement’s duration values. This new enhancement allows for a “No Due Date” configuration, so that you can provide Learners with information that is required for compliance or Curriculum completion, but does not have a strict completion timeline.
- You can activate the No Due Date value in the Training Requirement Due Date Calculation picklist, corresponding to the Due Date Calculation field on Training Requirements.
- When Vault creates a Training Assignment for a “No Due Date” requirement, it sets the assignment’s Due Date Timing accordingly and leaves the assignment’s Due Date blank.
- Assignments without a due date are still included in Curriculum completion calculations, ensuring Learners fulfill all necessary training to complete their curriculum.
When a Learner is assigned a Curriculum that requires a duration-based due date for a requirement they already have as a “No Due Date” assignment, Vault automatically updates the blank due date to a specific date based on the new configuration.
Curriculum Due DatesConfiguration26R1.3
Training Administrators can now manage due date configurations for all non-retired requirements within a Curriculum. This feature allows you to define due date settings at the Curriculum level and automatically apply those configurations to all associated Training Requirements.
Previously, managing due dates required updating each requirement individually, which could lead to inconsistencies. This enhancement streamlines your administrative workflow so that you can ensure standardized due dates across multiple requirements quickly and accurately.
This feature includes new Curriculum object fields, including Action on Due Date Calculation Change, Due Date Calculation, Duration, Duration Unit, Duration: Initial, and Duration Unit: Initial. These fields are also available for Training Change Requests.
- Action on Due Date Calculation Change allows Admins to choose to copy settings to all associated requirements, or only update those where the new Curriculum duration is shorter than the requirement’s existing duration.
- Vault automatically applies these settings when you add a new Training Requirement to a Curriculum, or when you modify the Curriculum’s existing due date configuration.
Pre-Training QuizConfiguration26R1.3
Pre-Training Quizzes allow your organization to assess a Learner’s knowledge before they review an assignment’s Training Materials. This is beneficial for both Training Admins who want to measure training effectiveness, and Learners who may already be proficient in a subject.
Previously, assessments occurred only after training was complete. With Pre-Training Quizzes, you can offer a “test-out” option so that Learners can demonstrate existing expertise and bypass unnecessary content, or you can compare pre- and post-training performance to better understand how much your Learners are improving.
- You can configure a Training Requirement so that passing the Pre-Quiz automatically completes the entire Training Assignment, saving time for experienced Learners.
- When a Pre-Quiz is made optional, Learners have the choice to skip the assessment and proceed directly to the Training Materials.
- To reduce the burden on Learners, you can configure Vault to automatically exclude Pre-Quizzes for assignments it creates as the result of document revision.
- New Training Assignment object fields track Pre-Quiz results, completion dates, and whether a Learner chose to skip the assessment, providing a clear audit trail of Learner knowledge.
Completion of E-Learning Completes Training AssignmentAuto-on26R1.3
When a Learner completes an E-Learning (SCORM) course, Vault prompts them to populate their eSignature upon closing the SCORM player, then completes the assignment’s workflow task and marks the related Training Assignment as Complete.
In some instances, after completing the eLearning, a Learner may forget to click the Mark Complete button. This feature skips that step by immediately prompting the Learner for their eSignature.
Quiz Dynamic SectionsConfiguration26R1.3
Dynamic Sections within a quiz allow you to create a pool of questions from which Vault randomly selects a subset for each Learner attempt.
Previously, quizzes used the same set of questions for every attempt. This made it possible for Learners to memorize incorrect answers and eventually guess the correct ones over multiple attempts, compromising the quiz’s validity. While Training Admins can increase the total number of questions to counteract this, doing so often makes quizzes too long and burdensome for Learners. Dynamic Sections solve this by keeping the quiz length short while ensuring that every attempt provides a unique, randomized combination of questions from a larger pool.
You should apply this feature when you need to quiz a Learner on procedural understanding but want to ensure the assessment remains a valid measure of knowledge. This is especially useful for compliance-related training where preventing “answer sharing” or memorization is critical.
- Add Dynamic Section Designs to a Quiz to include a mix of static questions (which always appear) and dynamic questions (which are pulled randomly from a pool) so that you can control core content while varying the rest of the assessment.
- Define a required number of questions to be pulled from a larger group so that learners receive a concise, ten-question quiz even if you have a library of 50 potential questions.
- Vault now automatically calculates the Passing Percentage based on the specific questions served during an attempt, ensuring that Learners are graded fairly even if different questions have different point values.
- Vault calculates the estimated Section Design Points based on the average points of all questions in the dynamic pool so that Training Admins can effectively plan and balance their grading criteria.
Curriculum Completion Percentage UpdateAuto-on26R1.3
This feature updates how Vault calculates and manages Curriculum Completion Status to ensure that Learner progress is accurately tracked, even when training requirements or roles change. Vault now introduces a method to archive completion data when a training path is broken, preventing incorrect 100% completion scores and unintended curriculum outcomes.
Previously, when an administrator removed a Learner from a Learner Role or retired a Curriculum, Vault could inadvertently mark the Curriculum Completion Status as 100% complete if the remaining assignments were no longer applicable. This can trigger Curriculum Outcomes, such as granting system access or certifications that the Learner hadn’t actually earned.
With this update, Vault preserves the Learner’s actual progress in a new historical record instead of forcing a completion, so that your training compliance data remains trustworthy and accurate.
- Vault now automatically moves records to the Historical Curriculum Completion Status object when an assignment is cancelled, for example when a Learner is made ineligible or removed from a role, or a Training Admin removes requirements or roles from a Curriculum.
- The system now checks for active associations between Learners, roles, and curricula before calculating progress, so that it can archive data rather than recalculating an incorrect percentage.
- When a document is superseded, Vault “freezes” the completion status until the new training is assigned, preventing Learners from being marked complete during the brief window between document versions.
- When a previously-removed training path is restored (such as re-adding a Person to a Learner Role or updating a Learner’s training eligibility), Vault copies the data from the historical record back to the active record so that the Learner does not lose their previous progress.
Training & Study Training EnhancementsAuto-on26R1.3
Document Relationships Panel: TRIA Section
A new Training Requirement Impact Assessments section is added to the QualityDocs Document Relationships Panel, listing any Training Requirement Impact Assessment records for that document that are open (not Completed or Cancelled).
Quiz Iconography Update
We have updated the iconography for quizzes “display of missed questions” so that a Learner can distinguish between the following types of answers:
- Selected correct answers
- Selected incorrect answers
- Non-selected correct answers
Previously, Vault displayed answer types 2 and 3 with a red X. The following describes all answer behavior:
- When a Learner selects the correct answer, Vault displays:
- A green checkmark iconography
- A light green background around the entire answer
- A green border around the entire answer
- When a Learner selects the incorrect answer, Vault displays a red X icon.
- When a Learner does not select a correct answer, Vault displays:
- A green arrow icon
- A light gray background around the entire answer
- A dashed border in black around the entire answer
- When a Learner does not select an incorrect answer, Vault does not display any special styling. This behavior is unchanged from previous releases.
Assign Training Requirement to LearnerAuto-on26R1.3
You can now assign a Training Requirement directly to one or more Learners using the new Assign to Learner user action. This feature allows Training Administrators to quickly add specific requirements to a Learner’s training matrix, ensuring they receive necessary updates and recurrences without the need for complex, manual configuration of traditional curricula.
In previous releases, adding a single Training Requirement for a specific Learner often required broader matrix changes. Now, you can perform this action directly from a Training Requirement record so that you can respond faster to unique training needs while maintaining data integrity. Vault automatically handles the creation of the necessary Individual Curriculum and Individual Learner Role records behind the scenes, reducing the administrative burden of manual setup.
This is primarily a Training Administrator feature, providing a streamlined way to manage individualized training needs. Learners also benefit from this change, as their assignments stay up-to-date automatically whenever the underlying requirement is modified or its recurrence is triggered.
When you execute a requirement’s Assign to Learner user action, you can select up to 1,000 Learners and provide a reason for the assignment. Additionally, a new related object section for Individual Assignment Requests is available on Training Requirement page layouts to help track these assignments.
- You can assign a single Training Requirement to as many as 1,000 Learners at once through a single user action.
- When a Learner does not already have an Individual Learner Role or Individual Curriculum, Vault automatically creates them to support the new assignment. Otherwise, Vault prevents the creation of multiple identical records for the same Learner and displays an error message if this occurs.
- To ensure system stability, Vault blocks Admins from updating an Individual Curriculum record’s Learner field, and also prevents these records from moving to the Inactive or Retired lifecycle state.
Validation Management
Test Authoring Interface Experience ImprovementsAuto-on26R1.2
This release includes several improvements to the Test Authoring interface:
- Loading the authoring interface and saving steps are faster.
- Field validations (required and minimum and maximum length) are actively handled when interacting with steps, rather than producing an error when saving.
- The maximum number of prompts shown on the step horizontally is also increased to mirror the layout of steps on Execution and Review interfaces.
This feature is auto-on and visible to all users who interact with the Test Authoring interface.
Populate Validation Viewer Role on Validation Management RecordsAuto-on26R1.3
With this release, users assigned the Validation Viewer application role are now populated and visible in the Sharing Settings on Validation Management object records. This allows Vault Owners to provide more granular access through atomic security configuration on Validation Management object lifecycles. This feature is auto-on, but there is no impact unless configuration changes are applied.
Copy Enhancements for Activities & ProtocolsAdmin Checkbox26R1.3
With this release, we have improved the deep copy engine for Validation Activities and Test Protocols to performantly handle high-volume copies, doubling the previous capacity from 5,000 to 10,000 records.
Additionally, to make troubleshooting easier, deep copies are no longer an all-or-nothing transaction. They now stop exactly at the point of failure so Admins can see what Vault successfully copied. Created records that are part of a failed hierarchy are moved to the new Copy Failed state.
Finally, to prevent database bloat, Vault automatically sweeps any records in the Copy Failed state to a Pending Deletion state, where Validation Management then deletes them in the background. This feature is enabled through an Admin setting, and will be auto-enabled in 27R1.
LIMS
Apply Default Filter For Owning SiteAdmin Checkbox26R1.3
This feature introduces automatic, default site-based filtering across LIMS-controlled screens based on a user’s assigned organizational location. For example, when a user adds a Sample to a Test Set or selects an Asset, the list of records that appears is filtered based on the Owning Site and the user’s Organization.
Retains TestingConfiguration26R1.3
This feature adds the ability to manage retains testing in Veeva LIMS, including:
- Defining Retain Samples, including any scheduled Tests, in a Batch Release Spec Data
- Defining and conducting retains testing according to a schedule until Sample disposal
- Tracking Sample quantity throughout the retains testing process
Stability Study BuilderConfiguration26R1.3
The Stability Study Builder introduces a purpose-built, top-down user experience for authoring, editing, and managing Stability Studies and Study Designs. This feature automatically creates Spec Data and Sample Plans as Timepoints are added. Users can edit a Timepoint’s Test assignment from Stability Builder.
Reduced TestingConfiguration26R1.3
This feature introduces Reduced Testing capabilities within Veeva LIMS. It allows organizations to apply a reduced set of Tests and Criteria to batches of material received from qualified suppliers, rather than performing full testing on every incoming lot.
The system automates the selection of the appropriate testing plan (Full, Reduced, or others) based on pre-defined schedules, supplier qualification statuses, and the history of processed batches. This helps laboratories optimize resource utilization, reduce testing cycle times, and focus analytical efforts on higher-risk materials while maintaining strict regulatory compliance and product quality.
Non-Routine & Ad Hoc Sample TestingConfiguration26R1.3
This feature introduces Non-Routine and Ad Hoc Sample Testing to Veeva LIMS, enabling analysts to perform testing on Samples outside the scope of Batch Release and Stability Studies. Non-Routine Spec Data can be created for typical business processes such as cleaning or method validation. Analysts can also define Ad Hoc testing for more freeform supplementary testing. Non-Routine and Ad Hoc Testing can be initiated and optionally reviewed by creating a Request record.
Calculated Result Can Use Input AttributesConfiguration26R1.3
This feature allows a Test Definition to use an Input’s attribute value in a Result’s calculation.
Spec Execution: Initiated Date & TimeAuto-on26R1.3
With this update, we have added the Initiated datetime field to Spec Execution records to track the date and time they were initiated.
Calculated Result Can Use Input AmountConfiguration26R1.3
This feature allows a Test Definition to use an Input’s Amount value in a Result’s calculation.
Reduced Testing: Obsolete Field & Object InactivationAuto-on26R1.3
This release inactivates obsolete fields and objects replaced by the new Reduced Testing feature. This includes the Reduced Testing Frequency, Reduced Testing Interval, Include in Reduced Testing, and Full Testing fields, as well as the Material Supplier object.
New fields will be added to support the Reduced Testing feature introduced in 26R2.
Test Definition & Spec Data: Updated Formatting Options Validation MethodAuto-on26R1.3
The following valid states for formatting options on a Result or Criteria have been added to triggers and Change Analysis checks:
- Rounding Rule, Precision, Precision Type, & Precision Rule all have data
- Rounding Rule & Precision Rule have data
- Precision & Precision Type can be blank or both populated
Print Label: Label Definition is SearchableAuto-on26R1.3
Users can now further filter the list of Label Definitions on Print Label actions. Previously, the field was a dropdown with no search capabilities.
Stability Study: Date/Time Fields for Pull WindowAuto-on26R1.3
The following Date/Time fields are now available on the Study Timepoint and Lab Sample objects:
- Earliest Pull Date/Time
- Latest Pull Date/Time
- Scheduled Start Date/Time (Study Timepoint object only)
Additionally, the Hours and Minutes values have been added to the Unit picklist.
Stability Study: Create Copy of Sample Plan & Spec DataAuto-on26R1.3
The Generate Lab Study and Generate Lab Study Timepoint actions now have a new option: Create Copy of Sample Plan and Spec Data. When selected, a new copy of the Sample Plan and Spec Data is used for each Timepoint.
Additional LOD Picklist ValueAuto-on26R1.3
The No More Than picklist value is now available as an entry option and formatted value for the LOD Entry Option picklist.
Design Data Builder: Chart DefinitionsAuto-on26R1.2
Spec Data Builder now allows Design Data Admins to configure various chart types, including control charts and regression analysis, directly through a centralized interface. Previously, this option was not available in the Design Data Builder.
Lab Test & Sample Field UpdatesAuto-on26R1.2
This update introduces the following data model changes:
- The following standard fields have been added to the Spec Data Sample Action object: Externally Tested?, Testing Location, Auto-Dispose?, Planned Disposal Interval, Reported Name
- The Lab Consumable Family object now has a lifecycle
- The Minimum, Maximum, and Decimal fields are now editable on Lab Test Definition Input, Lab Test Definition Variation Input, and Lab Test Input objects
Lab Consumable Family Object LifecycleAuto-on26R1.2
This feature adds a standard lifecycle to the Lab Consumable Family object.
Spec Data Builder: Option to Edit Fields on Sample ActionAuto-on26R1.2
For Select Actions (like Aliquot or Test Actions), there is now an Edit Metadata button in the UI. This opens the Action record in a new browser tab (VOF), allowing the user to quickly set metadata field values without cluttering the main Builder interface. Additionally, the Reported Name field is now directly visible and editable on the Test Action card within the Spec Data Builder.
Test Definition Result: Formatting Options Can Be OptionalAuto-on26R1.2
Previously, LIMS enforced Precision Type and Precision Number as required fields via standard validation rules. This feature allows Admins to inactivate these rules, and thus save a Result or Variation Result without applying formatting rules.
Spec Data Builder: Formatting Options Now EditableAuto-on26R1.2
In the Spec Data Builder, the Result Format section on the Criteria card is now interactive, allowing Admins to view and update the formatting options directly.
This feature loosens the rigidity around formatting fields. While Notation cannot be blank (it will default to Standard if unspecified), Admins may now leave the other fields blank. Admins can also use specific valid combinations, such as populating just the Rounding Rule and Precision Rule, or populating all fields.
Upper Level of Quantification (ULOQ)Auto-on26R1.2
This feature expands on LOD/LOQ with an Upper Level of Quantification (ULOQ). While LOD/LOQ cover the smaller range of possible measurement values, ULOQ provides the means to change how measurements above acceptable ranges are reported, displayed, and calculated. Design Data Builder has been updated to support the individual use of LOD, LOQ, and now ULOQ for Lab Test Result Definitions.
Test Execution: Display Spec CriteriaConfiguration26R1.2
This feature allows Design Data Admins to display detailed product specification descriptions to Lab Analysts directly within the Test Execution and Sample Results Entry (SRE) UI.
Test Execution: Updated Exception Icon Hover DisplayAuto-on26R1.2
Because some spec descriptions can be very long, the existing Criteria icon hover tooltip has been redesigned to prevent text from clipping off the screen. The new tooltip shows a maximum of three Criteria, truncates descriptions on the third line, and includes a new View Details link that opens a dialog containing the full, untruncated text for all applicable Criteria.
Test Execution: Optional Expected AmountAuto-on26R1.2
For some cases, Analysts do not have to enter an Expected Amount for Consumables. In this case, the Expected Amount can be left blank in the Test Definition and thus is not required in LIMS.
Assets & Consumables StandardizationAuto-on26R1.2
This feature standardizes commonly used components (objects, object types, lifecycles, lifecycle states, workflows, and fields) across Assets and Consumables. No new logic is introduced as part of this update. Establishing these standards provides a foundation for building future functionality in the Assets and Consumables domain.
Regulatory
RIM Publishing
Automatic On-Demand Validation for Submission Validation Version ChangesAuto-on26R1.2
When Publishing users select a new Validation Criteria Version field value, the system will now automatically trigger on-demand validation. Validation will be triggered upon saving the new value on the Submission record, and only when Enable Continuous Publishing is set to Yes.
What’s New
Previously, publishers would need to remember to revalidate in-progress submissions if the previously selected validation criteria was updated. Now, the system will automatically trigger the action.
Key Benefits
When validation criteria is mistakenly chosen, or when new validation criteria becomes required while submissions are in-progress, the automatic validation trigger allows for the most current validation results to be made available.
Taiwan eCTD 3.2 (TW 2.1) DTD 2.0Configuration26R1.2
Submission Publishing now supports the TFDA eCTD v2.1 specification for publishing and validation of Taiwanese submissions.
Key Benefits
The updates will ensure that customers publish and validate compliant Taiwanese eCTD v2.1 submissions to TFDA
Additional Considerations
The TW eCTD v2.1 format will be required for new Marketing Authorizations from 01 July 2026.
Auto-Append STFAuto-on26R1.3
The system will automatically set the XML Leaf Operation to the status of Append for any Study Tagging File (STF) that has been previously submitted as New in the same eCTD v3.2.2 application.
What’s New
For any Submission Content Plan (SCP) containing an active Content Plan Item (CPI) with Node Type = STF-Leaf, system logic will now determine if the same STF has already been submitted in a previous eCTD sequence in the same application. The system will search previously Published and Imported sequences within the same application for the Study ID (clinical_study__v or nonclinical_study__v).
When the STF CPI reaches Baselined state, the system will search the previous sequences and:
- Use the Submission Metadata object to determine if the current STF Study ID value is exactly identical to a previously submitted Study ID
- Confirm the Submission containing the previously submitted STF is:
- in a status of Publishing Inactive and populated with an Actual Submission Date value, or
- has Dossier Status = IMPORT SUCCESSFUL
- Automatically update the XML Operation of the most recently submitted STF to Append
- Calculate and update the XML Modified File Path
Key Benefits
Publishing end users will no longer need to manually use the Set Leaf Operation action for STF Content Plan Items (CPIs). This will automate the manual and repetitive effort that was previously required for every ongoing submission to support a nonclinical or clinical study in Module 4 or 5. This feature will also reduce the risk of users forgetting to change the STF leaf operation to Append, or mistakenly selecting the wrong STF file to lifecycle for a particular study.
Additional Considerations
- The Auto Append system action does not apply to any eCTD 4.0 submissions, and only applies to CPIs in Module 4 or Module 5 where Node Type = STF-Leaf.
- STF CPIs will still be created as New. The Append operation update will only be completed upon the CPI reaching Baselined state.
- Only the STF CPI will be automatically updated. Users will still need to use Set Leaf Operation to lifecycle any files that are submitted under the STF.
- The previous submission need not have been published using Veeva Submissions Publishing. STFs previously included in Imported submissions will also be included in the automatic updates.
- If the Study ID is not found in a previous sequence (new study), or any differences are detected in the Study ID value, the STF CPI will remain New.
Set Reference Leaf Updates for eCTD 4.0 SupportAuto-on26R1.3
An update to the Set Reference Leaf user interface (UI) allows Publishing users to reference eCTD v3.2 submission documents while working in an eCTD 4.0 submission. The user interface allows for eCTD 4.0 functionality to blend into the existing and familiar UI used for eCTD 3.2 submissions.
What’s New
This feature introduces forward compatibility with eCTD 4.0 on the backend (including compatibility with new Submissions Archive APIs) without disrupting the current process flow for setting reference leafs. Both eCTD 3.2 and 4.0 submissions will have the option to inherit the existing CPI title upon saving.
Key Benefits
Forward compatibility for eCTD 4.0 allows for maximum document reuse, even if the target of a Reference Leaf was originally submitted in an eCTD 3.2 application. All existing Set Reference Leaf actions and process steps remain unchanged, reducing any retraining burden on end users who have adopted eCTD 4.0.
Publishing Validation Results Help FieldsAuto-on26R1.3
Two new standard fields have been added to the standard Publishing Validation Criteria (publishing_validation_criteria__v) object. These fields are intended to provide end users with helpful tips or instructions to resolve open Validation Results. The fields will be populated by an Admin on the Publishing Validation Criteria object, and will also be available as a Lookup field on the Submission Validation Result object (submission_validation_result__v) for easy access at the time of troubleshooting.
What’s New
- Veeva Help Text (
veeva_help_text__v): Veeva-provided suggestions and notes will be made available via VPK at a later date - Additional Instructions (
additional_instructions__v): Rich text field available for customers to populate with internal SOPs, specific points of contact, or any other desired information
Key Benefits
These new fields are being added to provide Publishing end users and Admins convenient access to find troubleshooting tips, links internal processes and publishing standards, notes for local markets & affiliates, or other helpful information to support resolving validation failures and warnings. New fields are being introduced to the data model to preserve the integrity of existing resolution suggestions provided by some Health Authorities, but still allowing customers the flexibility and convenience of information at-hand.
Veeva Help Text will provide suggestions for common fixes related to Publishing tool usage, such as a reminder to use a unique Published Output Location. Additional Instructions is available for customers to add their own process details and preferences, such as a note to ignore a particular low- or medium-level validation warning.
Other Considerations
- Both the Veeva Help Text and Additional Instructions fields will be editable by an Admin, and can be populated via VPK or Loader. Admins will also need to add the fields to Layouts or any in-use Reports for them to be visible to end users.
- Upon release, the Veeva Help Text will not be populated, but values will be made available via the Veeva Product Support Portal at a future date. Customer values added to the Veeva Help Text field will be overwritten when the VPK is deployed.
RIM Submissions
Copy into Global Content PlansAuto-on26R1.3
Global Content Plan (GCP) users now only need to manage Document Sets at the CPI level. Instead, users will only assign Document Sets at the Content Plan Item (CPI) level.
What’s New
The new Document Set object data model no longer requires users to assign Document Sets at the Content Plan (CP) object level. GCP dispatch logic has been updated to automatically identify & exclude empty Content Plan sections from the dispatch, and the system will now manage the valid combinations of Document Set Assignments.
Key Benefits
This feature eliminates the manual effort required to assign Document Sets in multiple places (CP sections + CPIs). Now users can assign Document Sets at the level they are needed, at the CPI level where they would be matched. Vault will also calculate when higher level CP sections are in-scope vs. out-of-scope for a particular local market Submission, based on the lower-level CPI assignments. Empty CP sections are not dispatched to a target SCP when there are no CPIs in scope, indicating there are no expected documents.
Other Considerations
- With this feature, the Document Set Content Plan relationship is deprecated and all existing records are deleted.
- This feature will be automatically enabled in any Vault with the Improved Document Set Management Admin setting already enabled. For customers who have not yet enabled Improved Document Set Management, this feature will be automatically included when that setting becomes enabled.
- The new functionality only applies to Dispatch Comparisons completed after the release, and does not apply retrospectively. To use the new logic on existing dispatches, use the Regenerate Comparison action.
RIM Submissions Archive, RIM Submissions Publishing
Empty Section Indicator Scalability ImprovementAuto-on26R1.3
This feature utilizes a new system-managed object, Application Metadata, to facilitate the processing of the Empty Section Indicator (ESI) within Submissions Archive Viewer. The underlying functional behavior remains unchanged: If your Vault has the Application Setting: Enable identifying empty sections in the Viewer enabled when viewing submission content in the Viewer, any section in the Name column that does not contain documents displays the Empty Section icon so users know it contains no content.
As Submissions Archive contains more and more data each day, we are implementing this feature to change how we access data behind the scenes. This drives future scalability and enables future features in Submissions Archive.
The changes driving this feature include:
- New Metadata Object: The scalability update utilizes a new system-managed object called Application Metadata (
application_metadata__v) - Shifted Job Triggers: The trigger for the ESI Job has shifted from the Legacy Document Field
esi_update_pending__vto the ESI Last Modified Date field on the new Application Metadata object. - Automated Datetime Tracking: The ESI Last Modified Date field captures a DateTime value automatically following document Import or Removal actions, as well as Publishing-in-Vault events.
- Active Status Maintenance: The Empty Section Indicator is maintained as long as your Application is in an Active status.
Key Benefits
- Enhanced Oversight: Users save time by identifying empty sections in the Application they’re viewing at a glance in the hierarchy, without needing to expand the deepest parts of the structure.
Additional Considerations
- Enablement: This feature is Auto-On. However, there are no changes to the enablement of the underlying Application Setting Enable identifying empty sections in the Viewer, which remains a one-way setting that cannot be disabled once turned on.
- Legacy Field Deprecation: Following the 26R3 release, Submissions Archive will no longer maintain the legacy document field
esi_update_pending__v. - View Restrictions: The Empty Section Indicator only displays when using the Application View; it will not appear when one or more Submissions are actively applied in the filter.
Update Active Dossier from Country Decision DetailConfiguration26R1.3
What’s New?
A new system action is available for configuration in the Country Decision Detail (CDD) lifecycle to automatically update Active Dossier. When triggered by a lifecycle state change, such as entry into the Approved, Rejected, On Hold, or Withdrawn states, Vault updates related Active Dossier records to the appropriate status (such as Pending Current, Pending Superseded, Rejected, Withdrawn, No Decision, or Replaced) via the Active Dossier Calculation.
This release also extends the Active Dossier Processing Status and Issues Tracking feature, released in 26R1 to support CDDs:
- Extended UI Placement: You can now add the Active Dossier Processing Status control, including status icons and the hovercard, to the Country Decision Detail layout.
- Targeted Visibility: The CDD hovercard exclusively shows processes filtered for that specific record. These processes are excluded from the parent Regulatory Objective hovercard.
- Extended Object Tracking: The Active Dossier Processing and Processing Issue objects now include a Country Decision Detail field to link tracking logs to the triggering CDD.
- New Filter Value: The Active Dossier Processing Type picklist now includes the Update from Country Decision Detail value, allowing you to filter the Processing Issues page for CDD-triggered jobs.
Key Benefits
This enhancement supports workflows in multi-country procedures, such as the EU MRP or DCP, where final verdicts are captured at the Country Decision Detail level.
- Replacing Workarounds: You can now configure Active Dossier updates based on the CDD lifecycle state rather than relying on the Regulatory Objective state.
- Automated Date Syncing: Vault populates the ADID Approval Date or End Date based on the CDD Country Decision Date field when the decision status is Current, Rejected, or Withdrawn.
Additional Considerations
- Configuration Required: This feature requires configuration by an Admin.
- Handling Inactive Countries: When the automated update triggers, Vault checks the status of the associated country. If the
country__vrecord is inactive, the system skips it and excludes it from the calculation. - Global Data Model Impact on Processing Validations: The Validation Description field (
validation_description__v) on the Active Dossier Processing Validation object used for the Active Dossier Processing Status and Issues Tracking feature is being updated from an editable field to a locked, non-editable field to ensure consistency across all Vaults.
RIM Publishing, RIM Registrations, RIM Submissions, RIM Submissions Archive
HAQ Response Automations: Default Fields on Response DocumentAuto-on26R1.3
What’s New?
In this release, Vault enhances the Initiate Response user action to automate more of your data entry. When you create a response document through the guided user flow, Vault now populates additional fields based on the selected Health Authority Response (HAR) record.
Beyond the standard fields already populated—such as Health Authority Response, Health Authority Question, Application, Regulatory Objective, Submission, and Product Family—Vault now maps any fields where the name and data type match between the HAR record and the response document’s classification.
Field Mapping Logic
To ensure accuracy when mapping custom or standard fields, Vault applies the following rules:
- Name Matching: Vault matches field names regardless of the namespace (for example,
__cor__v). - Data Type: The data type must be identical between the HAR record field and the document field.
- Active Fields: If multiple fields share the same name, Vault only uses the active field.
- Namespace Priority: If multiple matching fields exist with different namespaces, Vault prioritizes them in this order:
__v__rim__vs__sys__c
Key Benefits
- Increased Efficiency: Reduces manual data entry by automatically pulling relevant information from the HAR record into the response document.
- Improved Data Integrity: Ensures consistency between your Health Authority records and the resulting documentation by utilizing automated mapping.
- Flexible Configuration: Allows organizations to map custom fields simply by aligning field names and data types.
Additional Considerations
- This feature is auto-on for all RIM Vaults.
- If you want to map custom fields, ensure the field names on both the HAR object and the document classification match exactly.
- Vault continues to adhere to existing limits, such as the 250-record limit for Health Authority Question records.
- If you need help configuring matching fields, contact your Veeva Representative.
RIM Registrations, RIM Submissions, RIM Submissions Archive
Active Dossier: Enhanced Metadata ExportAuto-on26R1.3
This release enhances the existing Export Excel Tree with All Descendants action.
What’s New?
- Filter Adherence: The export now respects filters applied in the Active Dossier Viewer. All in-scope records are extracted, regardless of whether they appear in the viewer due to row limitations.
- Expanded Field Support: The exported Excel file supports the following new columns if these fields are included in the Active Dossier Item Detail Layout: ADID Registrations, ADID Product Variant, ADID Manufacturer, ADID Inactive Ingredient. Values for these multi-select fields appear as comma-separated text and can hold up to the Excel cell limit of 32,767 characters.
- Direct Vault Links: Supported fields now include clickable links, allowing you to open specific document versions or object detail pages directly from the spreadsheet.
- Formatting Updates: Datetime fields in the export reflect your local time zone.
- Security Context: The system performs the export under your specific access context. Vault only extracts fields you have permission to view.
Key Benefits
- Targeted Information Sharing: Effortlessly extract and share specific sections of a dossier with stakeholders by using filters.
- Support for Large Dossiers: Successfully export records from dossiers exceeding one million records by filtering data to remain under Excel limits.
- Seamless Navigation: Eliminate manual searching by navigating directly from Excel to Vault records.
Additional Considerations
- Permissions Required: This feature is automatically enabled, but you must have Read permissions for the following fields:
- Active Dossier Structure:
display_name__v,hierarchy_path__v, andparent__v - Active Dossier Item:
section__v,hierarchy_path__v, anddocument__v - Active Dossier Item Detail:
section__v,hierarchy_path__v,application__v,ad_status__v,document__v, anduniqueness_key__v
- Active Dossier Structure:
- File Delivery: This action runs in the background. Once complete, Vault sends a notification and an email with a download link. The link remains active for 7 days, and the file is downloaded as
Active_Dossier_as_of_{datetime}.xlsx. - Built-In Limits: To prevent file corruption, the export fails if it exceeds Excel’s limit of 1,048,576 rows. If you receive an “Active Dossier Export Failed” notification, narrow your filters and try again.
Extracting Proposed Active Dossier Relationships for RegistrationsAuto-on26R1.3
What’s New?
You can now execute the Extract Active Dossier Records bulk action on the Application object without enabling the Active Dossier Registrations Tracking application setting. This update allows you to run the action even when the broader registration tracking functionality is turned off.
To execute this action, the following must be true:
- You have permissions to view and execute the record action on the Application object.
- The Active Dossier Item Detail (ADID) records exist in the Vault.
- The Vault has Registrations licensed.
- Admins have configured at least one state type in the Valid Registration State Types field.
Key Benefits
This enablement update provides greater flexibility for managing your Active Dossier (AD) data. Key benefits include:
- Controlled Testing: You can extract and analyze proposed records to test registration matching logic without triggering the unintended creation of ADID Registrations, ADID Product Variants, ADID Inactive Ingredients, or ADID Manufacturers.
- Migration Readiness: Customers can complete data migrations for existing ADIDs before enabling the Extract Proposed Active Dossier Relationships for Registrations & Product
- Information Tracking feature for all users.
- Process Integrity: You can analyze output without affecting day-to-day AD population processes.
Additional Considerations
- Admin Setup: To set the required state types, an Admin must briefly enable the Active Dossier Registrations Tracking setting, set the Valid Registration State Types field, click Save, and then disable the setting by clicking Edit and Save again once the setting is unchecked. Changes to the Valid Registration State Types field are only saved when the Active Dossier Registrations Tracking setting is saved.
- Record Creation: While the Active Dossier Registrations Tracking application setting is off, Vault does not create ADID Registrations, ADID Product Variants, ADID Inactive Ingredients, or ADID Manufacturers during population.
- Support: If you need assistance with licensing or configuration, contact your Veeva representative.
RIM Registrations, RIM Submissions
Active Dossier Population for Activities with No SubmissionAuto-on26R1.3
What’s New?
This enhancement automates Active Dossier (AD) record generation for markets that allow immediate change implementation without formal regulatory submissions. During the Global Content Plan (GCP) dispatch, Vault now automatically generates AD records without creating a Submission Content Plan based on two primary use cases:
- Immediate Implementation without additional activity: For changes that do not require a regulatory submission at all. You can now generate Active Dossier Item Detail (ADID) records regardless of whether the Related Submission field in the Activity points to an active or inactive Submission, or is blank.
- Submission after Implementation:For changes implemented first and filed later. To trigger automated AD generation without a Submission Content Plan, the Related Submission must be either blank or contain an inactive Submission. (If the Related Submission points to an active Submission, Vault performs a standard dispatch to create a Submission Content Plan).
For these dispatches, the target Activity must have a populated Application and an active Regulatory Objective. Vault uses these relationships to filter documents accurately, bypassing the creation or comparison of a Submission Content Plan. To use this feature, you must select specific Activities via the updated GCP dispatch dialogue. For more details, see Active Dossier Population for Activities with No Submission: Dispatch Dialogue Updates.
Key Benefits
- Automated Updates: Eliminates manual AD population for non-submission changes.
- Simplified Workarounds: Removes the need for “dummy” Submissions or manual additions via the Active Dossier Editor.
- Accurate Tracking: Provides a real-time view of product supply status for all markets.
Additional Considerations
- Disposition Setup: In alignment with existing logic, ADID statuses are determined by how your Local Dispositions are mapped to the Populate Active Dossier categories on the Dispatch Global Content Plan user action. Vault continues to support both standard and custom local dispositions.
- Immediate Implementation without additional activity:
- Mapping: Dispositions mapped to the “immediate implementation without additional activity” category (e.g., the standard No Regulatory Action Required to Implement).
- Result: Generates ADID records in a Pending Current status. The Needs Submission field is automatically set to No. The calculation logic supersedes ADIDs for previous document versions based on the relationships on the Activity’s Related Regulatory Objective.
- Submission after Implementation:
- Mapping: Dispositions mapped to the “requiring submission after implementation” category (e.g., the standard Implement Then File).
- Result: Generates ADID records in a Pending Current status. The Needs Submission field is automatically set to Yes. The calculation logic supersedes ADIDs for previous document versions based on the relationships on the Activity’s Related Regulatory Objective.
- Document Filtering Logic: In alignment with already existing GCP dispatch functionality, document filtering depends on your User Action configuration:
- Copy Relationships Off: Vault filters documents by matching the target Regulatory Objective relationships (Product, Active Substance, etc.) against the global Event.
- Copy Relationships On: Vault bypasses the comparison and includes ADIDs for all dispatched documents in the scope for the target Activity.
- Auto-On: This feature is automatically enabled for all Vaults using Active Dossier population within the GCP dispatch if the Enable Improved Document Set Management checkbox is on in your environment. To utilize this feature, the Enable Improved Document Set Management checkbox must be on.
Active Dossier Population for Activities with No Submission: Dispatch Dialogue UpdatesAuto-on26R1.3
What’s New?
To support the new automated Active Dossier (AD) population logic (see Active Dossier Population for Activities with No Submission), the Global Content Plan (GCP) dispatch dialogue now provides specific controls for Activities that do not require a Submission Content Plan. You can explicitly select these Activities to ensure Vault generates their Active Dossier records.
You can see the following checkbox options in the dispatch dialogue:
- Dispatch to Submissions and populate Active Dossier: Set this checkbox to dispatch the GCP to target Activities’ Submissions for AD population and Submission Content Plan creation.
- Populate Active Dossier for Activities that do not require a submission: Set this checkbox to enable a required drop-down menu where you can select specific Activities for AD record creation only.
The new Activity drop-down menu dynamically filters to show only eligible records that meet specific criteria:
- Immediate Implementation: To appear in this menu, the Activity must not be eligible for a standard GCP dispatch which results in submission content plan creation or updates. Because standard dispatches are hardcoded to always skip the No Regulatory Action Required to Implement disposition, these Activities will always appear as eligible, regardless of their Related Submission status. For other dispositions like Implement then File, they will only appear if the Related Submission is blank or points to an inactive Submission.
- Relationship Requirements: The Activity must have an active Related Regulatory Objective and Related Application.
- Security & Validation: You can only see Activity records you have permission to read. If an Admin sets the Enable Dispatch Validation checkbox to on, only Activities in supported lifecycle states appear.
Key Benefits
- Granular Control: Choose exactly which non-submission activities should receive Active Dossier updates. The updated dialogue is only presented if the Event includes an eligible Activity (see criteria above). If all Activities include an active Related Submission, then the original dialogue is displayed.
- Process Clarity: Distinguishes between a dispatch to a Submission Content Plan and a dispatch used solely for Active Dossier generation.
Additional Considerations
- Custom Disposition Visibility: If your Vault uses custom local dispositions, Activities will still dynamically appear in the new drop-down menu according to your mapping to the appropriate Populate Active Dossier category (e.g.,”immediate implementation without additional activity” or “requiring submission after implementation”).
- Auto-On: This feature is automatically enabled for all Vaults using Active Dossier population within the GCP dispatch if the Enable Improved Document Set Management checkbox is on in your environment.
RIM Publishing, RIM Submissions Archive
eCTD 4.0 Viewing: Stylized Submission Unit XMLAuto-on26R1.2
Regional details for eCTD 4.0 submissions are now presented in a user-friendly rendition that displays metadata and details that are not included in the Submissions Archive Viewer grid matrix. The stylized Submission Unit XML will be viewable for all currently supported eCTD 4.0 regional DTD versions, though each supported region will include its own set of information in the display.
What’s New?
Submission Administrative Information (SAI) for eCTD 4.0 submissions now displays in a stylized rendition, replacing the machine-readable plain text Viewable Rendition available previously. The new stylized rendition will be available via the Submission Administrative Information section of the Submission record layout, and both the Embedded Document Viewer and Mini-Browser within the Submissions Archive Viewer.
Consideration: For final, imported or published submissionunit.xml files that display with a plain text rendition, open the document in the Mini-Browser, then use the Re-render Document action. This will re-render the submissionunit.xml plain text rendition, replacing it with the stylized rendition.
Key Benefits
The stylized Submission Unit XML rendition provides a seamless viewing experience and aligns the SAI display with that of eCTD 3.2 submissions. The stylized rendition will be available for both imported submissions and those generated by Veeva Submissions Publishing.
RIM Registrations
IDMP Output Record PurgeAuto-on26R1.2
To maintain database hygiene and system performance, Vault now includes an automated cleanup process for Identification of Medicinal Products (IDMP) output records. For a fully built-out Medicinal Product, Vault can generate up to 1.6 million records across 45 objects. This feature ensures these redundant records are purged once the authoritative FHIR message is secured.
What’s New?
Vault manages record cleanup through a two-tiered process:
- Immediate purge: Vault purges all related Product Data Submission (PDS) output records immediately after a FHIR message is successfully generated.
- Daily maintenance (90-Day backstop): A daily IDMP Output Cleanup job runs to catch any old records or records missed during immediate purges.
- Logic: The job purges records if the related PDS Medicinal Product Element has not been modified in 90+ days or when the Marked for Deletion (
marked_for_deletion__v) field is Yes. - Failure handling: If an immediate cleanup fails, Vault flags the record by setting the Marked for Deletion field to Yes.
- Logic: The job purges records if the related PDS Medicinal Product Element has not been modified in 90+ days or when the Marked for Deletion (
- Failure notifications: When a cleanup job fails, Vault notifies the Vault Owner and includes a link to Admin > Operations > Job Status > History in the notification.
Key Benefits
- Performance Stability: Prevents high-volume IDMP data from impacting system responsiveness over time.
- Authoritative Records: Establishes the structured FHIR message as the single source of truth for submission history.
- Troubleshooting Safety: When the FHIR Message Generation job is not successful, IDMP output records are not immediately purged. Instead, they are preserved under the Product Data Submission for manual review and are only removed by the automated daily cleanup job once their related PDS Medicinal Product Element record has not been modified in over 90 days.
Additional Considerations
- Availability: This feature is provisioned to all RIM Registrations Vaults. The new Marked for Deletion field is provisioned as inactive. This field should be activated to support the purge operation, but it does not need to be added to the Product Data Submission object page layout. The daily IDMP Output Cleanup job runs daily at 12:00 AM EST by default, however you can modify the schedule.
- Operational guardrails: To prevent data conflicts, Vault blocks new IDMP data generation while a cleanup job is in progress (applies to both individual and bulk jobs).
Link Packaging to MP-Registration Bulk ActionAuto-on26R1.2
What’s New?
This feature introduces the ability to execute Link Packaging to a Medicinal Product Registration (MP-Registration) as a bulk action, allowing you to enrich MP-Registration records at the packaging level at scale. This update supports the EMA’s requirement for both XEVMPD and IDMP data to be submitted at the packaging level. When you execute this as a bulk action, the system processes records sequentially in batches of 500 to prevent race conditions.
How to Execute Link Packaging to MP-Registration as a Bulk Action
- Navigate to the Registered Packaging object.
- Select the desired records for enrichment.
- From the All Actions menu, select Link Packaging to MP-Registration.
- Review the confirmation dialog and click Finish.
- Vault sends an email notification with the results once the process is complete.
Key Benefits
- Scale and efficiency: Facilitates the enrichment of data in RIM Vaults where XEVMPD or IDMP data is already managed.
- Automated notifications: Vault sends a notification to the user who executed the bulk action upon completion of each batch.
- Detailed reporting: Notifications include summary results and CSV files for both successes and failures.
- Regulatory compliance: Helps maintain the granularity required for EMA structured data reporting.
Additional Considerations
- Sequential processing: If you include more than 500 records in a bulk action, Vault runs the jobs one at a time until all batches are complete.
- Success criteria: A record is categorized as a success if the job runs successfully, even if it results in no changes to the Medicinal Product Registration records.
- Failure logs: For any records that fail to process, Vault includes the specific error details in the results CSV.
- Permissions: Ensure your security profile grants the necessary permissions to execute bulk actions on the Registered Packaging object.
Enhanced IDMP Support for UDI-DIAuto-on26R1.2
IDMP requires the Unique Device Identifier (UDI-DI) at the medical device model level recorded in EUDAMED for drug-device combinations. This feature improves how you manage and report medical device identifiers within the IDMP data standard for these products.
Vault now allows for more than one of the same devices to be included in packaging by shifting data storage from the Product record to specific Packaging Product Variant records.
What’s New
- New Device Identifier Fields: Vault provisions new fields to manage identifiers at the individual level:
device_identifier__v: A text field on the Packaging Product Variant(packaging_product_detail__rim) object.device_identifier2__v: A destination field on the PDS Container Reference Item(idmp_container_reference_item__v) object.
- Updated Aggregation: The IDMP aggregation algorithm now sources the
device_identifier2__vfield from thedevice_identifier__vfield. - Fields Replacement:
- The new
device_identifier2__vfield replaces thedevice_identifier1__vData Carrier reference on the PDS Container Reference Item. - The IDMP data aggregation algorithm no longer sources the UDI-DI from the
basic_udidi__vfield on the Product record, but instead pulls it from the new text field on the Packaging Product Variant.
- The new
- Repeating Records: You can now enter multiple devices with the same Product and Product Variant combination as repeating Packaging Product Variant records with unique identifiers.
Key Benefits
- Improved granularity: Shifting data storage from the general Product level to specific Packaging Product Variant records allows Vault to accurately track multiple identical devices in a single package.
- Standard alignment: These updates better support the device identifier granularity required by the IDMP data standard and EUDAMED.
Additional Considerations
- Specific data requirements:
- To include devices in the IDMP output, you must create the device as both a Packaging Product Variant record and a Registered Product related to the drug Registration.
- In both records (Packaging Product Variant and Registered Product), you must set the Co-Packaged or Integral checkbox to Yes.
- Data Migration: Migration is not in scope due to the limited number of Data Carrier records where the Data Source is EUDAMED.
Important: Aggregation behavior changes automatically in existing Vaults configured for IDMP. The new device_identifier__v and device_identifier2__v fields are provisioned as inactive, so Admins must enable them.
Support for User Groups in Registration Verification WorkflowConfiguration26R1.2
This feature enhances Registration Verification by supporting user groups and manual participant selection within the workflow.
What’s New?
The Registration Verification workflow now supports the Allow workflow task owners to select participants and Use Vault user group as participants settings. These enhancements provide greater flexibility for the four-eye principle review process.
Key updates include:
- Participant selection: When the Allow workflow task owners to select participants setting is enabled on the workflow and the Select Verification Groups Admin setting is configured, Vault ignores the Select Verification Groups setting.
- User group support: When the Use Vault user group as participants setting is enabled, Vault ignores the Select Verification Groups Admin setting.
- Workflow visibility: The Verification Workflow Details section now displays the participants selected for the initial workflow task.
Key Benefits
- Enhanced Flexibility: Leverage standard Vault user group configurations for more granular control over registration change reviews.
- Process Efficiency: Dynamically identify correct reviewers during the process by allowing workflow initiators or task owners to select participants.
- Transparency: View assigned participants directly within the Regulatory Objective Details.
Additional Considerations
- Initial task limitations: The Allow workflow task owners to select participants setting does not apply to the initial task in a workflow.
- When the initial task is assigned to the workflow owner group, Vault assigns the task to the workflow owner and indicates this in the Regulatory Objective Details. Vault displays the following message: “This workflow will be assigned to the workflow owner.”
- Unsupported configurations: If the verification workflow is configured with the Verify Registration Data task as the initial task, users cannot submit Manage Registered Details (MRD) or advance to the next page.
- In this scenario, Vault displays the following error: “The verification workflow configuration is not supported. The workflows will not be generated based on your current settings. Please contact your administrator.”
- Configuration errors: When Vault cannot determine initial participants, it displays the following error: “Unable to determine initial participants for workflow. This configuration may not be supported.”
Allow Disabling of Event Change Action ValuesAuto-on26R1.2
Admins can now inactivate Event Change Action picklist values used in the Create & Manage Event Details action. This update provides flexibility to control which action types are available for selection.
What’s New?
In this release, Vault Admins can inactivate specific values within the Event Change Action picklist. This allows you to restrict the options users see when they run the Create & Manage Event Details action. For example, you can hide actions like Replace or Update and only display Add and Withdraw.
Key Benefits
- Process control: Limits users to specific actions that align with your organization’s business requirements.
- Improved adoption: Reduces user confusion by removing options which are not supported by your business process from the interface.
- Targeted selection: Ensures users only see relevant values based on your configured picklist settings.
Additional Considerations
- Action behavior: The underlying behavior and associated validation for the Create & Manage Event Details action have not changed.
- UI visibility: This change only affects which values are available for selection; it does not impact existing data.
- Note: We recommend reviewing your current business processes before inactivating picklist values to ensure users still have access to necessary actions. At least one picklist value must be active for the Create & Manage Event Details action to be executed.
Wizard Updates for Enhanced Handling of Local DifferencesAuto-on26R1.2
Vault RIM now includes updates to various Registrations wizards to improve the handling of Active Substance, Inactive Ingredient, and Manufactured Product Dosage Form details on Events, Registrations, and other local records. These updates ensure data flows accurately across regulatory transactions and local regulatory information when you use the local differences feature to capture local Product Variant attributes.
What’s New?
Suppressing Special Records in Create Event Details
Admins can now set the Suppress Special Records attribute to Yes on the Event Change Detail Field subcomponent. This attribute is available for Event Change Details referencing the Event Inactive Ingredient object and Event Change Detail fields referencing the Inactive Ingredient field.
When you set this to Yes, the Create Event Details wizard dropdown filters out any Inactive Ingredients with a non-blank value in the Special Record Classification field. Additionally, Vault does not create or propose records for these inactive ingredients, ensuring that Compendial or Non-Compendial records are only created for Manufacturing Details where appropriate.
Updating Join Field Logic for Content Planning
Validation logic for join field handling is updated in the Create Related Records and Update Local Regulatory Information wizards. Vault now only considers target relationship records where Use for Content Planning is set to Yes.
This update ensures join fields for local Inactive Ingredient, Clinical Study, and Extended eCTD Keyword relationships are set correctly, preventing duplicate record creation.
Enhancing Create Registrations & Manage Registered Details
The Create Registrations and Manage Registered Details wizards now use updated validation logic to ensure only relevant records are added to a Registration based on the country’s requirements:
- Registered Active Substances: Vault matches the Active Substance Strength or Active Substance Name when the value is a valid preferred value or local exception for the registration’s Country. At least one Registered Product on the Registration must match the referenced Product Variant from the Product Variant Active Substance (PVAS).
- Registered Inactive Ingredients: Vault matches the Inactive Ingredient Strength or Inactive Ingredient Name when the value is a valid preferred value or local exception for the country. At least one Registered Product must match the referenced Product Variant from the Product Variant Inactive Ingredient (PVII).
- Registered Manufactured Dosage Forms: Vault constrains and adds these only when they are a valid preferred value or local exception for the registration’s Country.
When a Registered Product is a complex product, Vault also uses the Product Variants from its product components in the matching logic.
Key Benefits
- Improved data accuracy: Ensures Vault creates the correct data on Registrations and Events without the need for manual configuration workarounds.
- Reduced duplication: Prevents duplicate records by focusing on Content Planning details for specific relationship fields.
- Process efficiency: Eliminates the need for users to ignore specific error messages, leading to a smoother end-to-end regulatory process.
Additional Considerations
- Enablement: This feature is auto-on, but some configuration is required.
Create Related Records: High-Volume Data SupportAuto-on26R1.2
What’s new?
Vault now supports a 20MB technical payload size for the Create Related Records action (CASRO) initiated from an Event. This 10x increase from the previous 2MB limit allows Vault to process a significantly higher volume of records in a single action.
Multi-Market IDMP Data Model & Aggregation UpdatesAuto-on26R1.3
What’s New?
This release introduces updates to the Veeva RIM data model, aggregation algorithms, and accelerators to support market-specific details for non-EU IDMP compliance. These changes facilitate managing preferred terms and market-level exceptions. You can now use additional support for EEA procedures and manage the Applicable Product Type (Drug vs. Device) for medicinal product accelerators.
Data Model Changes
Administrable Dosage Form Change
- Before: The Administered Product (AP) object type Administered as Manufactured used the Administrable Dosage Form (Reference) lookup field (
administrable_dosage_form__rim). - Now: Vault now uses the Administrable Dosage Form field (
administrable_dosage_form__rim) directly on the Administered as Manufactured object type where Preferred Term is “Yes”. - Reasoning: This facilitates the transition to the model described in the 25R1 feature IDMP Support for Local Differences in Product Variant Attributes where Product Manufactured Dosage Form is a separate object linked to Product and supporting preferred terms and exceptions.
Other Changes
- Medicinal Product: Added an Application field (
application__v) to the Medicinal Product object (medicinal_product__rim). - Administered Product (AP) Relationships: New object types and fields (Preferred Term and Country) support market-level details:
- AP Active Substance: Added Active Substance Preferred Value, Active Substance Strength Exception, and Active Substance Name Exception object types. Added Preferred Term and Country fields.
- AP Inactive Ingredient: Added Inactive Ingredient Preferred Value, Inactive Ingredient Strength Exception, and Inactive Ingredient Name Exception object types. Added Preferred Term and Country fields.
- AP Route of Administration: Added Route of Administration Preferred Term and Route of Administration Exception object types. Added Preferred Term and Country fields.
- AP Reference Strength: Added Preferred Term and Country fields.
Algorithm Changes
Administrable Dosage Form
Aggregation algorithms for IDMP, UPD, and XEVMPD source the Administrable Dosage Form based on the AP object type:
- Administered As Manufactured: The algorithm sources from the legacy lookup field (
administrable_dosage_form_lu__rim) if populated. If blank or inactive, it sources from the new field (administrable_dosage_form__rim). - Modified After Manufacturing: The algorithm sources data directly from the
administrable_dosage_form__rimfield.
Pharmaceutical Product Element Data
IDMP and UPD algorithms evaluate preferred values and market-level exceptions in this sequence:
- Identify Market: Identifies the market related to the Medicinal Product.
- Check for Exceptions: Uses market-specific exception records if available.
- Check for Preferred Terms: Uses the Preferred Term record if no exception exists.
- Fallback: Selects the first detail with a unique attribute combination.
Accelerator Changes
- Administered Product Accelerator: Automatically copies Active Substances and Inactive Ingredients from the Product Variant when you create an AP record. It also populates the Administrable Dosage Form field.
- Medicinal Product Accelerator: Supports European Union Centralised Procedures. For Centrally Authorised applications, Vault now generates Medicinal Product Registration records for EEA countries (Iceland, Liechtenstein, and Norway) based on the lead registration.
New Setting
When you enable the Automatically Generate Medicinal Product Records Application Setting, you can now use a new Applicable Product Type subsetting to control which product types (such as Drug or Device) the accelerator processes.
Key Benefits
- Compliance Readiness: Enhances the data model for future non-EU IDMP.
- Efficiency: Upgrades accelerators to automate complex record creation and ingredient data carrying.
Additional Considerations
- Vault provisions the new object types and fields as inactive by default.
- Vault continues to support the Administrable Dosage Form lookup field. The algorithm prioritizes this field if it is populated.
- The Medicinal Product accelerator updates for non-EU are preparatory. The accelerator currently only generates EU records.
UDI Submissions to GUDIDConfiguration26R1.3
What’s New?
Veeva Registrations now fully supports GUDID including the electronic submission of Unique Device Identifier (UDI) data directly to the FDA Global UDI Database (GUDID) via the Electronic Submission Gateway NextGen (ESGNG). This feature introduces a streamlined machine-to-machine (M2M) connection, allowing you to transmit UDI data individually or in bulk groups of up to 300 submissions.
Key Benefits
- GUDID XML Support: Vault will generate the GUDID data in HL7 SPL Release 5 format and validate the data with technical and business rules in the UDI viewer.
- Direct Transmission: Submit UDI data for the United States directly from the Next Steps page of the U.S. UDI Viewer or via a user action.
- Efficient Bulk Processing: Use UDI Submission Groups to simplify the user experience when managing multiple transmissions at once.
- Automated Status Updates: Vault automatically updates the state of UDI Submission and Registered Device Identifier records to Accepted by HA or Rejected by HA based on the gateway response.
- Flexible Gateway Administration: Admins can provision a standard gateway profile or create custom profiles to support organizations registering devices under multiple legal entities.
Additional Considerations
- Permissions: You must have the Submit UDI Data application permission to invoke electronic transmissions.
- Resubmission Logic: If a submission encounters transmission issues, you can use the Resubmit UDI Data action. This action only includes submissions in the Ready to Submit or Sending to HA states to prevent duplicating already accepted records.
- Notifications: Vault sends a summary notification once all submissions within a group reach a final state.
Assess Local Activities EnhancementsAuto-on26R1.3
In the 26R2 release, the Assess Local Activities page includes updates that provide greater flexibility and efficiency for users. You can now invoke the Assess Local Activities page directly from an Activity or assess activities across multiple Events simultaneously.
To improve usability, the page supports Saved Views and Saved Grid Settings. While Vault automatically persists grid settings such as column widths and grouping preferences across sessions, you can also save specific field and column filters into reusable Saved Views.
Additionally, the grid supports inline editing and fastfill for Date fields.
Key Benefits
- Streamlined Access: Start an assessment directly from an Activity record using the Actions menu.
- Automated Context: When opening Assess Local Activities from an Event or Activity, the filters will automatically include the record where the page was invoked from.
- Multi-Event Assessment: You can now manage local activities across multiple Events in a single interface, which reduces the need to navigate between different records. To enable multi-Event assessment, Vault Administrators must configure the new Include Events in the following states application sub-setting.
- Simultaneous Collaboration: Multiple users can work in the same Event or Activity at once without disrupting other workflows.
- Reduced Manual Setup: Saved Views allow you to skip repetitive filtering and layout configuration by saving your preferences for future sessions.
Create Event Details: Improved FilteringConfiguration26R1.3
Veeva RIM now supports more efficient portfolio management by allowing you to capture the stage of your products and events. This release introduces a streamlined process for managing large portfolios across clinical and commercial stages by reducing manual data entry and improving accuracy.
What’s New?
Veeva RIM includes a Product Stage field on several objects to help you track whether a product is in a nonclinical, clinical, or commercial phase.
- Filtered Event Details: When using Create Event Details, Vault pre-filters Product Family, Product, and Product Variant fields based on the Product Stage set on the Event.
- Clinical Study Product Extension: The Clinical Study Product record now includes the relevant Product Variant.
- Data Model Updates: Veeva RIM adds the Product Stage picklist (
product_stage__v) to the following objects:- Product Family (
product__v) - Product (
drug_product__v) - Product Variant (
product_detail__v) - Event (
event__rim)
- Product Family (
Key Benefits
- Improved Accuracy: Automatic filtering ensures you only select values applicable to the specific Event you are working on.
- Simplified Search: Finding relevant products is faster because non-applicable values are removed from the list.
- Granular Tracking: You can accurately track impacted product variants which are specifically related to your clinical studies.
Additional Considerations
- Picklist Configuration: The
product_stage__vpicklist includes Nonclinical, Clinical, and Commercial values by default. While you can edit the picklist label or make values inactive, the API name is not editable. - Out of Scope: This feature does not include automatic triggers to set the product stage, nor does it auto-populate product fields based on a Clinical Study selection.
- Veeva Connections: This update does not impact RIM connections with PromoMats, Safety, or Medical.
EUDAMED XSD Version 3.0.30 SupportAuto-on26R1.3
Vault RIM now supports the latest EUDAMED XSD Version 3.0.30 for UDI submissions. While this update primarily involves a default version number change for UDI submissions, it ensures your Vault remains compliant with the most recent technical requirements from the European Database on Medical Devices.
EUDAMED UDI Submission Generation Restricted to Supported LanguagesAuto-on26R1.3
What’s New?
Vault now restricts the languages included in UDI submission generation for specific properties to all official EU languages, as well as Icelandic (IS), Norwegian (NB), and Turkish (TR). When you generate a UDI submission for EUDAMED, Vault automatically skips any language-specific content that falls outside of these supported languages.
Additional Considerations
- Admins: Ensure your users are aware of these restrictions when they enter data into language-specific fields.
- Vault Data: Content in languages other than those supported remain in your Vault but do not appear in the generated UDI submission file.
RIM Submissions, RIM Submissions Archive
Active Dossier: Exclude Submission Country from GenerationAuto-on26R1.2
What’s New?
Vault RIM now allows you to exclude specific submission countries from Active Dossier Item Detail (ADID) creation. This is particularly useful for EU Centralized Procedures where you may need to include all EMA countries in a submission but only wish to generate EU-level ADIDs.
Vault now includes an Exclude from Active Dossier Generation yes/no field on the Application Country and Submission Country objects. When this field is set to Yes on a Submission Country, Vault ignores that country during automated Active Dossier (AD) generation.
To further automate this process:
- Field Defaulting: When you set the value on an Application Country, Vault defaults that value to the corresponding Submission Country record created via the Submission Wizard or Create Related Records Wizard.
- Procedure Constraints: You can use the new Exclude from Dossier by Constraint field on the Country object alongside Procedure Type Country Constraint records. When configured, Vault automatically sets Exclude from Active Dossier Generation to Yes on Application Country records based on the procedure type, such as a Centralized Procedure.
Key Benefits
- Simplified EU Management: Efficiently manage Centralized Procedures by generating only required ADIDs while maintaining a complete list of submission countries for tracking.
- Increased Automation: Reduce manual data entry by defaulting exclusion settings from the application level to the submission level.
- Improved Accuracy: Ensure ADID creation consistently aligns with specific procedure types through automated country constraints.
Additional Considerations
- Enablement: This feature is auto-on, but some configuration is required.
- Manual Generation: This exclusion logic only applies to automated generation. If you manually drag and drop a document into the Active Dossier Viewer, Vault creates ADIDs for all submission countries regardless of the exclusion field value.
- Layout Updates: Vault provisions updates to the
application_country_layout__vandsubmission_country_layout__vstandard layouts as part of RIM Core Data Model changes.
Active Dossier: Improved Uniqueness Key CalculationAuto-on26R1.2
Vault has improved the Active Dossier Uniqueness Key calculation to enhance performance and prevent the creation of duplicate Active Dossier records during generation. As part of this enhancement, Vault introduces system safeguards that prevent users from deleting Application, Submission, and Country records if they are actively referenced on existing Active Dossier Item Detail records.
Key Benefits
- Maintaining Data Integrity: Active Dossier Item Detail records capture the context of a document within a country using regulatory transaction references. Preventing the deletion of related foundational records avoids orphaned records.
- Ensuring Accurate Automated Calculations: Vault scopes automated calculations, such as the Latest for Authoring (LFA) value, based on Application, Country, and Product metadata. Maintaining these records ensures LFA and document status calculations remain accurate.
- Improving System Performance: Refining uniqueness key calculations and preventing duplicates improves the speed of Active Dossier generation and updates.
Additional Considerations
This update is a backend enhancement. No additional Admin configuration is required.
Safety
For the latest central dictionary updates for your Safety Vault, see Safety Central Dictionary Updates.
Features in the Veeva Connections section also affect the Safety application family.
Safety
Web Intake FormsConfiguration26R1.2
To facilitate structured data intake for spontaneous adverse event reporting, Veeva Safety introduces Web Intake Forms, which enable the direct collection of structured adverse event details from consumers and service providers managing post-marketing programs. This streamlined intake process reduces manual entry and ensures data consistency from the point of origin.
Key capabilities include:
- Configurable Web Forms: Allows Admins to design and deploy customized web-based forms tailored to specific programs or reporter types, capturing correct information upfront.
- Direct-to-Vault Intake: Streamlines submissions directly into the Safety Inbox as structured records, minimizing the need for manual transcription from PDFs and emails.
- Standardized Data Mapping: Enhances data quality by enforcing field-level validation at the source, leading to faster triage and more efficient case processing.
These updates bridge the gap between external reporters and the Veeva Safety database, reducing administrative overhead and accelerating the timeline for case processing.
Learn More
- Enablement: Enable Web Intake Forms
- Admin Help: Configure Web Intake Forms
- User Help: Web Intake Forms
Safety Rules Troubleshooting Tool for Localized CaseAuto-on26R1.2
To streamline the validation of specialized regional requirements, the Safety Rules Troubleshooting tool has been expanded to support Localized Cases, providing full visibility into regional rule evaluations. Previously, this diagnostic tool was limited to global Case records, limiting visibility into regional reporting logic, such as Japan reportable cases or domestic cases with a local localization scope.
With this release, the troubleshooting tool has been updated to accept both standard Case IDs and Localized Case IDs. When troubleshooting a Localized Case ID, Vault executes a detailed, asynchronous log of every registration, Safety Rule Set, and Safety Rule Parameter processed to provide technical insight into why a specific localized assessment passed or failed a reporting requirement.
Key capabilities include:
- Japan Localized Reporting Log: Vault now provides a dedicated reporting log for Japan scenarios that can be downloaded as a CSV. This log includes critical data points such as the Reporting Scenario, Destination ID, Rule Name, and the specific Localized Case Assessment IDs that passed or failed.
- Transmission Profile Selection Reasons: For all submission evaluations, the troubleshooting log now includes a Transmission Profile Selection Reason column. This identifies the exact logic Vault used to choose a profile, such as Product Registration, Study Registration, Profile Scope Matching, or Fallback logic.
Learn More
- User Help: Troubleshooting Safety Rules
Case Link ReasonAuto-on26R1.2
Veeva Safety now provides a standardized way to define relationships between linked cases, improving traceability and clarity. This update removes the need for custom fields and ensures that important context such as shared literature sources or familial relationships is captured consistently and in alignment with international regulatory standards.
Key enhancements include:
- Standardized Link Reasons: A new Link Reason field has been added to the Case Relationship object, providing pre-defined values for common scenarios including Parent/Child, Same Patient, Same Reporter, and Same Literature Article.
- Regulatory Compliance Alignment: The introduction of this standard field supports compliance with ICH and GVP Module VI (C.1.10.r) requirements for identifying and evaluating related reports together.
- Immediate UI Visibility: To streamline the review process, the Link Reason column appears by default in the Related Cases section of the standard Case layout, allowing users to understand case connections at a glance without navigating to sub-records.
- Data Persistence: Vault ensures that link reasons are preserved during deep copy actions, such as when creating a follow-up Case via a user action, maintaining the integrity of the relationship history.
Learn More
- User Help: Case Field Reference
FDA MedWatch 3500A: September 2025 VersionConfiguration26R1.2
With this release, Veeva Safety introduces support for the September 2025 version of the FDA MedWatch 3500A form. Vault incorporates the required data model for this latest version, which is available for preview, submission, and distribution of ICSRs for drug, biologic, medical device, and clinical trial cases. This update includes an application setting that enables Admins to manage the version cutover independently. Previously, Admins had to contact their Veeva Representative to activate a specific version of the form. The application setting defaults to the version of the form your Vault currently exports. If you want to continue with the same version, you do not need to take any action.
Learn More
- Enablement: Enable FDA MedWatch 3500A
- Admin Help: Manage Products
- User Help: FDA MedWatch 3500A (September 2025 Version) Generation Mapping
Configurable Event Reported Verbatim on ImportConfiguration26R1.2
Streamlining how adverse events are captured from international data sources is critical for global safety reporting. This update introduces refined logic for populating event fields when the E2B(R3) or JSON file includes an English translation value but is missing native language terms. With this release, Veeva Safety ensures that if the (E.1.1.1a) event fields are blank, Vault automatically leverages the Event Reported (English) (E.i.1.2) value. This reduces manual data entry and ensures consistency across global transmissions.
Key capabilities include:
- Automated Data Mapping: If both the native event term (E.i.1.1a) and the language code (E.i.1.1b) are missing, Vault maps the English translation (E.i.1.2) into the Event (Reported) field and defaults the language to English (ENG).
- Configurable Admin Control: A new Transmission Profile setting controls this logic. In Vaults with the Enable Localization-based Mapping on Import setting enabled, Vault evaluates specific E.i elements to determine the best available data source.
- Visual Source Transparency: A new visual indicator icon appears on the Source Data Panel next to the Event Reported field. Users can hover over this icon to see the tooltip stating Import from Event Reported (English), providing clear lineage for the data.
- Preservation of Coding: To maintain data integrity, this automated logic does not override any MedDRA coding already performed on the record.
Learn More
- User Help: E2B Case Import Data Mapping
PMDA Comments AssistanceConfiguration26R1.2
To streamline compliance with PMDA reporting requirements for section J.2.11 (Other References), Veeva Safety now captures more detailed expectedness information on localized records. This enhancement makes the context for unexpected events, such as whether a term is listed on the datasheet or considered unexpected due to its seriousness, clear through additional information on localized records.
Key enhancements include:
- Granular Localized Data Model: For added clarity, Vault populates the reason for an expectedness evaluation, such as Unexpected Due to Seriousness, in the new Expectedness Details field on Japan Localized Case Assessments.
- Localized MedDRA Term Mapping: Based on the Event (LLT) on the global Case, Vault now populates a Localized Event (LLT) on Japan Localized Case Adverse Events, providing the necessary Japanese MedDRA translation for localized regulatory workflows.
Learn More
- Enablement: Enable PMDA Local Case Processing Automations
- User Help: PMDA Case Field Reference
Partially Completed Follow-up QuestionnairesConfiguration26R1.2
With this release, Veeva Safety introduces the ability to capture partially completed follow-up questionnaires abandoned by the reporter. This feature allows organizations to adopt online questionnaires ensuring that Vault brings in all data, including incomplete information.
Key capabilities include:
- Automated “On Behalf” Submission: Vault automatically submits online questionnaires on behalf of the respondent, ensuring that all entered data is captured.
- Configurable Auto-Submission Schedules: Admins can define a specific schedule (measured in hours) after which a distributed questionnaire is considered abandoned and triggered for auto-submission.
- Inbox Item Generation: Vault creates a new Inbox Item for each auto-submitted questionnaire. This allows safety teams to review the partially provided data and determine if further follow-up or case updates are required.
Learn More
- Enablement: Enable Follow-Up Questionnaires
- Admin Help: Set Up Follow-Up Questionnaires
- User Help: Send a Follow-Up Questionnaire to Case Contacts
Case Compare: MedDRA Preferred Term MatchingAuto-on26R1.2
When you recode a MedDRA Lowest Level Term (LLT), the Inbox Item to Case Compare and Case Version Compare pages now broaden matching criteria to the Preferred Term (PT) level. Previously, a change in LLT caused Vault to identify the record as New, leading to potential redundant entries. This feature is auto-enabled in all Vaults to increase matches for coding updates, though organizations that prefer strict LLT-level matching can disable this application setting.
Learn More
Unknown Formulation Product VerificationAuto-on26R1.2
For organizations using Company Product Match Verification and Unknown Formulation Product Selection, when verifying whether a reported product with incomplete data matches a Product Registration Vault previously returned low Product Match Confidence results. Veeva Safety now improves the accuracy of the Product Confidence Match to increase early-stage product identification.
With this release, when a product with an unknown formulation is reported, Veeva Safety performs verification checks against all Product Registrations within the Product Family, rather than failing due to missing registration details. By validating reported data such as Country, Dose Form, and Route of Administration against broader registration records, the update improves matching accuracy and confidence evaluations.
Learn More
- User Help: Inbox Item Study and Product Matching
Validation Criteria: Failed Validation Error HandlingAuto-on26R1.2
Veeva Safety now automatically clears failed validation results when underlying data has been removed from a Case. This improves compliance reporting accuracy and reduces manual cleanup by keeping validation results aligned with the current state of Case data, so users do not spend time reviewing outdated errors.
Key enhancements include:
- Automated Obsolete Result Cleanup: The Evaluate Regulatory Conformance action identifies and removes validation results that reference records, such as Case Products, that have been deleted since the previous Evaluate Regulatory Conformance run.
- Redirected Object Support: This update supports scenarios where validations for child objects, such as Case Product Dosage or Case Product Indication, are associated with the parent Case Product. Validation results linked to deleted child records are now properly cleared.
- Unified Validation Consistency: The removal of outdated results is reflected at both the Case level and the Transmission level during the Generate Transmission Document(s) entry action, ensuring a consistent view of data integrity across the submission lifecycle.
Blind Isolation: Simplified Japan Registration UnblindingAuto-on26R1.2
To ensure data integrity and submission accuracy for clinical trials in Japan, Veeva Safety now synchronizes Case Product Registrations (CPRs) following unblinding. This enhancement prevents the creation of duplicate or conflicting registration records that previously occurred when the Isolate Blinded Clinical Trial Information feature generated dual records for blinded and unblinded product states.
Key enhancements include:
- Automated Blinded Record Cleanup: Upon generating new CPRs, Vault identifies and removes existing registrations from when the Case Product was blinded.
- Accurate Unblinded Generation: Vault creates new CPRs specifically for the unblinded Case Product, ensuring that the most current and accurate data is used for regulatory reporting.
- Follow-Up Case Consistency: The simplified registration logic extends to follow-up cases, ensuring that when unblinding occurs later in the case lifecycle, Vault does not preserve orphaned or redundant blinded registrations.
- Manual Entry Support: The robust cleanup identifies and removes even manually created blinded CPRs upon unblinding, maintaining a standardized record state across Vault.
Japan Reporting: Cross Reporting Registration ImprovementsAuto-on26R1.2
To enhance the precision of Japanese regulatory reporting, Veeva Safety now automatically filters out Combination Products during substance-based cross reporting. This update ensures that your registration data remains clean and focused solely on the submittable product entities required by the PMDA.
Key enhancements include:
- Redundancy Prevention: By excluding parent-level Combination Products from cross reporting evaluations, Vault avoids the creation of unnecessary registration records that were previously triggered by shared substances.
- Constituent-Level Accuracy: Vault continues to evaluate individual Product Constituents, ensuring that all standalone Products within a combination are accurately captured for Japanese reporting.
- Reliable Data Mapping: This refinement resolves issues where unexpected combination records appeared in matches, significantly reducing the manual effort required to clean up registration logs.
Japan Reporting: Improved Validations for Blinded Product SubmissionsAuto-on26R1.2
To ensure regulatory submissions for clinical trials in Japan remain compliant without generating unnecessary errors, Veeva Safety has refined the PMDA.G.k.2.3.r.1 validation logic. This update acknowledges that for actively blinded products, substance information is inherently unknown and cannot be reported. By making this validation conditional, Vault prevents “missing data” flags on blinded products, allowing Safety teams to proceed with PMDA submissions more efficiently.
Key enhancements include:
- Conditional Substance Validation: Vault suppresses the PMDA.G.k.2.3.r.1 validation check for any Case Product where blinding is active, preventing false-positive errors for unknown substance data.
- Targeted Compliance: Vault continues to enforce substance data requirements for all non-blinded products, ensuring that open-label or unblinded records maintain full regulatory compliance.
- Reduced Submission Friction: Eliminates the need for manual workarounds or data “placeholders” previously used to bypass substance-related validation errors in blinded clinical trial cases.
- Support for Active Blinding: Directly supports the workflow where actively blinded products are submitted to the PMDA, ensuring Vault reflects the reality of clinical trial data constraints.
Validation Criteria: MFDS Validation Improvements for Device ProductsAuto-on26R1.2
To improve the accuracy of regulatory conformance checks for the South Korean market, Veeva Safety has updated the MFDS validation logic to better distinguish between drug and device products. Previously, several validations did not account for product type, leading to false-positive errors when device cases were missing drug-specific identifiers. This update ensures that device products are no longer evaluated against inapplicable criteria, streamlining the path to submission.
- Product-Aware Validation Logic: Vault now evaluates the Case Product type before triggering specific MFDS validations to ensure that medical devices are only measured against relevant criteria.
- Exclusion of Drug-Specific Data Points: Validation rules for identifiers that do not exist for devices, such as MPID or Substance information, are no longer evaluated or failed for device Products.
- Broad Case Scope Coverage: These logic improvements apply across various reporting scenarios, including Spontaneous Domestic, Foreign Localized, and Other Study cases.
NMPA Validation Criteria: Exclude External ProductsAuto-on26R1.2
Veeva Safety now excludes External Products from the NMPA.G.k.CN.4-1 validation criteria to streamline regulatory reporting for the Chinese market and reduce false-positive errors. This update ensures that case processors are only prompted to provide local registration details for relevant investigational products, while continuing to verify that CTA Approval Numbers are populated for all applicable localized products. By filtering out non-relevant products, teams can focus on resolving valid data gaps more efficiently without unnecessary warnings.
Safety PBRER Case Listing for Summary Tabulation of ADRAuto-on26R1.2
Providing a structured list of cases within aggregate reports is essential for detailed safety analysis and regulatory transparency. Previously, while clinical trial cases included Interval Line Listings, the Summary Tabulation of ADR from Postmarketing Sources lacked a usable case listing, and existing log files were insufficient for deep data analysis.
With this release, Veeva Safety enhances the listing of cases in Safety aggregate reports. Vault now generates a detailed format for the list of cases included in the Summary Tabulation of ADR from Postmarketing Sources, mirroring the format used for Clinical Trial Interval Line Listings.
Key capabilities include:
- Comprehensive Data Access: Vault now generates a dedicated Case Listing for all cases used in the calculations for the PBRER Summary Tabulation, ensuring no data points are left unverified.
- Improved Analytical Utility: Moving beyond basic log files allows teams to perform deeper reconciliations on the postmarketing data used within the report.
- Standardized Reporting Structure: The listing format for postmarketing ADRs now mirrors the OOTB (out-of-the-box) structure used for Serious Adverse Reactions in Clinical Trials, providing a consistent user experience across different PBRER sections.
Learn More
- User Help: Generate Aggregate Report Tabulations
Responsive UI for Safety Auto-on26R1.2
To provide a modernized and intuitive user experience across varying device sizes, Veeva Safety optimizes the responsive framework introduced with the Responsive UI Platform feature. This transition ensures that critical case processing tools, such as the MedDRA and WHODrug browsers, remain fully functional on smaller screens by displaying full search bars and action buttons during navigation.
Furthermore, certain complex control fields have been redesigned to render correctly without overlapping data or hiding essential labels. Beyond these functional updates, the UI now features improved label alignment and standardized spacing in key areas like the Patient section of Inbox Items and the Narrative section of Cases to facilitate a faster, more accurate review process.
To maintain a consistent brand identity, Vault applies the same font across all metrics pages and literature sections, while optimizing comparison tools, such as the Case Version Compare page, to support flexible browser resizing.
Inbox Item Data ArchivingAuto-on26R1.2
To maintain optimal system performance and data organization, Veeva Safety now automatically archives Inbox Items following 180 days of inactivity after promotion. This enhancement ensures that the Inbox remains clear of processed records, allowing intake teams to focus exclusively on active items without the clutter of legacy data. Automating the transition of promoted records to an archived state improves overall dashboard efficiency and streamlines the end-to-end data lifecycle management process.
Automatically Run Early Notification Rules on Case PromotionAdmin Checkbox26R1.2
To assist with regulatory compliance and reduce the risk of missed ICSR deadlines, Veeva Safety provides the ability to evaluate Early Notification rules during the Case promotion process. This enhancement addresses a gap where reporting rules previously failed silently if executed before the asynchronous generation of Case Assessments was complete.
Vault now provides consistent identification and tracking of urgent early notifications (often due within 24 hours). Key enhancements include:
- Evaluation Trigger: Automatically evaluates Early Notification rules at the precise moment Vault completes Case Assessment generation upon case promotion, ensuring the Safety Rule Engine has access to all necessary data.
- Comprehensive Promotion Support: Applies the automated evaluation to initial Case promotions, as well as follow-up and merge-to-in-flight actions performed through Case Compare.
- Reduction of Manual Overhead: Eliminates the need for Admins to configure complex user actions or entry actions on early Case lifecycle states, which were historically prone to failure due to asynchronous processing.
Learn More
- Enablement: Safety Application Settings & Configuration Options
- User Help: ICSR Transmissions Overview
Email Addresses for Manufacturer SitesAuto-on26R1.2
To meet FDA reporting requirements for medical devices, manufacturer email addresses must often be exported to specific regulatory forms. Previously, the standard Email field was restricted to the Registration Holder object type, which created a data gap when manufacturer details were stored under different organization types. This release expands the availability of the Email field to Sites. This ensures that regardless of how a manufacturer is classified in Veeva Safety, you can capture and map their contact information to FDA device forms.
Exclude Reporter Information by Destination on CIOMS I FormsConfiguration26R1.2
To comply with partner agreements and data privacy standards, organizations often need to withhold specific investigator details when distributing CIOMS I forms externally. Previously, information mapped to Box 25b (such as the investigator’s name, address, and country) was always exported, regardless of the recipient’s privacy requirements.
This release introduces a partner-specific configuration that allows users to redact this information on generated CIOMS I forms. While the investigator data remains fully visible within Veeva Safety for internal use, it is automatically excluded during the transmission process based on the destination’s settings.
Key updates include:
- Partner-Level Privacy Control: A new field for Partners allows organizations to specify whether reporter information should be suppressed for that entity.
- Automated Form Redaction: During transmission generation, Vault evaluates destination partner configuration. If redaction is enabled, Box 25b is blank on generated CIOMS I forms.
Learn More
- Enablement: Enable Exclude Reporter Information by Destination on CIOMS I Forms
- Admin Help: Manage Organizations
- User Help: CIOMS I Generation Data Mapping
Standard FDA eMDR Transmission Profile Document TypeAuto-on26R1.2
With this release, Veeva Safety streamlines the Submission workflow by setting the Outbound Format to FDA eMDR on the standard Transmission Profile for device Submissions to the FDA. This update reduces manual configuration steps and ensures that device-related Submissions consistently include the required documentation for successful electronic Medical Device Reporting (eMDR).
FDA E2B(R3) Relabeled to FDA FAERS E2B(R3)Auto-on26R1.2
To provide clarity across reporting streams, Veeva Safety has updated its naming conventions to better distinguish between the FAERS (drug/biologic) and VAERS (vaccine) reporting systems. With this release, Safety renames the FDA E2B(R3) report to FDA FAERS E2B(R3), assisting users to easily identify and classify documents intended for the FDA’s Adverse Event Reporting System during the submission process. The Vault UI and picklists now match current regulatory nomenclature, reducing the risk of selection errors between reporting channels. While these updates align the Vault interface with the FDA’s latest standards, the underlying document export and data mapping remain the same.
Learn More
- User Help: E2B Generation: FDA FAERS E2B(R3) Mapping
Vault No Longer Creates Gateway ProfilesAuto-on26R1.2
With this release, Vault no longer creates a corresponding Gateway Profile when an Admin creates an AS2 Gateway type Transmission Profile for use with an AS2 Connection. This update modernizes the AS2 connection process for users; there are no changes to the underlying transmission logic or data integrity.
Transmission Profile Creation Limit RemovedAuto-on26R1.2
With this release Admins can create Transmission Profiles without encountering errors previously caused by reaching the Gateway Profile creation limit. Vault now allows for a seamless Transmission Profile creation process, providing Admins with greater flexibility and reducing administrative friction.
MedDRA International Sort OrderAuto-on26R1.2
The MedDRA Expert Working Group has established an international sort order to ensure System Organ Classes (SOC) terms remain consistently ordered across all languages. Previously, International SOC order was not available in the MedDRA dictionary. As a result, reports typically defaulted to alphabetical order, which led to inconsistent data presentation across different languages or MedDRA versions.
With this release, Veeva Safety introduces the International SOC Sort Order on the SOC terms of the MedDRA dictionary. Reports can utilize this information to ensure that SOCs appear in a standardized, medically logical sequence, regardless of the user’s language or the specific MedDRA version in use.
Key updates include:
- Standardized Sort Order Metadata: Vault now includes the official MedDRA-defined numeric sequence (1-27) for SOC terms within the MedDRA dictionary.
- Automated Dictionary Updates: This sorting logic is automatically applied to existing MedDRA versions and will be maintained for all future dictionary uploads.
Learn More
- User Help: Code MedDRA Terms
Web Intake Forms: Support Custom TranslationsConfiguration26R1.3
To simplify adverse event collection across international markets and support localized partner protocols, Veeva Safety introduces comprehensive custom translation capabilities for Web Intake Forms. This update allows organizations to present Web Intake Forms in a respondent’s preferred language, improving data clarity at the point of intake.
Key enhancements include:
- Dynamic Language Selector: Introduces a language dropdown on single-use and repeat-use public gateway landing pages, allowing respondents to select a preferred display language.
- Automated CSV Translation Templates: Provides utility actions to generate, up-version, or bulk-export translation CSV files containing form header keys, giving Admins a structured format to upload string translations.
- Static Text Translations: Extends translation coverage to all static text components, including email verification prompts, document upload cards, timeout dialogs, form review screens, and footer legal notices.
- Respondent-Language Email Alerts: Syncs the communication engine dispatching security links and submission receipts in the respondent’s preferred language.
Document Intake Panel: Support for Additional FieldsAuto-on26R1.3
Efficiently triaging incoming safety data requires immediate access to key document details. Previously, users processing documents using the document intake panel often had to toggle between views or manually enter metadata that resided within the source document on the inbox manually. This process slowed down the initial intake process and increased the likelihood of data entry discrepancies during intake.
With this release, Veeva Safety streamlines the intake process by expanding the available Inbox Item fields on the Document Intake panel. This update allows users to map and import essential data points directly from the document view into the Inbox Item record. By reducing manual navigation and centralizing data entry, organizations can accelerate triage timelines and improve the accuracy of incoming safety information.
Key updates include:
- Enhanced Data Entry & Validation: The Document Intake panel now includes additional fields in edit mode, such as a multi-select Invalid Reason field. If a user selects an Invalid Reason, the Promote to Case action remains enabled, and corresponding sections display an X icon to denote missing critical data.
- Expanded View Mode Visibility: Users can now view comprehensive details directly in the panel’s summary view without entering edit mode. This includes clinical details such as Onset, Cessation, and Outcome, as well as expanded Reporter information (including Middle Name and Address) and Product details (including Indication, Dosage, Frequency, and Administration dates).
- Intelligent Age & Gender Display: The panel now provides logical data visualization by moving patient Gender next to Initials and dynamically displaying Age Group and Age. For example, if an Age Group is present, the Age displays in brackets immediately following it for quick reference.
- Case Identifier Grouping: The view mode now displays up to five Case Identifiers. These are visually grouped with existing ID fields, allowing users to cycle through available annotations.
- UI Consistency & Banner Logic: The panel displays banners for Invalid Reason and Special Situation in view mode.
- Global Date Localization: To ensure consistency, date fields within the panel including Date of Birth, Administration dates, and Event dates now strictly reflect the Vault-level date format setting.
Automatic Inbox Item Creation for Adverse Event Report DocumentsAdmin Checkbox26R1.3
To further simplify the intake process and reduce manual data entry steps, Veeva Safety now automatically generates and links an Inbox Item immediately upon the upload of an Adverse Event Report (AER) document to the Library. This enhancement ensures Vault captures new safety reports without delay, allowing for faster triage and processing.
Key enhancements include:
- Document Routing: Vault automatically identifies uploaded supported file types to trigger the appropriate import process, routing XML files to E2B import and CSV or ZIP files to tabular data import.
- Universal Intake Support: For text and image-based formats (such as PDF, Word, JPEG, and email), Vault automatically extracts case data; otherwise, it creates a blank Inbox Item linked to the source document.
- Zero-Click Linking: This feature removes the manual requirement to run the Create Inbox Item from Document action, ensuring every Case > Source > Adverse Event Report document is immediately actionable within the intake lifecycle.
- Flexible Administrative Control: Organizations can disable this tool for specific workflows by enabling the Disable Automatic Intake of AER Documents application setting or by uploading documents in migration mode.
Invalid & Special Situation Case PromotionAuto-on26R1.3
To align with GVP Module VI (Section VI.B.6), this release automates the tracking of critical case classifications to ensure regulatory compliance. By automating the transfer of these classifications, organizations can significantly reduce the manual administrative burden, minimizing the risk of human error during data entry. This ensures that cases are processed faster and with higher data integrity, directly improving the accuracy of regulatory reporting and maintaining a clear, compliant audit trail for case validity.
Previously, users had to manually re-enter Special Situation and Invalid Reason data when promoting an Inbox Item to a Case. This manual process increased the risk of data discrepancies and created inefficiencies during the promotion and follow-up phases. With this release, Veeva Safety seamlessly transfers Special Situation and Invalid Reason data from the Inbox Item to a Case. This automation streamlines the workflow and ensures that key regulatory markers are maintained throughout the Case lifecycle.
Key updates include:
- Automated Data Mapping: During initial Case promotion and subsequent follow-ups, Vault automatically maps values from the Inbox Item fields directly to the corresponding Case fields.
- Support for Validity Transitions: Vault now supports dynamic logic for scenarios where an “Invalid” Case must transition to “Valid” as new information is received, ensuring the case status is always current.
Admins should ensure that the Special Situation and Invalid Reason fields are visible on both Inbox Item and Case object layouts to allow users to review and manage these automated values.
Case SignificanceAuto-on26R1.3
To streamline case prioritization and ensure data integrity, Veeva Safety now automatically promotes the Significance value from Inbox Items to the corresponding Case record. This enhancement ensures that the prioritization assigned to incoming information is accurately reflected throughout the Case lifecycle.
Key enhancements include:
- Automated Significance Mapping: During both initial case promotion and follow-up creation, Vault maps the Inbox Item Significance to either the Significant or Non-Significant field on the Case.
- Data Consistency Across Versions: Vault ensures that the significance level remains consistent from the intake stage through case promotion, reducing discrepancies that can arise from manual edits.
- Merge Visibility: On the Inbox Item to Case Compare Page page, a blue information icon now appears when merging into an active Case version. Vault displays a hover message clarifying that while merging new information, Vault will not update the Significance of the matched in-flight Case, preserving existing triage values.
Local Case ResyncConfiguration26R1.3
To eliminate manual data entry and prevent reporting delays, Veeva Safety now allows users to refresh foreign Localized Cases with the latest global data at any time. This new capability ensures that local affiliates have immediate access to the most up-to-date case information, even after a Transmission has been generated or if records were missing during migration.
Key improvements include:
- On-Demand Data Refresh: The new Resync Foreign Cases action maps updates from a global Case to all of its associated foreign Localized Cases whenever changes occur post-approval.
- Migrated Data Healing: During data resyncing, Vault generates missing localized child and grandchild records for Cases created via migration, ensuring full data integrity for historical records.
- Standardized Administrative Control: Admins can configure Resync Foreign Cases as a user action, entry action, or workflow action, providing flexibility to match specific organizational business processes.
Safety Case InvestigationConfiguration26R1.3
To streamline compliance monitoring and support audit-readiness, Veeva Safety introduces a standardized approach to safety case investigations. This framework leverages Vault Platform’s dashboards and analytics tools for tracking submissions and transmissions across the organization. By using standardized objects and lifecycles, organizations can maintain a consistent audit trail for all investigational activities without the need for manual tracking in external spreadsheets.
Key capabilities include:
- Dedicated Case Investigation Management: Includes standard investigation objects, layouts, and business lifecycles designed to manage late case investigations from initiation through review and closure, replacing manual or external tracking approaches.
- Standardized Compliance & Audit Tracking: Introduces dedicated lifecycle states for both case investigations and transmissions, providing a clear, audit-ready history of compliance actions and investigation progress.
- Enhanced Submission & Transmission Visibility: New fields, report types, and layouts provide direct linkage between regulatory submissions and investigations, helping teams monitor submission flows to agencies such as AEMS and quickly identify bottlenecks or delays.
Remove Blind Protection for Investigator CausalityAuto-on26R1.3
To provide greater flexibility in clinical trial safety reporting, Veeva Safety removes the mandatory blind protection for investigator causality. This enhancement responds to early adopter feedback from organizations that prefer to allow unblinded users to view investigator causality assessments even when the product itself remains blinded. By making this information visible by default, companies can better align their safety review workflows with specific study protocols or internal SOPs without compromising the integrity of the blinded study medication.
Key capabilities include:
- Standardized Causality Visibility: Investigator causality fields are now visible to users with appropriate permissions, even when the associated Case Product remains blinded.
- Targeted Information Exposure: Vault reveals causality data on Case Assessment and Case Adverse Event records while ensuring the Case Product and Substance information remain strictly isolated and protected.
- Simplified Compliance Reporting: Facilitates certain regional reporting requirements where the causality assessment must be evaluated by unblinded staff regardless of the treatment assignment.
- Maintained Data Integrity: Even with causality protection removed, the core Blinded Reference logic remains active to ensure that the actual product identity is never inadvertently exposed to blinded roles.
Improved Postmarket Combination Product ManagementAuto-on26R1.3
Veeva Safety delivers several improvements to postmarket combination product management, reducing manual effort and improving accuracy across case processing workflows. This release introduces the Case Combination Product object, which displays all product constituents of a Combination Product in a single view. The new structure, which is also available for combination products on clinical trials, simplifies and centralizes coding during case processing.
With this feature, it’s much easier to add, update, or remove combination products and their constituents as more information becomes available. When a Combination Product changes, Vault updates all existing product constituents and downloads any missing ones. This feature also introduces an action for bulk removal of non-constituent Case Products that no longer apply to the Case. To simplify promoting Inbox Items with combination products, the Inbox Item to Case Compare page now groups product constituents, making it easier to review and reconcile product data across case versions. In addition, Vault now supports including multiple instances of the same postmarket combination product on a single Case.
Clinical Trial Combination ProductsAuto-on26R1.3
Veeva Safety extends support of combination products, including the latest improvements, to clinical trial combination products. On clinical trial study Cases with study combination products, Vault automatically adds all constituents when any study product constituent is coded on the case. During case processing, Vault protects blinded information for study combination products. When a blinded primary constituent is unblinded, Vault automatically generates Case Product records for the remaining combination product constituents.
This feature is Auto-on in Vaults with Study Products that include a Combination Product value.
PMDA: Combination Product Registration Variant MatchingAuto-on26R1.3
To improve the precision of regulatory reporting for combination products and reduce manual reconciliation effort, Veeva Safety introduces enhanced logic for the Retrieve Case Product Registration action to better support products using variants. This update ensures that the automated selection of registrations is more accurate for complex product presentations, such as drug-device combinations with multiple delivery systems.
Vault now filters Case Product Registrations by matching the Product Variant when a Combination Product is identified on a Case. By considering only registrations that align with the specific product variant, safety teams ensure that regulatory reports are generated against the correct marketing authorization for that specific delivery system or device variant. In scenarios where no registrations have a Product Variant, Vault defaults to its standard behavior by considering all available registrations, while cross-reporting logic based on substance remains unchanged to ensure global reporting continuity.
Readability Updates to Rules Based Narratives Follow-up SummaryAuto-on26R1.3
Ensuring that case narratives remain concise and professional is essential for clear regulatory communication. Previously, follow-up summaries often contained agranular data points that cluttered the report, making it difficult for reviewers to quickly identify pertinent clinical changes. With this release, Veeva Safety streamlines Summary Statements by simplifying language and refining assessment logic to focus on high value clinical information.
Key updates include:
- Streamlined Narrative Logic
- Simplified Summaries: Data updates for demographics, studies, drug and medical history, and treatment products now use more concise, natural phrasing.
- Focused Assessments: Summaries are now limited to Investigator or MAH assessments, using generic “updated” statements rather than specific result values.
- Cleaner Terminology: Field references in summary outputs no longer include “normalized”, improving readability.
- Enhanced Clinical Tracking
- New Product Alerts: Summary statements now include a specific statement for when a new suspect product has been added to a Case.
- Critical Data Updates: Summary statements now address changes to Cause of Death and Dechallenge information.
- Standardized Formatting: Aligned formatting ensures a professional, consistent tone across all rules-based narratives, reducing the need for manual edits.
Japan Rules-Based NarrativesConfiguration26R1.3
Generating regulatory narratives for the Japanese market often requires specific linguistic nuances and data structures that differ from global standards. Previously, localized narratives for premarket cases required significant manual translation or post-generation editing, which caused inefficiencies during processing ofJapanese regulatory submissions.
With this release, Veeva Safety extends the rules-based narrative framework to generate narratives in Japanese for submission to the PMDA. Admins can now configure localized Narrative Outlines that automatically pull data into Japanese-specific formats, leveraging localized fields and child objects to ensure PMDA compliance.
Medical Timeline: Sorting & Unknown DatesAuto-on26R1.3
To streamline the medical assessment process and provide reviewers with a more flexible view of patient data, Veeva Safety introduces several key user interface enhancements to the Medical Review Timeline. These updates specifically address feedback regarding the chronological flow of events and the handling of incomplete data, allowing medical reviewers to better contextualize adverse events within a patient’s clinical journey. By optimizing how data is displayed and accessed, reviewers can perform more efficient safety assessments without losing track of the primary case information.
Key capabilities include:
- Chronological & Reverse-Chronological Sorting: Users can now toggle the timeline to display events in chronological and reverse-chronological order, making it easier to prioritize the most recent clinical occurrences during a review.
- Visual Indicators for Unknown Dates: To improve data transparency, events with unknown or partially missing dates now feature a distinct visual indicator, ensuring reviewers can immediately identify entries with incomplete temporal data.
- Modeless Popup Window: Reviewers have the ability to open the Medical Review Timeline in a modeless popup, allowing the timeline to remain visible as a reference while the reviewer navigates and edits other sections of the Case.
- Intelligent Date Placement: For events with unknown start and end dates, Vault utilizes a standardized logic to place these items on the timeline, ensuring they are accessible even without precise chronological data.
- Enhanced UI Controls: New UI elements provide a more intuitive experience for adjusting the timeline view, including improved sorting icons and placement indicators for unknown dates.
MedDRA Upversion for Business AdministrationAuto-on26R1.3
To significantly reduce administrative maintenance, Veeva Safety now automatically updates hierarchical MedDRA terms on active Datasheets, Watchlists, MedDRA Queries, and Product-Event Combinations during the dictionary upversion process. Previously, when the MedDRA dictionary was updated, safety teams were required to manually review and update hierarchical changes for HLT, HLGT, and SOC terms on these records to ensure they remained synchronized with the latest dictionary version. This enhancement automatically updates the terms to reflect the latest hierarchical changes, ensuring your safety data reflects the most current dictionary changes removing the need for manual intervention.
Product Browser: Allow Multiple Trade NamesAuto-on26R1.3
Veeva Safety introduces the ability to display multiple matching trade names within the Product Browser to improve the speed and accuracy of the product coding process. This enhancement addresses a previous limitation where the browser would only return a single trade name during a search, potentially obscuring other valid options.
By providing a more comprehensive view of available trade names, the Product Browser search behavior now returns all trade names that match the user’s search criteria rather than restricting the output to a single result. Users can now view the full list of trade names associated with a product record directly within the search interface, ensuring all relevant data is available for selection. Furthermore, the search interface is optimized to handle these multiple results effectively, offering a more intuitive and efficient browsing experience during case intake and processing.
WHODrug Company Product Auto-CodingAuto-on26R1.3
Accurately capturing medicinal product identifiers is critical for regulatory reporting, yet gaps in internal libraries often lead to incomplete data. Previously, while Veeva Safety would snapshot Company Product and Registration information, many organizations lacked WHODrug MPID data within their product libraries. This gap necessitated manual workarounds or third-party supplemental solutions to fill in missing identifiers, increasing the risk of inconsistent data across the Case Product records.
With this release, Veeva Safety streamlines the enrichment of product data by integrating directly with the WHODrug dictionary. When a Company Product is coded, Vault now automatically leverages the WHODrug Koda tool to bridge information gaps. If a match is identified, Vault populates the UMC MPID field on the Case Product automatically, ensuring compliant data without external manual intervention.
Key updates include:
- MPID Enrichment: Vault queries the WHODrug dictionary via Koda to map missing UMC MPID data when a Case Product Registration field is populated or updated, using the reported trade name, substance, country, and dose form.
- Efficient Lookup Caching: Stores verbatim search strings and resolved MPID matches in a new WHODrug Koda MPID Cache object, allowing identical future requests to resolve instantly without re-querying the external dictionary.
Case Validation: Masked and Blinded DataAuto-on26R1.3
To ensure high data quality and a more compliant validation process, Veeva Safety introduces full support for validating Case data that is either masked, blinded, or undergoes data transformation during file generation. This update transitions legacy direct-file validations into Vault’s Case Data Expression framework, resolving previous technical limitations associated with complex data scenarios. By aligning these validations with the modern configuration engine, organizations can ensure data accuracy throughout the case lifecycle without compromising the integrity of the blinded study medication.
Vault transitions approximately 411 legacy file-based validations into the Case Data Expression framework while introducing new expression functions specifically designed to evaluate data that is masked or blinded in the UI. Furthermore, it optimizes validation levels by automatically moving certain submission-only checks to the Case level, allowing users to identify and correct errors earlier in the lifecycle without altering the familiar end-user experience. This shift fully aligns the application’s validation offering with regulatory standards by evaluating the actual data transformations that occur during E2B file generation.
Validation Criteria: EMA Identifiable Patient UpdatesAuto-on26R1.3
To ensure consistent alignment with EMA E2B(R3) data privacy standards and to streamline the reporting of blinded or masked cases, Veeva Safety introduces updates to the EMA Identifiable Patient validation criteria. This enhancement specifically modifies the validation logic for EMA.D.1 to support the use of “MSK” (Masked) across all patient-identifiable fields. By allowing masked values in these fields, organizations can fulfill EMA identifiability requirements without compromising data privacy or triggering unnecessary validation errors.
Key capabilities include:
- Support for Masked Data (MSK): Updates the EMA.D.1 validation rule to allow “MSK” as a valid entry for all identifiable patient fields, ensuring that masked data does not result in failed regulatory conformance checks.
- EMA E2B(R3) Compliance Alignment: Aligns the application’s validation framework with the latest EMA technical specifications for patient identifiability.
- Reduction in False Validation Errors: Eliminates conformance errors for cases where patient details are intentionally masked for privacy reasons or blinded trial protocols.
- Standardized Validation Logic: Applies the updated logic consistently across Vault’s core validation engine for all Cases requiring EMA submission.
View E2B Readable Renditions in VaultAuto-on26R1.3
Navigating between E2B(R3) reports and their readable formats often required manual file management, as the E2B Readable Rendition previously triggered a local download. This interrupted the user workflow by forcing them to leave the Vault interface to view the generated PDF. Additionally, managing these files across different document versions could lead to versioning confusion.
With this release, Veeva Safety streamlines the review process by opening the E2B Readable Rendition directly in a Vault pop-up document viewer. By transitioning the file from a standard rendition to a version-controlled attachment, users can now view, compare, and access these reports without leaving their browser.
FDA FAERS E2B(R3): Replace Unsupported CharactersAuto-on26R1.3
To ensure successful transmissions to the FDA AEMS (Adverse Event Management System, formerly known as FAERS) and eliminate rejections caused by incompatible text formats, Veeva Safety introduces automated character replacement and validation for FDA FAERS E2B(R3) submissions. This feature addresses a critical gap where UTF-8 characters (such as smart quotes or non-English symbols) would previously cause file rejections at the FDA gateway. By automatically sanitizing the export file, safety teams can significantly reduce the need for manual data corrections and ensure a higher first-time acceptance rate.
Key capabilities include:
- Automated Character Replacement: During the generation of an FDA FAERS E2B(R3) file, Vault automatically replaces any unsupported UTF characters with their ISO 8859-1 equivalents.
- Enhanced Validation Logging: Provides clear, actionable feedback to case processors when unsupported characters are detected, allowing them to address potential rejections before submission.
FDA Reporting: Unique Device Identifiers UpdateAuto-on26R1.3
Accurate reporting for medical device adverse events requires precise alignment with FDA labeling standards. Previously, to populate a Unique Device Identifier (UDI) on FDA eMDR and FDA MedWatch 3500A (September 2025 version) forms, Vault mapped the Unique Device Identifier from Device-type Case Products. However, per FDA guidance, a complete UDI is a composite of the Device Identifier (DI) and the Production Identifier (PI). Relying on a single field often resulted in incomplete submissions that did not meet the latest regulatory expectations.
This release updates the export to ensure compliance on both FDA forms. Vault now automatically concatenates the Unique Identifier and Unique Production Identifier values on the Case Product to provide a complete value in the D4 field of both forms.
FDA FAERS E2B(R3) Primary IND SelectionAuto-on26R1.3
Accurate regulatory reporting to the FDA requires precise mapping of Study Registration Numbers to the FDA.C.5.5a data element. With this release, Veeva Safety introduces the FDA Primary IND setting in Transmission Studies, enabling Admins to override the most relevant Study Registration for cross reporting.
Now, during FDA FAERS E2B(R3) generation, if no Case Study Registration for the United States exists, Vault now executes a specific hierarchy to identify the correct registration number. Vault first checks for a Transmission Study with FDA Primary IND enabled and a Study Registration for the United States. If no primary FDA IND exists, Vault exports the Study Registration Number from the Study Registration with the FDA Primary IND setting enabled and with a Study Product that matches the primary suspect Case Product. In cases where multiple Study Registrations meet these criteria, Vault automatically exports the earliest-created registration to ensure consistent and compliant FDA.C.5.5a population.
FDA Reporting: Support for Individual Study Device SubmissionsAuto-on26R1.3
A single clinical trial case can include multiple device-type products. To simplify clinical trial reporting for combination products and multi-device study protocols, Veeva Safety now supports populating individual study devices on FDA MedWatch 3500A (August 2024 version) forms. By using the existing Case Product field on Transmissions, Vault can isolate a single study device per submission.
Key enhancements include:
- Targeted Device Filtering in Sections D and H: When you select a device study product on a Transmission, Vault maps data to all Section D and Section H fields for that product exclusively.
- Preserved Concomitant Capture in Section C: Standard data mapping logic for Section C is maintained, ensuring all non-device products on the case continue to export in full to preserve comprehensive therapeutic context.
- Study Identifier Optimization in Section G: For clinical trial study Cases, Vault checks for active FDA Investigational New Drug (IND) registrations and maps the earliest-created registration number. If no IND registration is found, Vault falls back to the Case Product Registration.
- Device Qualification Logic: Before applying form filtering, Vault evaluates whether the selected product qualifies as a device by checking core object type configurations, destination jurisdiction country registrations (Device or OTC Device), and external suspect product types.
- Blank Field Fallback: If the Transmission does not specify a Case Product, Vault continues to map all devices on the Case to the form, ensuring no disruption to current workflows.
Enhanced E2B Generation Warning NotificationsAuto-on26R1.3
This enhancement clarifies unsupported unit of measurement notifications during regional E2B generation. Previously, when unsupported units of measurement were exported, the Vault notification and email could reference the incorrect document type, making troubleshooting more difficult for users reviewing the submission. With this update, Veeva Safety provides more accurate and detailed information to help users quickly identify and resolve data issues during E2B generation.
Key improvements include:
- Accurate Document Type Identification: Notifications now specify the regional transmission document type, for example, PMDA E2B(R3) or FDA FAERS E2B(R3), providing clearer export context.
- No Workflow Changes Required: The enhancement continues to use the existing export notification email framework, allowing organizations to maintain their current operational processes and monitoring workflows.
FDA Reporting: NullFlavor Export UpdatesAuto-on26R1.3
To streamline safety submission workflows and reduce data entry overhead for regulatory compliance, Veeva Safety optimizes nullFlavor exports during FDA reporting. When generating FDA E2B(R3) files with the FAERS and VAERS formats, Vault now evaluates required context such as patient and reporter identifiers. If certain text fields remain unpopulated during case processing, Vault populates the standard regulatory code placeholders; such as ASKU (Asked but Unknown) or UNK (Unknown), on the submission.
Embedding these regional requirements minimizes validation noise following data entry. As such, numerous legacy Validation Criteria that previously flagged missing contact or demographic details as errors are now inactive. This alignment ensures that transmissions comply with health authority rules without requiring manual data correction, accelerating submission cycle times and lowering technical gateway rejections.
Process eMDR ACKsAuto-on26R1.3
Managing medical device reporting requires seamless integration with regulatory feedback. Previously, while Veeva Safety effectively processed standard E2B ACK files, the specific nuances of FDA eMDR (Electronic Medical Device Reporting) submissions required more robust handling to ensure full visibility into submission status and documentation.
With this release, Veeva Safety expands its ingestion capabilities to support the unique acknowledgment package provided by the FDA for eMDRs. This update ensures that Safety teams can reconcile device submissions with the same automation and accuracy as for drug safety reports.
Secure DTD Extended to Swissmedic EMA E2B SubmissionsAdmin Checkbox26R1.3
Vault extends secure DTD hosting support to Swissmedic submissions, aligning with modern cybersecurity requirements across the SDX pipeline.
As regulatory authorities move away from unencrypted HTTP communication, this update expands an existing application preference to ensure outbound files transmitted to Switzerland use HTTPS by preventing gateway security failures and compliance rejections.
Swissmedic Security Integration: Swissmedic (swissmedic_ch__v) is added to the secure DTD destination framework alongside existing secure routing profiles for the EMA and MHRA.
Dynamic Protocol Handling: When the global security toggle is enabled, the file generation engine maps the secure HTTPS path directly into file headers for Swissmedic destinations. If the setting is disabled, file headers revert to standard HTTP, preserving existing transmission behavior.
eMDR Device Form ValidationsAuto-on26R1.3
To ensure the highest level of regulatory compliance and data integrity, Veeva Safety has introduced automated Case Validations for eMDR device forms. By streamlining the data verification process before submission to health authorities, this feature reduces the risk of technical rejections and significantly alleviates the manual validation burden for case processors.
Key enhancements include:
- Comprehensive Device Validation: Vault now provides full validation coverage for the FDA’s eMDR.
- Standardized User Experience: These updates utilize the existing Case Validation framework, ensuring that case processors can identify and resolve data discrepancies using the same familiar interface as E2B(R3) submissions.
- Targeted Compliance Checks (or Report-Specific Logic): Validations are differentiated by report type, ensuring that specific requirements for serious incidents, field safety corrective actions (FSCAs), and trends are accurately verified.
- Customizable Rules: Admins can leverage new functions to configure custom validations, allowing organizations to tailor compliance rules to their specific business needs or localized safety protocols.
MIR Device Form ValidationsAuto-on26R1.3
To ensure the highest level of regulatory compliance and data integrity, Veeva Safety has introduced automated Case Validations for Manufacturer Incident Report (MIR) device forms. By streamlining the data verification process before submission to health authorities, this feature reduces the risk of technical rejections and significantly alleviates the manual validation burden for case processors.
Key enhancements include:
- Comprehensive Device Validation: Vault now provides full validation coverage for the EU’s MIR.
- Standardized User Experience: These updates utilize the existing Case Validation framework, ensuring that case processors can identify and resolve data discrepancies using the same familiar interface as E2B(R3) submissions.
- Targeted Compliance Checks (or Report-Specific Logic): Validations are differentiated by report type, ensuring that specific requirements for serious incidents, field safety corrective actions (FSCAs), and trends are accurately verified.
- Customizable Rules: Admins can leverage new functions to configure custom validations, allowing organizations to tailor compliance rules to their specific business needs or localized safety protocols.
Device Agency for EU CountriesConfiguration26R1.3
Managing regulatory compliance within the European Economic Area (EEA) requires nuanced handling of different governing bodies. While many countries fall under the EMA for medicinal products, medical device reporting is increasingly centralized under EUDAMED. Furthermore, certain countries (such as Turkey and Switzerland) operate outside the EMA’s jurisdiction entirely.
This release introduces the ability to link a secondary, device-specific agency to a country. This ensures that when a medical device is registered in a specific country, Veeva Safety correctly triggers EUDAMED reporting rules rather than defaulting to medicinal product logic.
Key updates include:
- Multi-Agency Country Mapping: Users can now define a primary agency for medicinal products and a secondary Device Agency within a single Country record. This eliminates the one country, one agency limitation.
- Automated Registration Lookup: A new lookup field on the Product Registration object automatically pulls the correct Device Agency based on the registration country.
- Rule Triggering: By identifying the specific Device Agency at the registration level, Vault can now accurately execute EUDAMED-specific reporting rules for device Cases while maintaining standard EMA workflows for medicines.
NMPA Validation Criteria: Localize G.K.2.2 and H.1 ValidationsAuto-on26R1.3
To reduce technical rejections from the NMPA gateway and ensure regional data integrity, Veeva Safety introduces localized validation criteria for the G.k.2.2 and H.1 fields. These updates ensure that cases submitted to China meet the specific formatting and content requirements mandated by the NMPA, allowing safety teams to process China-bound reports with higher confidence and less manual troubleshooting.
Key capabilities include:
- Localized Reporter Country Code (G.k.2.2): Updates the validation logic for the reporter’s country code to strictly align with NMPA-specific E2B(R3) technical requirements.
- Regional Case Identifier Validation (H.1): Refines the Worldwide Case ID validation to ensure total conformance with the NMPA’s expected format for inbound Individual Case Safety Reports (ICSRs).
- Targeted Conformance Checks: Ensures these specific localized validations only trigger for NMPA-bound transmissions, preventing unnecessary false positive errors on reports destined for the FDA, EMA, or other global agencies.
- Reduced Gateway Failures: Automatically flags data in the UI that would otherwise be rejected by the China regulatory gateway, allowing case processors to correct errors prior to the transmission phase.
Validation Criteria: Inactivate NMPA.F.r.1-2Auto-on26R1.3
To perform more efficient safety assessments and reduce the administrative burden caused by redundant conformance errors, Veeva Safety inactivates specific validation criteria for NMPA submissions. This update ensures Vault no longer flags specific drug-related fields that are no longer strictly required or have been superseded by other regional reporting logic.
Specifically, Vault disables the validation check for Drug Characterization (NMPA.F.r.1) and removes the requirement for Medicinal Product Name as Reported by Primary Source (NMPA.F.r.2) for the NMPA region.
By removing these unnecessary checks, safety teams can move cases to the transmission phase more quickly without being blocked by non-essential validation failures or false positive noise. This change specifically targets the NMPA validation profile, ensuring that mandatory data elements required for successful gateway acceptance are prioritized while maintaining standard ICH or other regional rules for the same data fields elsewhere in the application.
Validation Criteria: Use EVCTMPROD Destinations for EMA ValidationsAuto-on26R1.3
Vault updates several EMA validation criteria rules to recognize EVCTMPROD as a valid submission destination, alongside the existing EVCTM and EVCTMTEST destinations.
Previously, affected validation criteria only evaluated rules against EVCTM and EVCTMTEST destinations, which could cause validations to behave unexpectedly for submissions routed through EVCTMPROD. This update ensures consistent validation behavior across all EMA destination types.
Interval Tabulations for DSURConfiguration26R1.3
With this release, Veeva Safety maintains compliance with new MHRA requirements by supporting the generation of interval tabulations. Users can now generate an Interval Tabulation of SAEs and an Interval Tabulation of SARs directly within their DSUR Listing. This ensures that clinical trial data is presented in the precise format expected by UK regulators without additional manual intervention.
Key updates include:
- Expanded Reporting Scope: The DSUR generation process now includes dedicated options for Interval Tabulations of both Serious Adverse Events (SAEs) and Serious Adverse Reactions (SARs).
- Regulatory Alignment: These additions specifically address the latest MHRA mandates, ensuring that submissions are complete and compliant upon generation.
Ignore Non-Active Studies During Reporting Rules EvaluationAuto-on26R1.3
Ensuring reporting accuracy requires that only relevant, active Study Registrations are considered during reporting rule evaluation. Previously, while Vault filtered out Inactive and Deprecated states, the introduction of new states from the ClinOps Connection could occasionally trigger unnecessary rule evaluations, leading to false positives in regulatory reporting workflows.
With this release, Veeva Safety streamlines rule processing by explicitly ignoring specific Study Registration lifecycle states. This update ensures that administrative or historical study records do not impact Vault’s automated evaluation of reporting obligations.
Allow Marketing to Investigational (same agency) Reporting ScenarioConfiguration26R1.3
Managing reporting obligations for complex cases involving both marketed and investigational products often requires intricate rule logic. Previously, reporting rules may not have fully accounted for cases where a single agency requires submissions to both premarket and postmarket centers for the same event. This often led to gaps in compliance or manual workarounds for unique study types, such as Individual Patient Use (IPU).
With this release, Veeva Safety introduces the ability to configure reporting scenarios for Individual Case Safety Reports (ICSRs) that bridge the gap between marketing and investigational designations within the same agency. This ensures that when a Case involves a mix of Product statuses, Vault correctly evaluates each according to rule priority and agency-specific requirements.
JSON Data IntegrityAuto-on26R1.3
Veeva Safety strengthens the reliability of JSON-based case imports by introducing stricter field filtering and validation logic during intake processing.
Unsupported field types including custom objects, system fields, non-Vault fields, and fields on the ignore list are now automatically excluded during import rather than triggering errors or causing unexpected behavior. This ensures that incoming JSON payloads are processed cleanly, even when they contain fields outside Vault’s supported data model.
Additionally, improved error handling provides clearer feedback when validation issues are detected during import, reducing troubleshooting time for integration teams.
Activate the Safety Monthly Maintenance JobAuto-on26R1.3
To improve performance, Vault is activating the Safety Monthly Maintenance job in all Vaults. This job runs on the first day of every month at 11 PM in local Vault time. If this job is already active in your Vault, no action is required and there will be no change in behavior.
Study Content Protection ReevaluationAuto-on26R1.3
To eliminate critical compliance risks related to accidental unblinding and maintain strict data protection boundaries, Veeva Safety now updates Study Content Protection values during reporting rule reevaluation. Previously, if a case’s safety parameters were modified after transmission generation, Vault did not update the Study Content Protection setting, potentially leaving sensitive clinical trial data visible on subsequent transmission documents.
Key capabilities include:
- Retroactive Transmission Synchronization: Updates the Evaluate Regulatory Obligations action to monitor and adjust Study Content Protection on Transmission records.
- Dynamic Masking Overrides: Automatically updates masking for existing Transmissions if a newly triggered reporting rule mandates a different protection layout, such as transitioning from an unblinded reporting family to a masked reporting family for the same destination.
- Removal of Manual Workarounds: Restructures data copying mechanisms to synchronize fields without requiring case triage teams to manually override or delete and recreate partner distribution records when case unblinding contexts evolve.
- Maintained Study Blind Integrity: Guarantees that partner documents and MedWatch or E2B forms accurately align with the latest clinical trial security requirements right up until the point of final transmission.
FDA Summary Type Reporting Rules AutomationAuto-on26R1.3
To streamline the handling of specialized submissions, Veeva Safety introduces targeted reporting rule execution logic for Cases that specify an FDA Summary Type. Previously, such Cases triggered the full spectrum of global reporting and distribution rules, often resulting in redundant records that required manual cleanup. With this update, Vault restricts rule evaluation and Early Notifications for these Cases exclusively to the FDA Agency ruleset while suppressing general distribution rules. This granular approach ensures regulatory precision for FDA summaries without impacting One Last Time (OLT) or Nullification workflows, helping to eliminate reporting noise and reducing the risk of erroneous multi-agency submissions.
Aggregate Support of New Study TypesAuto-on26R1.3
Vault now expands aggregate reporting to recognize three new study types introduced under ICH E2D guidelines, ensuring cases tied to these programs are correctly classified and included in periodic safety reports.
Previously, cases associated with newer post-marketing evaluation methods could be flagged as invalid or miscategorized during aggregate generation. This update adds native support for the following study types across PBRERs, PSURs, and PADERs in Veeva Safety:
- Patient Support Programs (E2B Code 4;
patient_support_program__v) - Market Research Programs (E2B Code 5;
market_research_program__v) - Organized Data Collection Systems (E2B Code 6;
organized_data_collection_system__v)
Cases associated with these study types are processed as non-interventional or solicited (other) reports, depending on the requirements of the specific report template.
Safety & SafetyDocs
Safety Deletion LogsConfiguration26R1.3
To strengthen GxP compliance and safeguard critical system files against accidental loss, Veeva SafetyDocs introduces Safety Deletion Logs. This feature allows organizations to mandate a formal, auditable justification whenever documents (such as PVAs, PSMFs, or RMPs) are deleted from Vault, mirroring advanced compliance controls found in Clinical Operations. By creating a structured record of removals and introducing a temporary preservation buffer, safety teams can ensure thorough regulatory audit readiness while retaining the ability to recover documents mistakenly marked for deletion.
Key capabilities include:
- Enforced Deletion Justification: Replaces standard document deletion with a mandatory Delete Document with Reason user action, requiring users to select a predefined justification and provide an optional descriptive comment before a document deletion request can proceed.
- Automated Audit Record Creation: Seamlessly logs all request details in a Safety Deletion Log record, capturing essential document metadata for permanent compliance retention.
- Flexible Deletion Cancellation: Within 30 days of creating a document deletion request, authorized users can execute a Cancel Deletion Request action, which instantly moves the Safety Deletion Log to the cancelled state and logs a mandatory cancellation reason for the tracking archive.
- Automated Daily Cleanup Engine: Every 24 hours, Vault permanently purges documents marked for deletion that have surpassed their 30-day retention window, while systematically flagging blockages (such as legal holds, active workflows, or object references) and notifying the requester.
Safety, Safety Signal, Safety Workbench & SafetyDocs
26R2 Safety Suite StandardizationConfiguration26R1.3
To further unify our Safety ecosystem, the 26R2 Safety Suite Standardization introduces a comprehensive set of standard components across Safety, Safety Signal, Safety Workbench, and SafetyDocs. This foundational update aligns the data model across all applications, ensuring that shared entities such as Products, Studies, and Organizations behave consistently throughout the safety lifecycle. By adopting these standard configurations, organizations can more easily implement industry best practices, reduce manual data mapping, and accelerate the cross-application workflows essential for signal management and aggregate reporting.
Key capabilities include:
- Unified Data Model (CDM): Synchronizes core objects across the Safety suite, ensuring that a Product or Study defined in Safety is immediately and consistently available for detection in Signal and reporting in Workbench.
- Standardized Lifecycles & Workflows: Introduces preconfigured lifecycles for Case investigations and aggregate report authoring, while placing Signal Individual Case Review capabilities directly behind the SafetyDocs manifest to align cross-application Safety workflows.
- Cross-App Terminology Alignment: Updates standard picklists and field definitions across all four applications to ensure a single version of truth for critical data points like Seriousness, Case Type, and Event Terminology.
- Ready-to-Use Business Logic: Deploys standard Action Triggers and Validation Rules that enforce best practices, such as ensuring mandatory document links are present before a signal is promoted to an investigation.
- Suite-Wide Reporting Readiness: Standardizes the underlying data structures in Safety Workbench to support pre-built, suite-wide dashboards that provide real-time oversight across intake, processing, and signal management.
Vault AI for Safety
Narrative Agent: Custom InstructionsConfiguration26R1.3
To provide greater flexibility in narrative generation and ensure alignment with organizational writing standards, Veeva Safety introduces several enhancements to the Narrative Agent. These updates build upon the foundation released in 26R1, allowing safety teams to further customize AI-driven outputs and maintain a detailed audit trail of AI activity.
Key capabilities include:
- Supplemental Instructions: Admins can now define specific instructions such as “use a chronological order” or “apply company voice” to the standard Rewrite and Proofread actions to fine-tune the agent’s output to meet specific internal requirements.
- Configurable Custom Actions: Admin can define entirely new, custom actions for the Narrative Agent, allowing for the creation of unique prompts that cater to specialized narrative styles or regional reporting needs.
- Automated & Manual Flexibility: Vault supports custom actions in both manual user-triggered scenarios and as part of automated Narrative Outlines, ensuring consistent AI behavior throughout the case processing lifecycle.
Safety Workbench
Advanced Logic for Report Set FiltersAuto-on26R1.2
This feature ensures consistent Workbench Report data analysis and significantly reduces manual effort when managing large report volumes. Veeva Safety Workbench now supports advanced logic for additional Workbench Report Set filters, allowing users to apply complex, ad-hoc filtering criteria across multiple reports simultaneously.
Learn More
- Admin Help: Configure Workbench Report Sets
- User Help:
Dashboard Analysis with View ParametersAuto-on26R1.2
This feature provides the flexibility to focus on specific data subsets in Workbench Dashboards without restricted filtering. Veeva Safety Workbench now supports Workbench View Parameters in dashboards, allowing users to perform complex, calculation-heavy analyses, such as defining multiple periods for disproportionality or setting data cutoff dates for expectedness, directly within a dashboard. Users can dynamically drive SQL joins and layout logic across all dashboard components.
Although this feature is Auto-on, some components may require additional configuration.
Learn More
- Admin Help: Configure Safety Views in Safety Workbench
- User Help: Use Workbench Dashboard Filters
Sticky Dashboard TotalsAuto-on26R1.2
This feature enhances data analysis by anchoring total rows to the bottom of Workbench Dashboard Components, ensuring key metrics remain visible while users scroll through detailed records.
Although this feature is Auto-on, some components may require additional configuration.
Learn More
- Admin Help: Configure Safety Views in Safety Workbench
- User Help: Manage Workbench Dashboards
Dashboards: Embedded User GuidanceAuto-on26R1.2
This feature improves dashboard usability and provides essential guidance by allowing Admins to add static text components to Workbench Dashboards. This enables the display of persistent documentation, such as “Tips and Reminders”, or welcome pages directly alongside data visualizations to provide critical business context.
Learn More
- User Help: Manage Workbench Dashboards
Save Report Sets to the LibraryConfiguration26R1.3
This feature improves data compliance and reduces manual effort by eliminating local file downloads. Veeva Safety Workbench now enables users to generate a combined Excel workbook for an entire Workbench Report Set and upload it directly to the Vault Library with a single click.
Custom Report Naming from DashboardsAuto-on26R1.3
This feature improves dashboard usability and reporting efficiency when generating a Workbench Report from a Workbench Dashboard Component. Vault allows users to customize report names during generation by prompting them to edit the autopopulated default name before running and creating the report.
Dashboards: Long Text DialogsAuto-on26R1.3
This feature improves Workbench Dashboard usability when analyzing long text fields. For long text fields exceeding 1,500 characters, such as Narrative Previews or Case comments, Veeva Safety Workbench now offers a Show More action to view the complete text in a dialog.
MedDRA Query Scope FilteringAuto-on26R1.3
This feature simplifies safety reporting by delivering standard, out-of-the-box MedDRA scope filtering. Veeva Safety Workbench now supports selecting broad or narrow variations for standardised MedDRA Queries (SMQs) and custom MedDRA Queries (CMQs) across Workbench Reports, Workbench Views, and Workbench Dashboards, providing pharmacovigilance teams with standard control over query precision.
Workbench PMDA Post-Market Aggregate ReportsAuto-on26R1.3
With this release, Veeva Safety Workbench introduces the ability to generate compliant periodic safety reports in accordance with PMDA guidelines, including:
- J-PSR Form 3: Cumulative Tabulation
- J-PSR Form 4: Line Listing
- J-NUPR Line Listing
As well as the following additional components:
- Open Case Listing
- Case Series Listing
Admins can configure these reports to run individually or together within a unified report set. User locale must be set to Japan to display localized date formats.
Although this feature is Auto-on, some components require additional configuration.
Workbench Pregnancy Annex 3 ReportsAuto-on26R1.3
With this release, users can generate the following EMA Annex 3 pregnancy reports in Veeva Safety Workbench, in accordance with EMA regulations:
- Maternal Outcome Tabulation
- Paternal Outcome Tabulation
- Case Line Listing
- Event Line Listing
These reports automate complex case grouping into regulatory layouts, enhancing submission readiness and analysis of pregnancy cases.
Latest Case Version SupportConfiguration26R1.3
With this release, Admins can configure safety views to return the latest Case versions in Workbench Reports, Workbench Report Sets, and Workbench Dashboards. Admins can also configure safety views to allow users to choose whether versioning logic is applied. In addition, this feature introduces the ability for Admins to hide specific Workbench View Columns from Workbench Report, Workbench Report Set, and Workbench Dashboard filters.
Safety Signal
MedDRA Query Analysis for FAERS & VAERSConfiguration26R1.2
Veeva Safety Signal now supports running Signal Calculations at the MedDRA Query level using FAERS and VAERS data. This feature enables aggregated analysis across external datasets to improve signal detection accuracy.
Learn More
- Admin Help: Configure Signal Calculation Objects
- User Help: Run Signal Calculations
EVDAS Data Requests & Automated ProcessingConfiguration26R1.3
This feature eliminates error-prone, manual EVDAS data loading and the need for external tools for electronic Reaction Monitoring Report (eRMR) and line listing linking. Veeva Safety Signal now accelerates EVDAS data importing by automating the ingestion, cleaning, and MedDRA coding of eRMR files and line listings. While Admins can import EVDAS data for routine signal detection, users can also submit requests for EVDAS data outside of regular surveillance periods for ad-hoc signal evaluation. Vault can then link corresponding eRMR and Line Listing records together and automatically flag entries requiring manual intervention.
Signal Calculation Configuration SummaryAuto-on26R1.3
Veeva Safety Signal now generates a downloadable summary of Signal Calculation data to ensure data integrity and provide an overview of the calculations that ran. The summary file provides a snapshot of the filters and settings used to determine specific signal results.
Enhanced Logic Operators for Signal AlertsConfiguration26R1.3
Veeva Safety Signal now allows Admins to configure Signal Alert Rule Sets so that signal alerts only trigger when specific combinations of complex Signal Alert Rule criteria are met. This feature improves signal detection precision by allowing for more sophisticated signal monitoring configurations.
Safety Signal & SafetyDocs
Signal Review SchedulingConfiguration26R1.2
With this release, Veeva Safety Signal and SafetyDocs automate the scheduling of routine signal surveillance reviews by introducing automated creation of Signal Reporting Periods based on a predefined schedule for a Signal Product Profile. This feature eliminates the need for manual tracking in external tools like spreadsheets.
Learn More
- Enablement: Enable Signal Review Scheduling
- Admin Help:
Signal Detection Plans: Data ModelConfiguration26R1.3
To eliminate manual tracking and ensure compliance, Veeva SafetyDocs centralizes and versions product safety monitoring strategies. This comprehensive data model structures Signal Detection Plans, risk-based product tiers, and Safety Topics, directly linking high-level monitoring activities to active execution profiles, such as Signal Product Profiles, Signal Alert Rules, and Signal Data Sources. Automated plan execution is currently unsupported.
Weekly Signal Review SchedulingConfiguration26R1.3
To streamline high-frequency safety surveillance, Veeva Safety Signal now supports a weekly scheduling option for Signal Product Profiles. This allows users to automatically create weekly reporting periods and execute scheduled Signal Calculations without encountering validation errors.
SafetyDocs
PVA Effective Date AutomationConfiguration26R1.2
Managing the timeline of Pharmacovigilance Agreements (PVAs) requires coordination between administrative completion and regulatory activation. Previously, activating a PV Agreement was often a manual task, which created a risk of non-compliance if the status wasn’t updated exactly when the agreement became effective.
With this release, Veeva SafetyDocs automates lifecycle management for PV Agreements based on future-dated effectiveness, ensuring seamless transitions and reducing the administrative burden of manual status updates. PVA Managers can now finalize records in advance, allowing Vault to transition PV Agreements to the Effective state on the designated effective date. When a PV Agreement enters the Effective state, Vault additionally moves all of its associated Approved-state documents to the Effective state.
Learn More
- Enablement: Enable PVA Management
- User Help: Manage Pharmacovigilance Agreements
User License Warnings for SafetyDocs DocumentsAuto-on26R1.2
Maintaining strict adherence to Veeva licensing agreements is essential for organizational compliance and audit readiness. With this release, Veeva SafetyDocs identifies user license violations for Safety application users who might inadvertently access SafetyDocs features or documents without the appropriate specialized license, ensuring that access to sensitive regulatory content is aligned with user entitlements and providing immediate feedback when a license violation occurs.
Prevent Automatic PSMF Logbook GenerationConfiguration26R1.2
Managing PSMF revisions often involves minor changes. Previously, Vault automatically generated a PSMF Logbook Entry any time a new document was created, forcing users into a rigid workflow where even minor technical adjustments such as fixing a broken hyperlink or addressing drafts created in error required a formal entry. Secondary authors often struggled to update the auto-generated PSMF Logbook Entries with their own details, leading to data gaps or blank records in the document history.
With this release, Veeva SafetyDocs allows you to prevent the automatic creation of PSMF Logbook Entries when a PSMF document is created or up-versioned, providing greater flexibility to manage minor updates and accidental draft creations without cluttering the audit trail.
Learn More
- Enablement: Enable PSMF Automatic Logbook Generation
- User Help: Manage PSMF Binders and Logbooks
Generate Draft PSMF PDFConfiguration26R1.3
Maintaining a Pharmacovigilance System Master File (PSMF) requires frequent oversight and internal QC checks. Previously, users could only generate a comprehensive PDF version of the PSMF Binder after all related documents were finalized and the PSMF was electronically signed. This limitation often delayed pre-submission reviews and internal QC tasks, as teams could not visualize the consolidated binder in its final format during the authoring phase.
With this release, Veeva SafetyDocs introduces the Generate Draft PSMF PDF action, allowing users to produce a draft version of their PSMF Binders at any point during the authoring process. This feature provides immediate visibility into the formatted binder, facilitating earlier quality checks and more agile collaboration.
PSMF Request ManagementConfiguration26R1.3
Maintaining the Pharmacovigilance System Master File (PSMF) often involves responding to complex, time-sensitive inquiries from various requesters, such as Health Authorities (HA) or partners. Previously, tracking these requests was often handled through disconnected spreadsheets or manual trackers, making it difficult for PSMF Coordinators to maintain oversight of global compliance due to visibility gaps regarding due dates, team assignments, and the specific PSMF versions referenced in each request.
With this release, Veeva SafetyDocs introduces a dedicated mechanism for managing PSMF requests, centralizing all regulatory interactions and ensuring that teams can respond and meet strict compliance timelines.
Key updates include:
- A PSMF Request object that allows PSMF Coordinators to manage requests from end to end. A PSMF Request can be categorized by jurisdiction, urgency, and type, and is populated with specific request participants to ensure precise prioritization, due date tracking, and effective compliance monitoring.
- A dedicated workflow provides full visibility into PSMF Request lifecycles, allowing teams to track requests from initial receipt and response planning to document submission.
- The ability to map specific PSMF documents to a PSMF Request facilitates collaborative response planning and ensures a consistent regulatory narrative.
- The ability to generate ad-hoc PSMF Periodic Reviews as required from a PSMF Request streamlines the remediation process and ensures the PSMF is updated promptly based on feedback.
IMIC Literature IntakeConfiguration26R1.3
Efficiently managing global literature requires the ability to ingest data from diverse regional sources without manual data entry. Previously, users were limited to RIS or JAPIC formats to bring external citations into Vault.
With this release, Veeva SafetyDocs introduces the ability to import literature references from International Medical Information Center (IMIC) formatted files directly into Literature Articles, allowing for further processing and conversion to Inbox Items, and streamlining the transition from search results to actionable safety data. Users can now upload IMIC files, similar to the existing RIS and JAPIC import processes. During import, Vault maps data from structured fields in the IMIC file to Literature Article fields.This feature minimizes the need for manual transcription, reducing the risk of data entry errors and accelerating the screening process.
PVA Periodic ReviewConfiguration26R1.3
Maintaining oversight of Pharmacovigilance Agreements (PVAs) is critical for ensuring that safety data exchange obligations remain current and compliant. Previously, tracking the review cycles for these agreements often relied on manual monitoring or external trackers, which increased the risk of missing critical update windows as regulations or partner roles evolved.
With this release, Veeva SafetyDocs introduces periodic review for Pharmacovigilance Agreement documents, mirroring the established PSMF periodic review logic to automate the scheduling and task management of agreement evaluations. Vault automatically identifies PV Agreements with documents due for review and creates PVA Periodic Review tasks for assigned reviewers, eliminating the need for manual oversight. If a review determines that an amendment is necessary, Vault up-versions the PVA document, automatically adjusting the next scheduled review date upon completion. This feature ensures that every PV Agreement is systematically reviewed, updated, and synchronized with its corresponding documentation.
Specify Reply-to Value for PVA Distribution EmailsConfiguration26R1.3
Managing partner communications is important, especially when ensuring that regulatory and safety information are routed to the correct subject matter expert. Previously, outbound distribution emails sent via PV Agreements required that the reply-to value match the email address of the Sponsor Contact defined on the PVA Obligation. This automatic reply-to assignment prevented users from specifying a different preferred correspondent or shared inbox as needed for redundancy and auditability.
With this release, Veeva Safety introduces the Outbound Reply-To Person field to the PV Agreement object. This update allows organizations to define a specific contact or shared inbox at the agreement level, ensuring that any replies to automated PVA Document for Distribution emails are sent directly to the appropriate stakeholders. This streamlines partner correspondence and reduces the administrative burden of manual email forwarding.
Prevent Lifecycle State Change When Error Occurs While Generating PSMF PDFsAuto-on26R1.3
To ensure data alignment and prevent compliance gaps within SafetyDocs content management, Veeva SafetyDocs introduces enhanced error-handling logic when generating PSMF PDFs. By blocking PSMF Binders from changing state if an error occurs, Vault eliminates the operational hazard of PSMF Binders advancing to a new state while possessing broken, unmerged, or missing PDF attachments.
Specify Start Date for Multi-Agreement ActivitiesConfiguration26R1.3
In real-world partner obligation tracking, pharmacovigilance teams are frequently not the first to learn about a reportable milestone, occasionally discovering safety events days after they originally occurred. To provide better operational flexibility and prevent artificial compliance reporting, Veeva SafetyDocs introduces the ability to define specific start dates–even if they occurred in the past–for PVA Multi-Agreement Activities, which Vault uses to calculate due dates for PVA Activities generated using the Generate Related Activities action.
Signal Individual Case Review for SafetyDocsConfiguration26R1.3
Vault now enables users with Safety and SafetyDocs licenses to perform Signal Individual Case Reviews (ICRs). Signal ICRs are automatically created based on routing rules and linked to specific Case Products. Signal reviewers can assess individual Cases without affecting ongoing Case processing and determine whether a Safety Investigation is needed.
QualityOne
QualityOne
Teams: Advanced User SearchAuto-on26R1.3
When selecting team members from the Team section on a Team-enabled object record, users can now click the binoculars icon to open an advanced search dialog to perform a text search, filter on User object fields, and select a team member to assign.
Teams: User Name HovercardAuto-on26R1.3
This feature lets users hover over a team member’s name in the Team section of an object record to view a hovercard containing additional information about each user. Admins can configure which fields are displayed in hovercards by selecting the Display in default lists and hovercards option for fields on the User object.
Teams Bulk Management: Lifecycle State Filtering & Change Summary EnhancementsAuto-on26R1.3
With this release, the Teams Bulk Management interface now displays a filter icon on the Lifecycle States column of the record results grid, allowing users to filter records by lifecycle state directly from the column. Additionally, when previewing the changes to be applied in the Summary section, Vault now displays the Name field values for all records to be updated. Previously, Vault displayed the value of the ID field.
HACCP (QualityOne)
HACCP Flow Diagram: Display Risk Category Field as Clickable LinkAuto-on26R1.3
In the Risk Assessment section of the Information panel, the Risk Category field now displays as a clickable link. Users can click the link to open the Risk Category record in a small browser window.
HACCP Flow Diagram: Update to Label of Hazard Significance Filtering OptionAuto-on26R1.3
Currently, users can filter Process Hazard Analysis records in the Hazard Analyses section of the Information panel according to the value of the Is Significant Hazard? field. With this release, the label of the “Insignificant” filtering option is updated to “Non-significant”.
HACCP Flow Diagram: Intermediate State for ConnectionsAuto-on26R1.3
When creating, editing, or deleting a connection between two steps on the HACCP Flow Diagram, the connection’s line now changes to gray and users cannot make further edits to that connection while Vault saves the changes.
RegulatoryOne
Registration & Dossier Management
Registration Objective LimitAuto-on26R1.2
With this feature, the limit of Registration Objectives for a given Registration increases from 100 to 1,000.
Scheduled Job for Create Binder ActionConfiguration26R1.3
This feature ensures successful execution of the Create Binder entry action when invoked from a scheduled job by setting the Owner value of the Requirement as the entry action initiating user.
Veeva Claims
Veeva Claims
Clone Project: Sections OrderingAuto-on26R1.2
This feature updates the display order of records listed in related object sections displayed in the dialog after running the Clone Project action from alphabetical order to the logical order configured on the Project object layout.