Pre-Release 日: 2024 年 3 月 25 日 | リリース日: 2024 年 4 月 12 日および 2024 年 4 月 19 日
Safety、QualityOne Client Application、RegulatoryOne、Veeva Claims の各アプリケーションのリリース日はそれぞれ異なることがあります。
Vault 24R1 のご紹介です。以下の新機能をご覧ください。新機能の有効化に関する情報については、24R1 Release Impact Assessment をご覧ください。開発者向け機能 (API、VQL など) については、Developer Portal をご覧ください。
Platform
以下の Release Notes に加えて、Vault Platform Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモを提供しています。
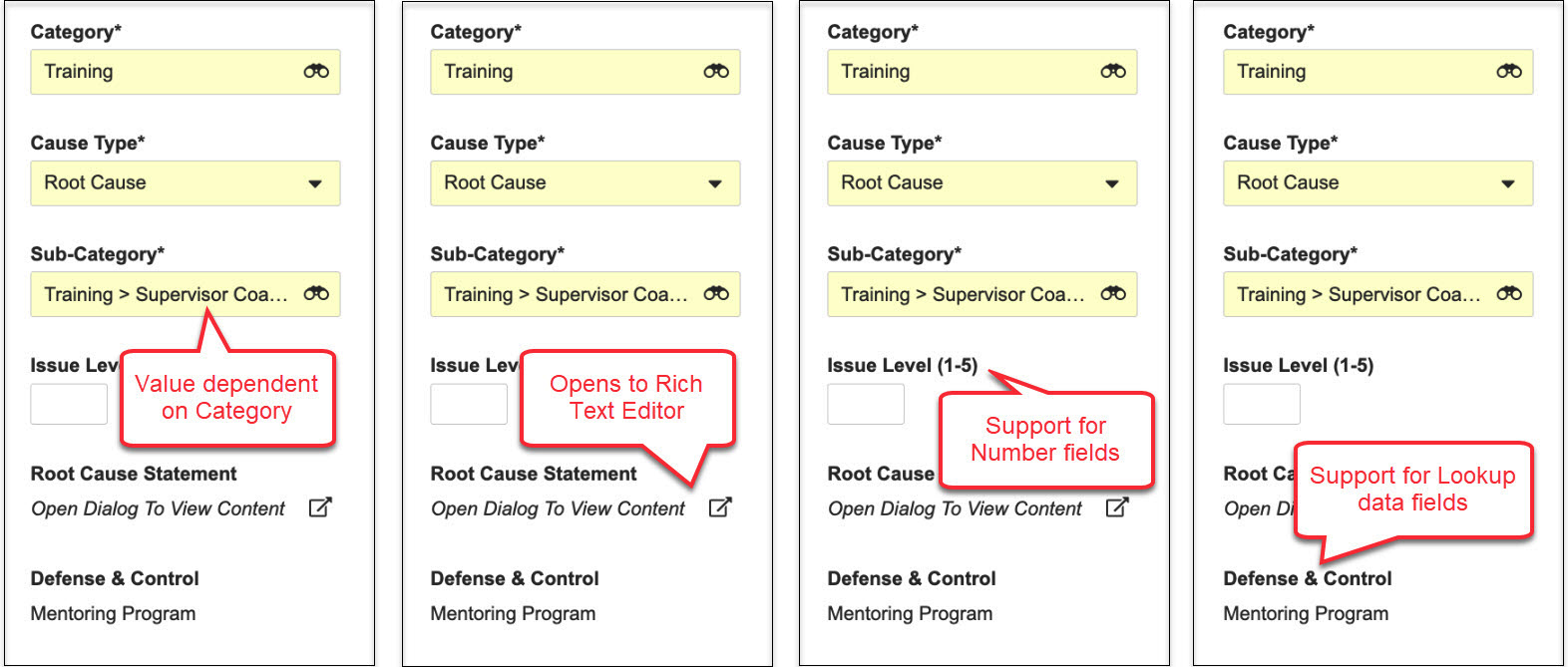
Action Layouts
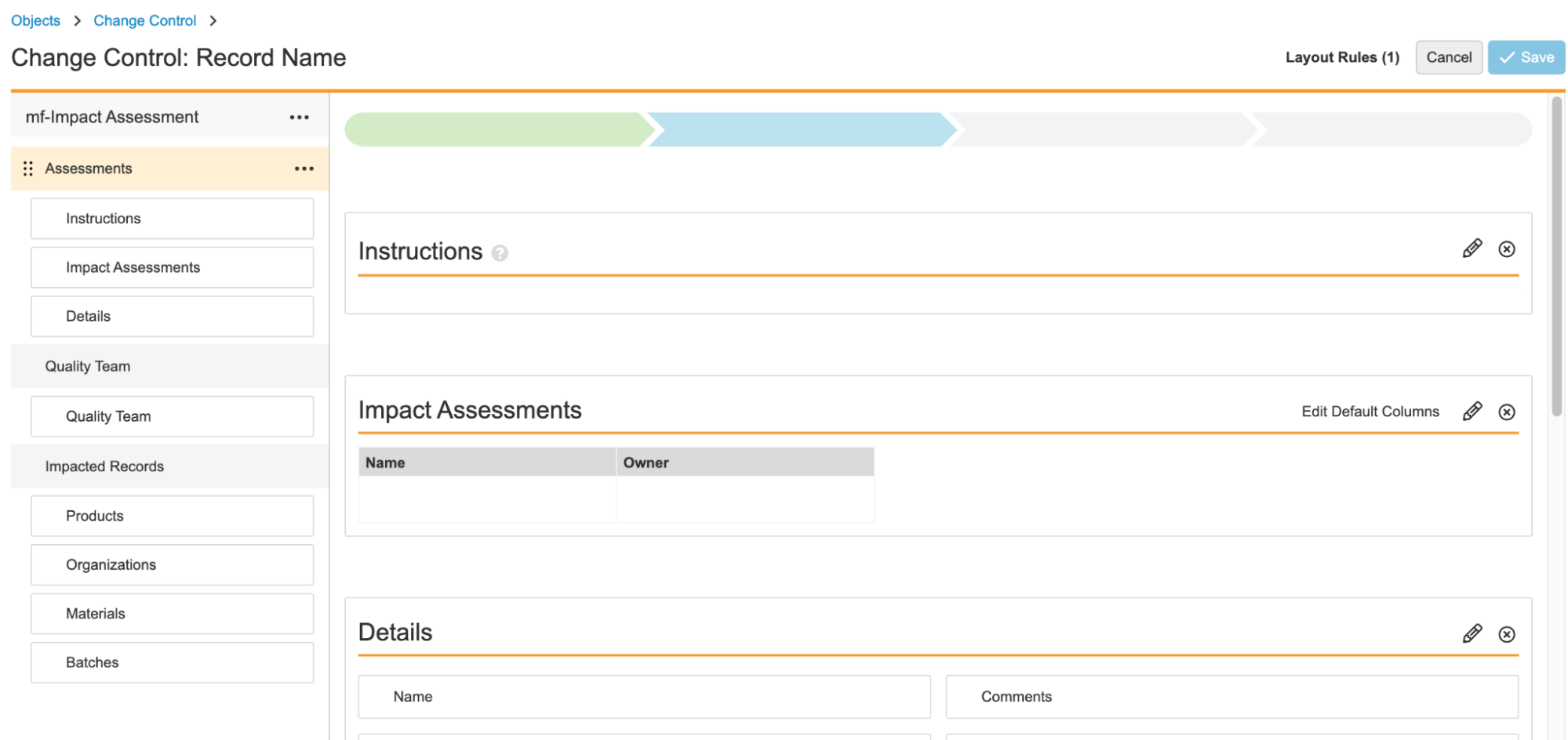
24R1では、アクションレイアウトを導入し、レコード詳細ページのユーザエクスペリエンスをカスタマイズするための新しくパワフルな手段を提供します。アクションレイアウトは、ページ、複数レイアウト、レイアウトプロファイルの 3 つの新機能と、ビジュアルスタイリング、レイアウトエディター、レイアウト規則における多くの機能強化によって支えられています。管理者は、さまざまなレイアウトを作成し、特定の使用事例および/または特定のユーザーを対象に、さまざまなユーザーが適切なタイミングで適量の情報を使用して、より合理化されたレイアウトでレコードを表示できるようになりました。この機能強化の詳細は以下で説明されています。これにより、お客様はさまざまなユーザー向けに、より整然として、文脈に沿い、焦点が絞られたレイアウトを作成できます。
Action Layouts: Layout Pages
現在、[レコード詳細ページ] レイアウトにはセクションのみが含まれています。このリリースで、管理者はレイアウトに最大 10 個別ページを設定して、セクションをより適切に整理できるようになりました。ページは、ユーザーが簡単に移動してタスクに必要な適切なデータに集中できるように、関連するセクションをグループ化するために使用されます。デフォルトでは、既存のすべてのレイアウトはセクションのみで変更されませんが、左側のナビゲーションのユーザーインターフェイスが若干すっきりと表示され、メニューを最小化して全画面表示できるようにするオプションがあります。
さらに、レイアウトエディタが拡張され、以下が可能になりました:
- 新しいページの作成
- セクションをドラッグ&ドロップして並べ替える
- ページとセクションの挿入
- カスタムヘルプコンテンツをセクション内とスタンドアロンセクションの両方に追加する
- レイアウト規則エディタの更新
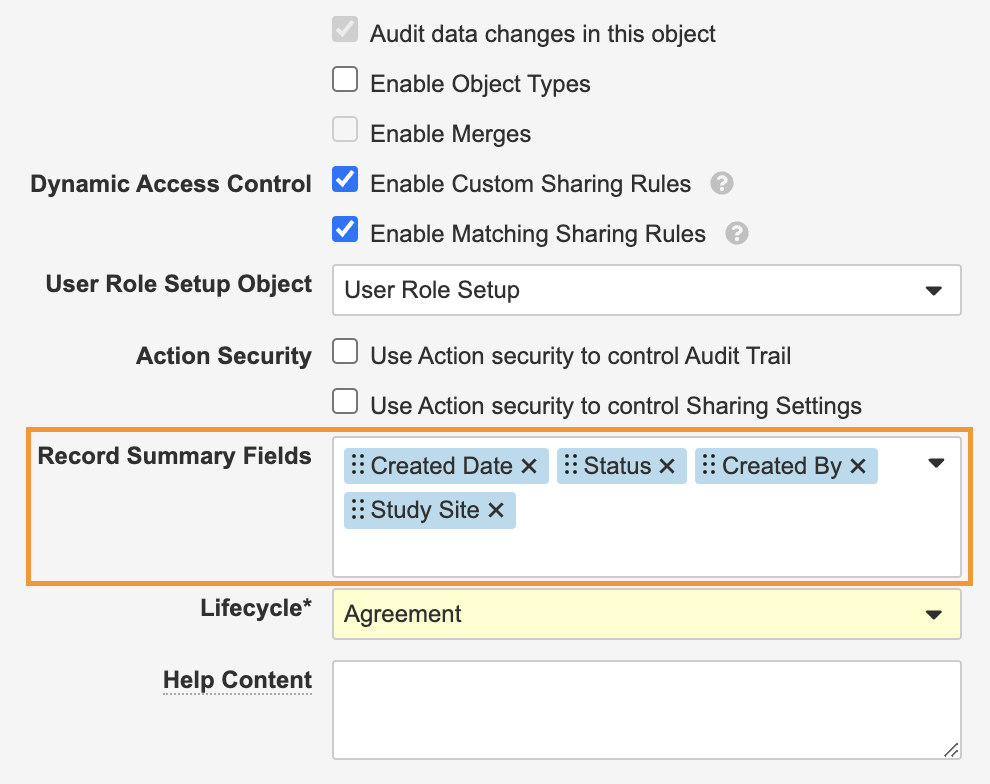
Action Layouts: Record Summary Fields
アクションレイアウトを使用すると、管理者はオブジェクトまたはオブジェクトタイプごとに最大 6 つのフィールドを設定して、常にレコードページの上部にレコードサマリーフィールドとして表示できます。これにより、ユーザがレコードを操作している間、重要な情報を常に表示できます。
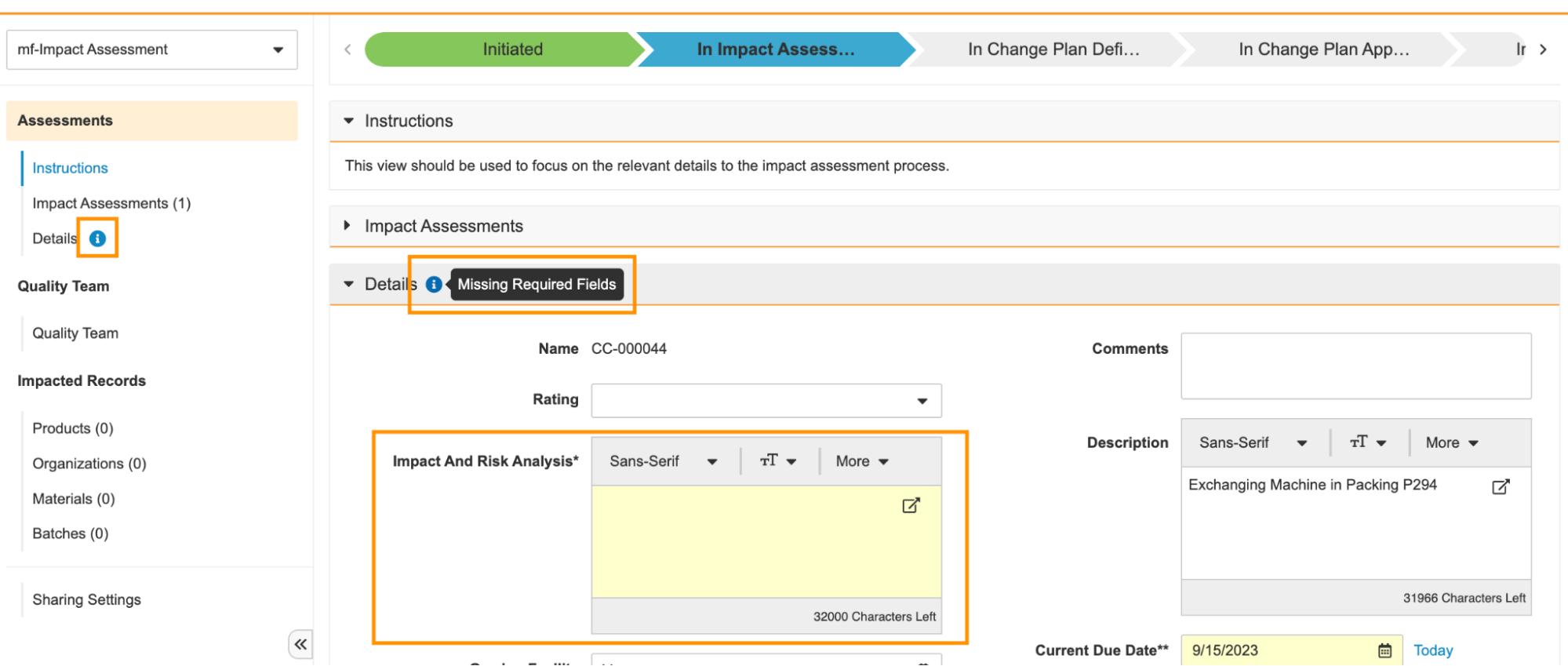
Help Section/Control
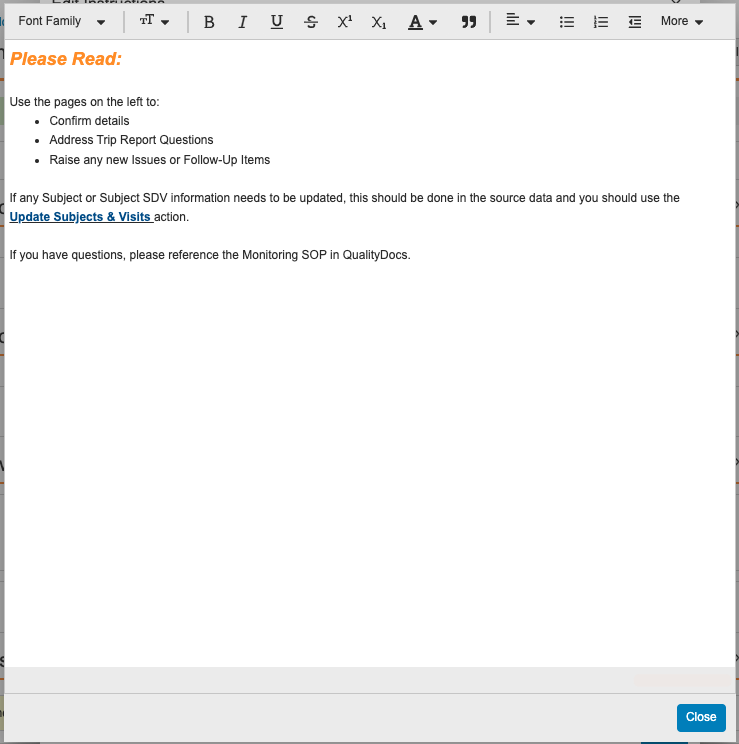
Action Layouts を使用すると、管理者は Help Sections と Help Controls を設定して、特定のセクション内にヘルプテキストを追加できます。これにより、ユーザは、所定の時間に所定のレコードに対してどのような処理が期待されるかをガイドする詳細なコンテキスト情報を受け取ることができます。
この機能により、ハイパーリンクや画像 URL のサポートなど、リッチテキストエディタを利用できるように機能強化され、管理者が Help Sections と Help Controls の書式設定を定義する際の柔軟性と使いやすさが向上します。
詳細に関しては、ヘルプセクションとヘルプコントロールをご覧ください。
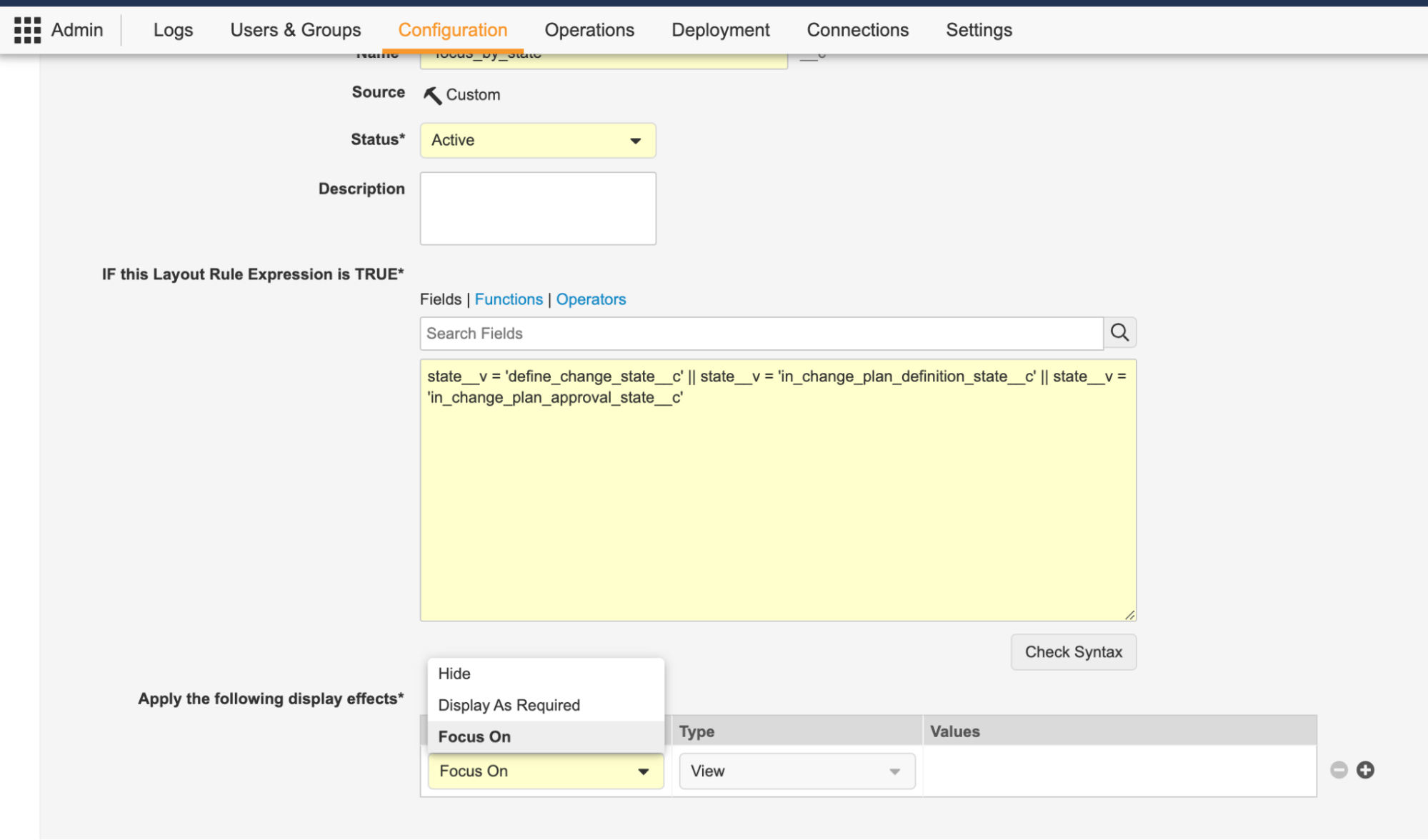
Action Layouts: Layout Rules Enhancement
これらのレイアウト変更の柔軟性をさらに高めるために、レイアウト規則も強化されました。24R1 以前のバージョンでは、管理者はレイアウト規則を使用して、レコードのライフサイクル状態などのビジネスロジック条件に基づいて、セクションまたはフィールドを表示または非表示にすることができました。
レイアウトにページが導入された 24R1 リリースでは、レイアウト規則により、レイアウト内のページを表示または非表示にすることもできるようになりました。さらに、ユーザにはレコードを表示するために複数のレイアウトが割り当てられている可能性があるため、管理者は、Focus On と呼ばれる新しいエフェクトで、最初に表示する特定のレイアウトを選択する基準を定義できます。たとえば、QMS の変更コントロールが In Implementation 状態の場合、そのレコードがプロセスのどこにあるかに基づいて、デフォルトで異なるレイアウトを表示することが合理的である場合があります。
新しい Display as Required エフェクトを使用すると、レイアウト規則式に基づいてフィールドが必須フィールドとして表示されます。これにより、フィールドが適切な状況で必須として表示されるようになり、ユーザが入力基準エラーに遭遇する可能性を減らすことができます。
新しい Display as Read Only エフェクトを使用すると、レイアウト規則式に基づいてフィールドが読み取り専用として表示されます。たとえば、レイアウト規則は、Draft などのライフサイクル状態に基づいてフィールドを読み取り専用として表示することができます。この機能強化により、フィールドの基本的なセキュリティは変更されませんが、特定のレイアウトでユーザに編集不可能なフィールドを表示する柔軟性が得られます。
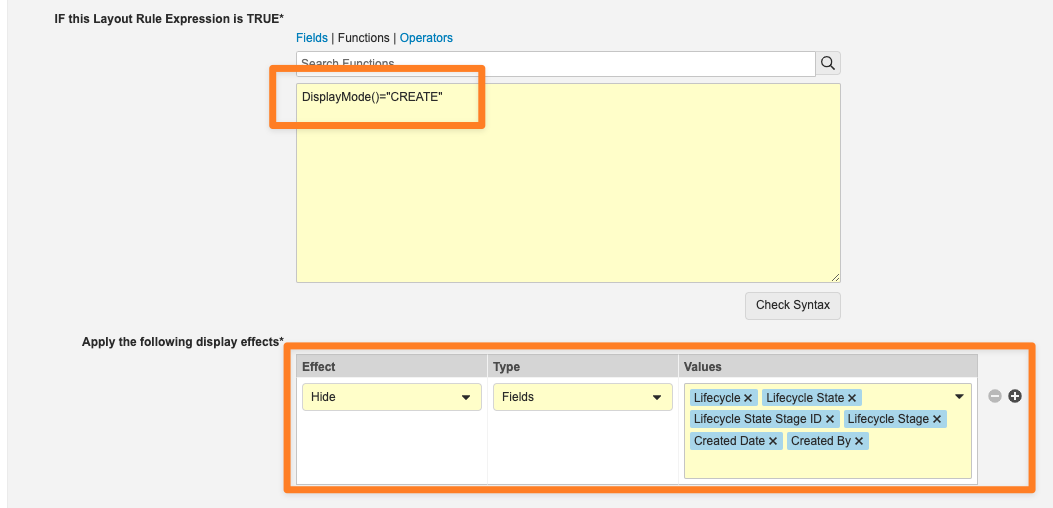
最後に、新しい DisplayMode() 関数を使用すると、管理者はレイアウト規則を設定して、ユーザがオブジェクトレコードをどのように操作しているかに基づいてエフェクトを適用できます。表示モードには、VIEW、CREATE、EDIT、COPY の 4 種類があります。
たとえば、この関数は、ユーザがレコードを作成するときに特定のシステム管理フィールドを非表示にするために使用されます。
これらのレイアウト規則の機能強化により、アクションレイアウトの変更による柔軟性がさらに向上し、ユーザーが適切なタイミングで適切な情報に集中できるようになります。
アクションレイアウト: レイアウトプロファイル
管理者は、オブジェクトごとおよびオブジェクトタイプごとに最大20の異なるレイアウトを設定できるようになりました。これにより、すべてのユースケースに適合する1つのレイアウトではなく、特定のユースケース用に構築された複数のレイアウトをサポートするはるかに高度な設定が可能になります。リリース後も、ユーザーには以前と同じレイアウトが引き続き表示されます。レイアウトプロファイルを活用して、ユーザーに異なるレイアウトを割り当てるために、管理者は新しいレイアウトを作成し、レイアウトプロファイルにそれらを関連付ける必要があります。管理者はレイアウトプロファイルを使用して、1 人のユーザーに 1 つのレイアウトを割り当てたり、1 人のユーザーに複数のレイアウトを割り当ててレコードを表示したりできます。
これにより、レイアウトページでサポートされる追加フォーカスを、ユーザーとその役割に基づいて柔軟に設定することができます。たとえば、QMSでは、QA チームのメンバーは、それぞれの役割に合わせて調整されたレイアウトによって、作業をより効率的に遂行できる可能性があり、一方内容領域専門家は、まったく異なるレイアウトの恩恵を受けることができます。
Action Layouts: Additional Improvements
24R1 では、アクションレイアウト機能の追加の機能強化は次のとおりです。
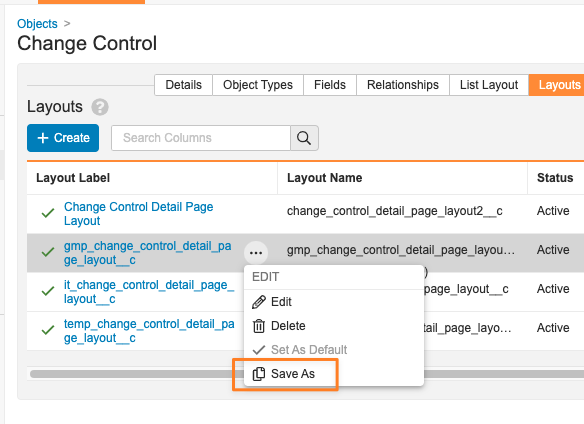
Save As (既存のレイアウトから新しいレイアウトを作成):
レイアウトを設定する際、管理者は既存のレイアウトを選択し、新しいレイアウトとして保存できるようになります。この機能強化により、レイアウトの作成と管理のプロセスが合理化され、管理者が標準レイアウトを利用できるようになります。
標準レイアウト:
アクションレイアウトの導入を可能にするために、Vault はアプリケーションチームによって展開される標準レイアウトをサポートするようになりました。標準レイアウトは読み取り専用です。ユーザは代わりに新しいカスタムレイアウトとして保存できます。標準レイアウトは、カスタムレイアウトの制限にはカウントされません。
カスタムレイアウト制限の増加:
オブジェクトタイプごとのカスタムレイアウトの最大数が 20 から 50 に増加しました。
Layout Editor - 複数選択のドラッグアンドドロップ:
Command (Mac の場合) または Control (Windows の場合) を押したままにすることで、管理者はレイアウトエディタ内で複数のセクションを選択し、それらをドラッグしてセクションの順序を変更できます。これにより、特定のセクションをまとめておく必要がある場合に、管理者はより簡単に調整できるようになります。
Layout Editor - レイアウト規則のフィルタリング
フィールドやセクションなどのレイアウト規則の影響を受ける要素は、fx アイコンで示されます。これらのアイコンがクリックできるようになり、クリックすることで、その要素のみに影響を与えるレイアウト規則のフィルタリングされたリストに移動できます。これにより、管理者はより的を絞った方法でレイアウト規則を管理できるようになります。
レイアウトプロファイル - 基本レイアウトの追加
特定のオブジェクトタイプのレイアウトを設定する場合、管理者は、そのオブジェクトタイプのレイアウトのみに限定するのではなく、基本レイアウトを追加するオプションを利用できるようになります。基本レイアウトを複数のオブジェクトタイプに使用できる場合、管理者はアクションレイアウトを活用して、レイアウトの合計数を減らすことができます。
Documents
Doc Viewer: Context Menu Usability
23R3 では、ドキュメントビューアコンテキストメニューを導入しました。これにより、ユーザはドキュメントビューア内で右クリックすることで、ドキュメントコンテンツに対する一般的なアクションに迅速かつ直感的にアクセスできるようになりました。
24R1 では、Grab ボタンがデフォルトで無効になり、Annotate Mode が有効になります。注釈が存在する場合、またはユーザに注釈権限がある場合は、デフォルトで Annotate Mode が有効になります。24R1 より前は、テキストの選択を必要とするコンテキストメニューアクション (用語集検索の実行など) を実行する場合、デフォルトで選択されている Grab ボタンを最初に無効にする必要がありました。
ユーザは必要に応じて、Grab ボタンを選択するか、キーボードの CTRL+Shift (または CMD+Shift) を使用して、Grab を有効にできます。
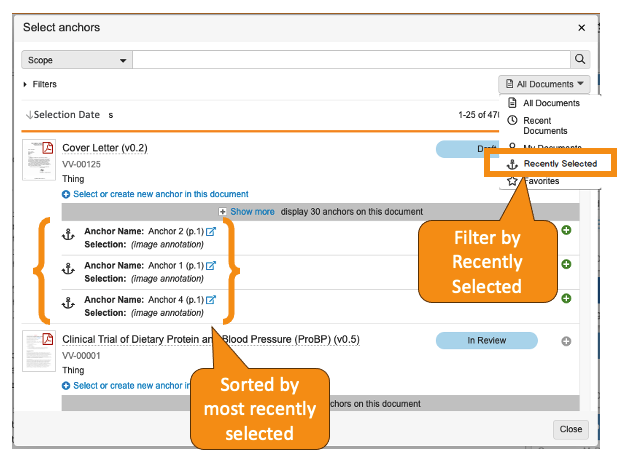
Select Anchor Improvements
アンカーを利用してドキュメント内の特定の場所にリンクするユーザにとって、ユーザエクスペリエンスが強化され、アンカーの検索、参照、選択が容易になりました。
Select Anchor ダイアログボックス内:
- 検索ボックスの下にある Views メニューに、ユーザが最近選択した 25 件のドキュメントを表示する、Recently Selected オプションが追加されました。
- ユーザがメニューでいずれかの項目を選択すると、次にそのユーザがアンカーの選択ダイアログを開いたときに、Vault によってその選択が再適用されるようになりました。
- 各ドキュメントの下に表示されるアンカーが、そのユーザの最新の選択に基づいて並べ替えられるようになりました。
これらの機能強化により、アンカーのヘビーユーザはドキュメント全体でアンカーをより効率的に適用・再利用できるようになります。
詳細については、アンカーをご覧ください。
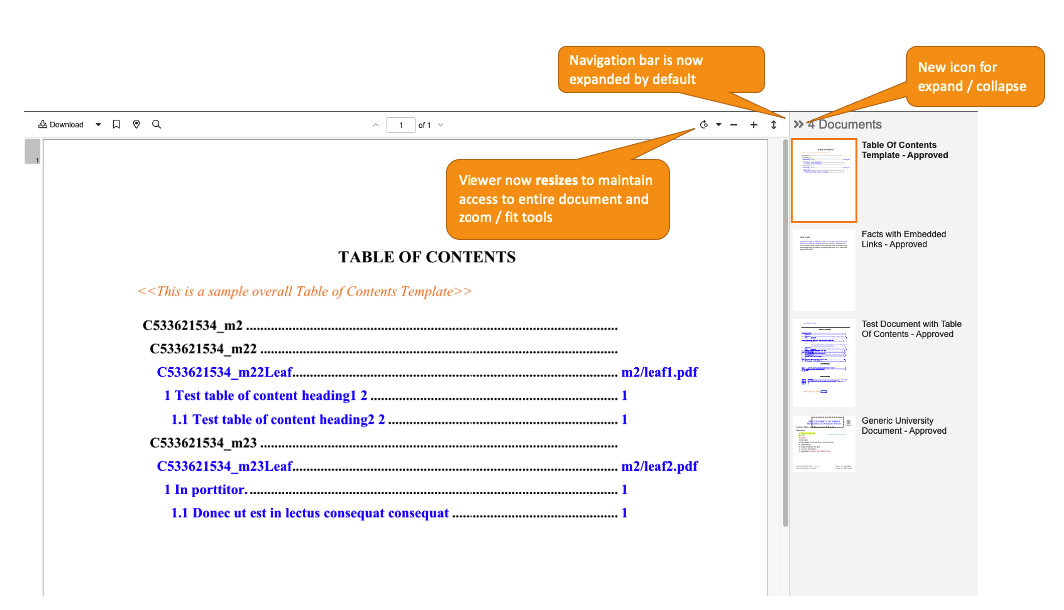
External Viewer: Improved Multi-Document Navigation
External Viewer で一度に複数のドキュメントを使用する場合のユーザエクスペリエンスが向上し、External Viewer がより直観的になる機能強化が施されました。
- ドキュメントのサイドバーはデフォルトで自動的に展開されるため、複数のドキュメントを確認する必要があることがユーザにわかりやすくなります。24R1 より前は、複数のドキュメントを受け取ったユーザは、ドキュメントのサイドバーを手動で展開しないと、追加のドキュメントが含まれていることを見逃す可能性がありました。
- ドキュメントのサイドバーは、ユーザが明確に折りたたむことを選択しない限り展開されたままになります。24R1 より前は、ユーザが特定のドキュメントを選択すると、ドキュメントのサイドバーは自動的に折りたたまれていました。
- ドキュメントのサイドバーを折りたたむ/展開するアイコンが、左/右矢印に変更されます。
- ドキュメントのサイドバーが展開されると、サイドバーがドキュメントやズームコントロール (回転、フィット、ズーム) を覆い隠さないように、External Viewer がドキュメントのサイズを自動的に変更します。
External Viewer には直接 URL 経由でアクセスされ、Vault ユーザ以外にドキュメントを送信するためによく使用されます。複数のドキュメントを直接 URL 経由で External Viewer に送信する一般的な使用例は次のとおりです。
- MedInquiry の回答パッケージの送信
- Medical と PromoMats の Approved Email
- SiteConnect を介した安全性情報の配布 (有効なアグリーメントのない施設に対して)
- SafetyDocs 内電子メールでの PVA ドキュメントの配布
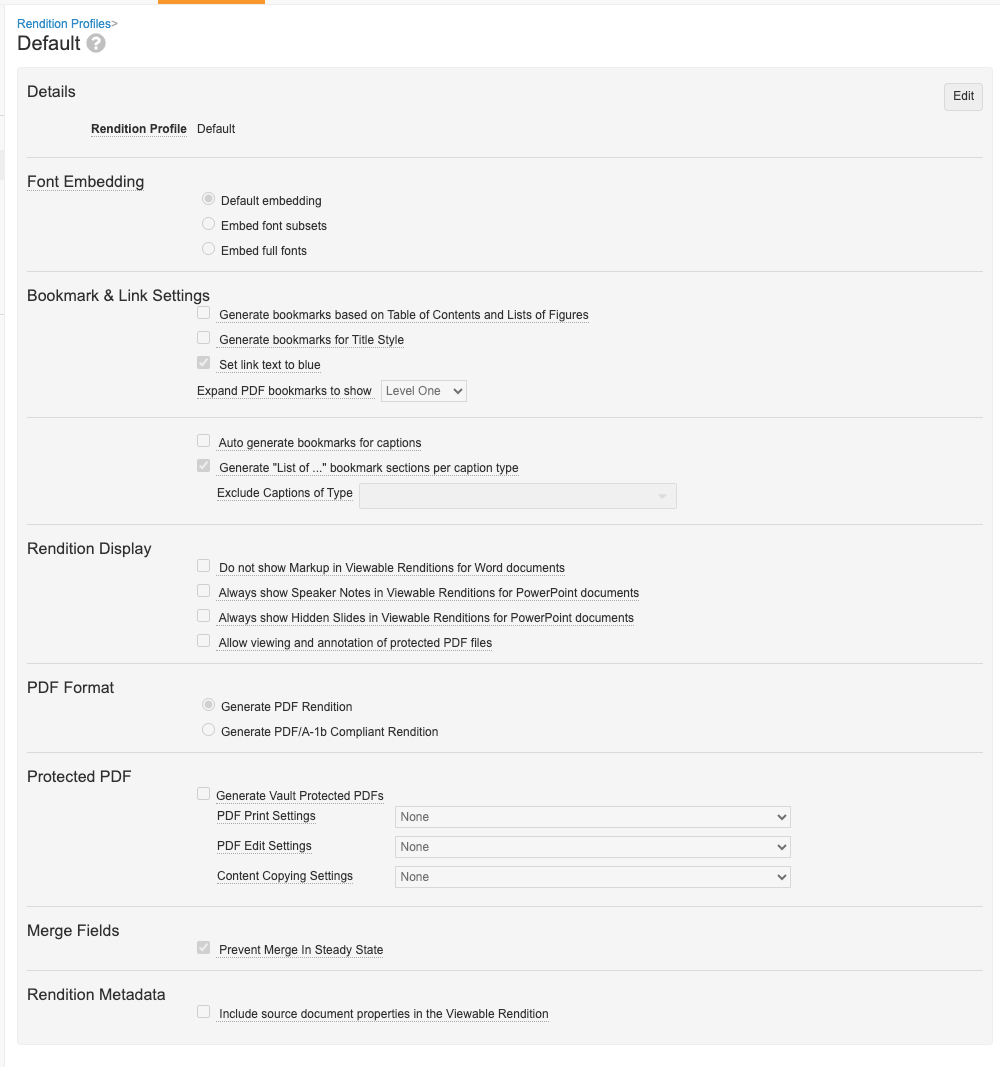
Consolidated Rendition Settings & Profiles
管理者は、設定のレンディションプロファイルページを介してすべてのレンディション設定を管理できるようになります。24R1 より前は、「Vault 全体」のレンディション設定は Admin > Settings > Rendition Settings で管理され、より具体的な設定はレンディションプロファイル内で管理されていました。
この機能強化により、すべての設定が 1 か所 (レンディションプロファイル) で管理されるようになり、プロセスが合理化され、管理者によるレンディションに関する質問のトラブルシューティングが容易になります。
現在、Admin > Settings > Rendition Settings で適用されている設定は、エンドユーザの動作の変更がないように、デフォルトのレンディションプロファイルに移行されます。
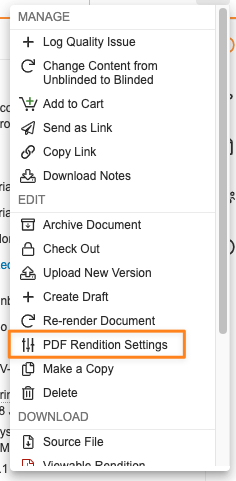
この機能強化により、エンドユーザが Word のマークアップ、Powerpoint のレイアウト、PDF の透かしの外観を個別に調整する機能は廃止されます。ユーザには、ドキュメントの Actions メニューに PDF Rendition Settings オプションが表示されなくなります。
今後は、Vault がマークアップとレイアウトの表示をどのように処理するかは、レンディション プロファイルによって決定されます。PDF レンディション設定が削除されたため、ほとんどのお客様が透かしの管理にオーバーレイを使用しているため、[PDF では非表示だが印刷可能な (「印刷のみ」) 透かしを表示する] オプションは利用できなくなりました。この変更により、お客様はさらなる制御が可能になり、マークアップが表示された状態でドキュメントが承認されるシナリオを回避できるようになります。
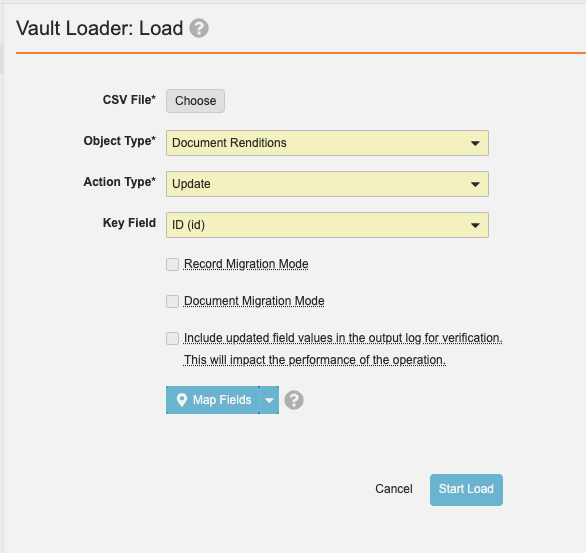
Bulk Rendition Operations
管理者は、UI または API で Vault Loader を使用してドキュメントを一括再レンダリングできるようになります。24R1 より前は、ドキュメントの一括再レンダリングを実行するには、お客様は製品サポートに問い合わせる必要がありました。
CSV ファイルには、ID、major_version_number__v、minor_version_number__v を含める必要があります。一括再レンダリングは、CSV ファイルを入力として使用し、ドキュメントレンディションの更新アクションを通じて Vault Loader によって実行できます。
この機能強化により、管理者は Vault サポートに問い合わせることなく上記のアクションを実行できるようになります。
ファイルが再レンダリングのキュー処理に失敗した場合もプロセスは続行され、操作が完了すると、失敗は概要ファイルに記録されます。
このキュー処理プロセスは非同期でも実行されます。つまり、ユーザはレンディションのキュー処理中もドキュメントにアクセスできます。管理者は、ジョブステータスページでもこのプロセスを監視できます。キュー処理されると、各レンディションリクエストのステータスが、Vault 管理者の操作タブにある管理者用レンディションステータスに表示されます。
詳細については、再レンダリングをご覧ください。
Support for High-Efficiency Image Rendering
Vault は、OCR、オーバーレイ、署名ページのサポートを含む、HEIF ファイルと HEIC ファイルのレンダリングをサポートするようになりました。ファイルがアニメーション化されている場合、Vault はファイルをビデオレンディションとしてレンダリングします。また、Vault は、これらのレンディションに対して光学文字認識 (OCR)、オーバーレイ、署名ページもサポートしています。
HEIC ファイルと HEIF ファイルは、モバイルデバイス (Android や Apple デバイス) のカメラアプリケーションで使用される一般的な形式です。これらのレンディションサポートを追加することで、お客様はこれらのファイルタイプをより適切に操作できるようになります。
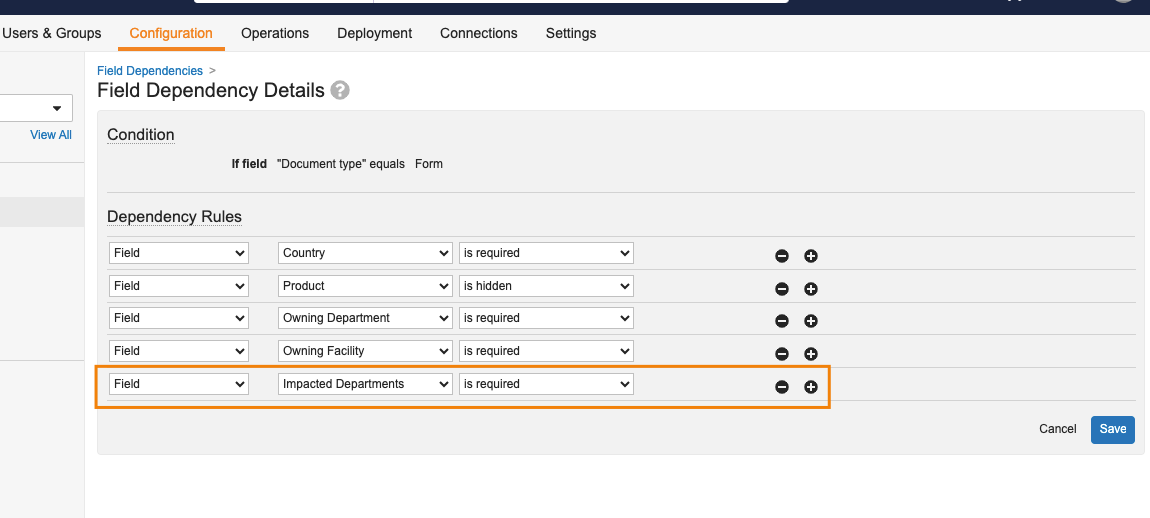
フィールド依存性の監査機能強化
管理者が既存ドキュメントフィールドの依存性を変更した場合、システム監査履歴にその変更内容がより明確に表示されるようになりました。
24R1 より前でもこれらの変更は監査されていましたが、具体的な変更内容は明確に表示されていませんでした。この機能強化により、お客様にとって発生した変更内容をより詳細に特定しやすくなったほか、監査証跡情報の堅牢性が向上しました。
たとえばこの例では、既存のフィールド依存性に、影響を受ける部門を必須とする新規条件が追加されています。
システム監査履歴には、この変更内容の詳細が次のように表示されるようになりました。
詳細に関しては、ドキュメントフィールド依存性および監査をご覧ください。
Non-Consecutive Document IDs
ドキュメントが作成されると、Vaultは自動的にドキュメントIDを割り当てます (注意: これはドキュメント番号とは異なります)。ドキュメント ID が必ずしも連番で割り振られるとは限りません。たとえば、ドキュメントを削除することにより ID が連番でなくなる可能性があります。
24R1 では、将来の機能に対応するため、ドキュメント ID の生成方法を変更しています。この変更により、ドキュメント ID は連番でない番号になる頻度が高くなります。
ドキュメントIDフィールドは、ドキュメントの作成順序に依存するため、使用しないことをお勧めします。その代わりに、[ドキュメント番号] フィールドを使用してドキュメントの順序を追跡してください。
Document Templates Limit
24R1では、Vault ドキュメントテンプレート (基本ドキュメントテンプレートと管理ドキュメントテンプレートの両方) の制限が明確に文書化され、適用され、信頼性の高いドキュメントテンプレート機能が確保されるようになりました。
現在、各 Vault には以下の制限があります:
- 基本ドキュメントタイプの 50 のドキュメントテンプレート
- 制限に達すると、新規テンプレートを基本ドキュメントに追加できなくなります
- ドキュメント階層ごとに 500 のドキュメントテンプレート
- 制限に達すると、新規テンプレートをドキュメント階層に追加できなくなります
- Vault ごとに 5,000 のドキュメントテンプレート
- 制限に達すると、新規テンプレートを Vault のどこにも追加できなくなります
ドキュメント テンプレートの制限には、アクティブおよび非アクティブなテンプレートの両方が含まれます。
ドキュメント階層の制限には、指定された分類におけるすべてのローカルドキュメントと継承ドキュメントのテンプレートが含まれます。たとえば、合計 500 のテンプレートがあり、制限に達しているドキュメントテンプレートを持つ、次のドキュメントタイプ階層について考えてみましょう:
VPS 有害事象レポートドキュメント (300) > eDMR (150) > 別紙 (50)
現在、お客様がお持ちのドキュメントテンプレートがこれらの制限のいずれかを超える場合は、Veeva アカウントチームが直接ご連絡いたします。
ドキュメントテンプレートについての詳細はこちらをご確認ください。
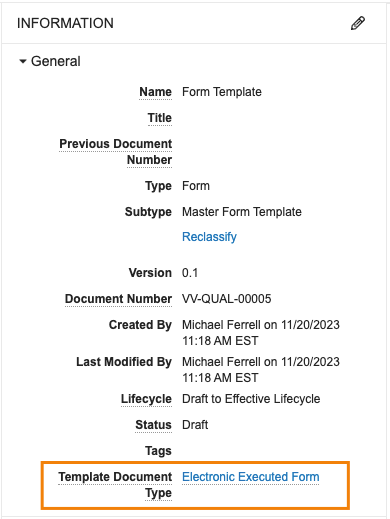
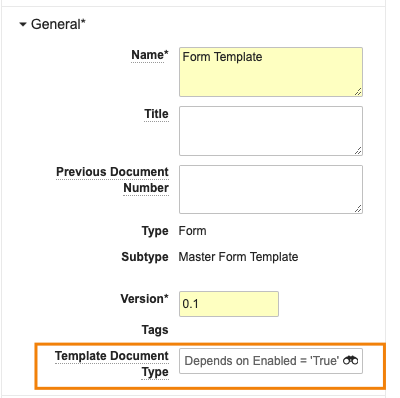
Preventing Unintended Document Template Creation
管理されたドキュメントテンプレートを活用する場合、テンプレートは [テンプレート ドキュメント タイプ] フィールドを使用して、ドキュメントタイプに関連付けられます。
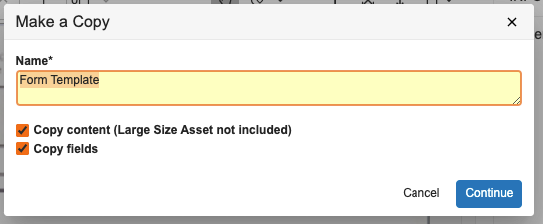
24R1では、ユーザーがテンプレートに対して [コピーを作成アクション] を実行した場合、[テンプレートドキュメントタイプ] フィールド値をコピーしなくなりました。これにより、不必要で意図しないドキュメントテンプレートが新たに作成されるのを防止できるようになりました。ただしユーザーは、必要に応じて、コピーを作成した後でも [テンプレートドキュメントタイプ] フィールドを手動で更新できます。管理者は、必要に応じてデフォルトでコピーするようにフィールドを設定することもできますが、この機能は、デフォルトで値をコピーしません。
以下は、Quality Vault のフォームテンプレートの例で、テンプレートドキュメントタイプフィールドが入力されています。
ユーザーが [コピーを作成] アクションを実行すると:
新しいドキュメントは、[テンプレートドキュメントタイプ] フィールドで空白になります:
Binder Limits
適切なバインダーパフォーマンスを確保するため、Vault バインダーの制限が明確に文書化され、実施されるようになりました。お客様とVeevaサービスは、予測可能で理解しやすい方法でバインダーを使用できます。
24R1 では、以下の制限がバインダーに適用されます:
- バインダーの上限は 50,000 ノードです。バインダーがこの制限に達すると、バインダーノードを追加できなくなりますが、削除することは可能です
- この50,000ノードの制限には、バインダーのコンポーネント バインダーのノードが含まれます。コンポーネントバインダーが、50,000ノードの制限に達していなくても、バインダーが限界に達している場合は、バインダーノードをそのコンポーネントバインダーのいずれにも追加することはできません。
- バインダーの合計サイズが 10,000 ノード以上になると、Create Draft 経由でのバインダーのバージョン管理や、コピーができなくなります。このノード数には、コンポーネントバインダー内のノードが含まれます。Set New Major Version Entry Action は、バインダーに 10,000 以上のノードがある場合でも、ブロックされません。
Vault 内のバインダーの数に制限はありません。Veeva アカウントチームは、Vault にこのような制限を超えるバインダーが含まれている場合、お客様に直接連絡します。
Vault のバインダーについての詳細に関しては、こちらをご覧ください。
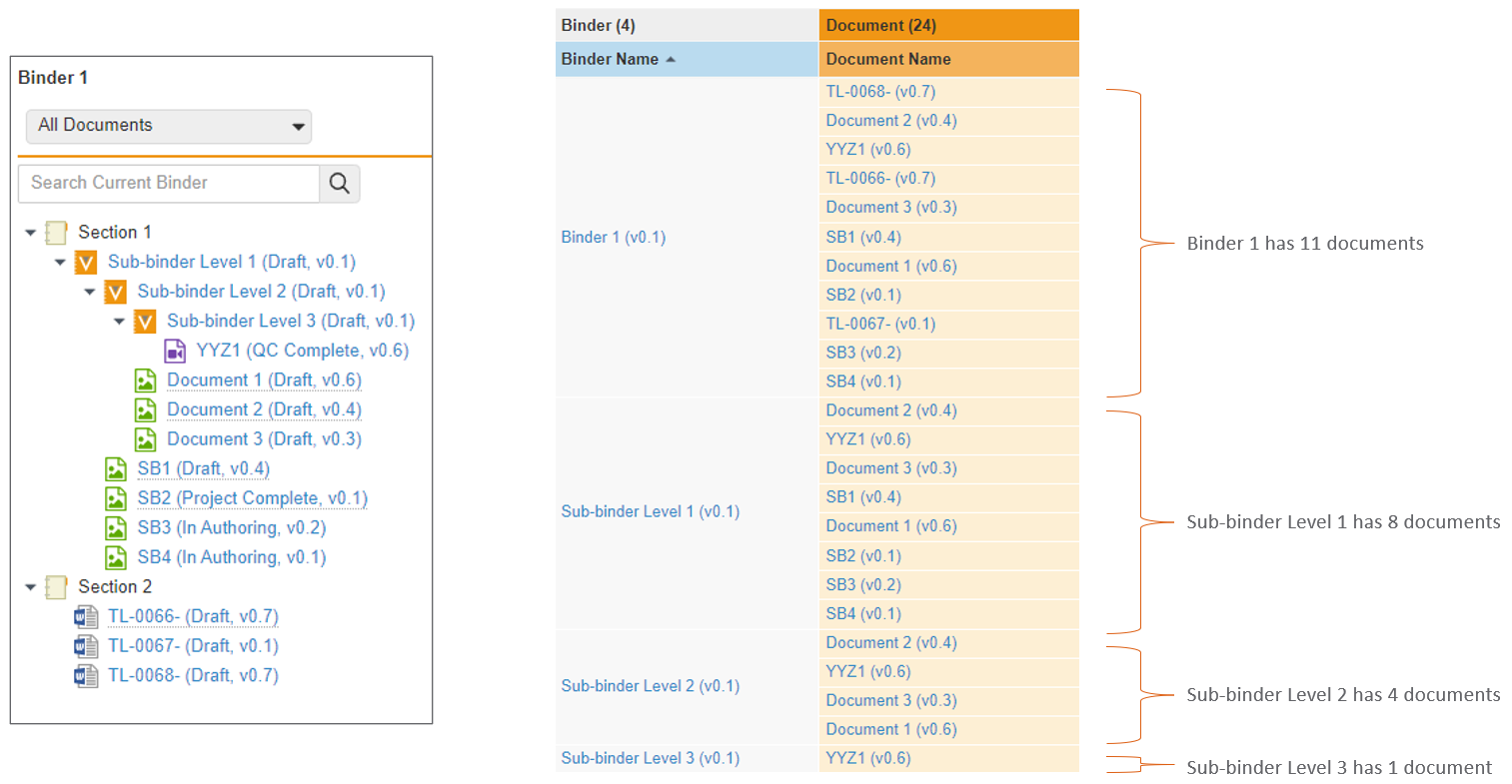
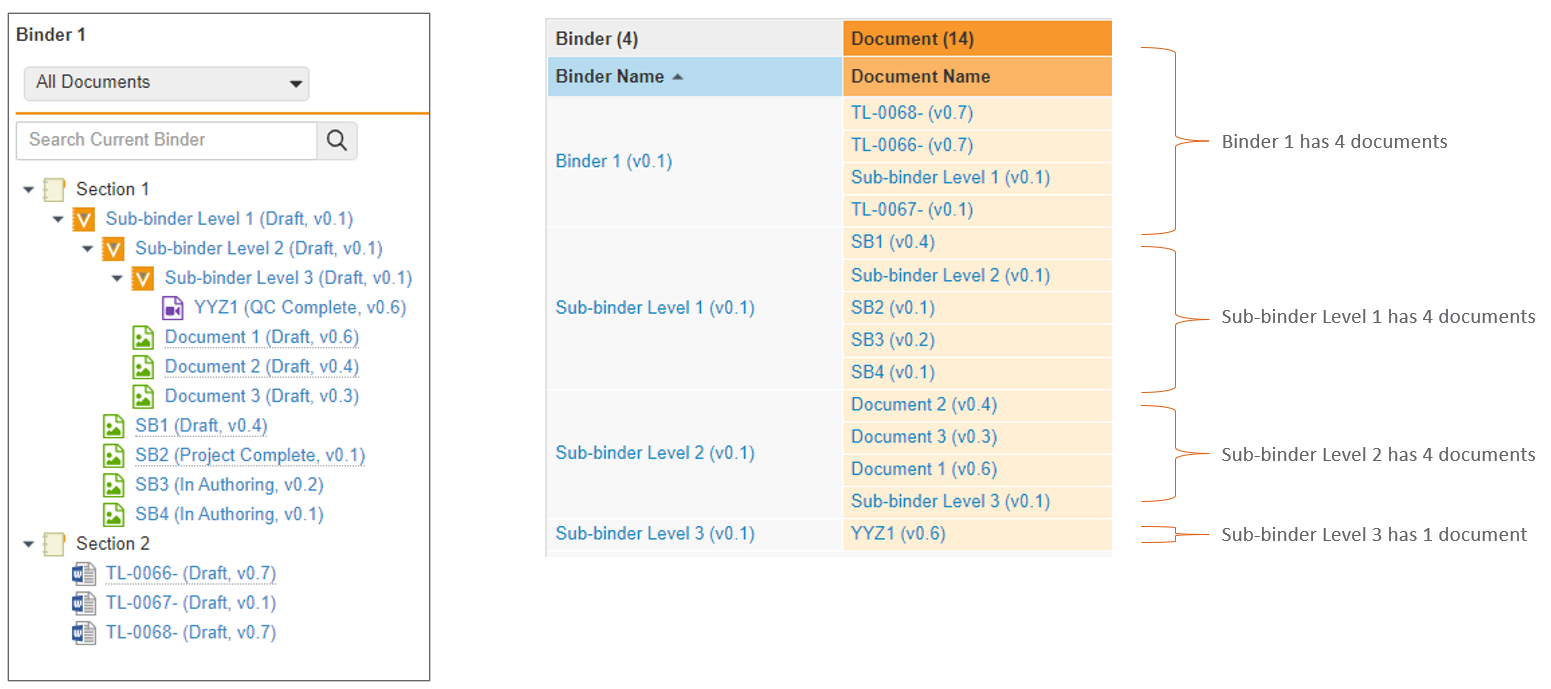
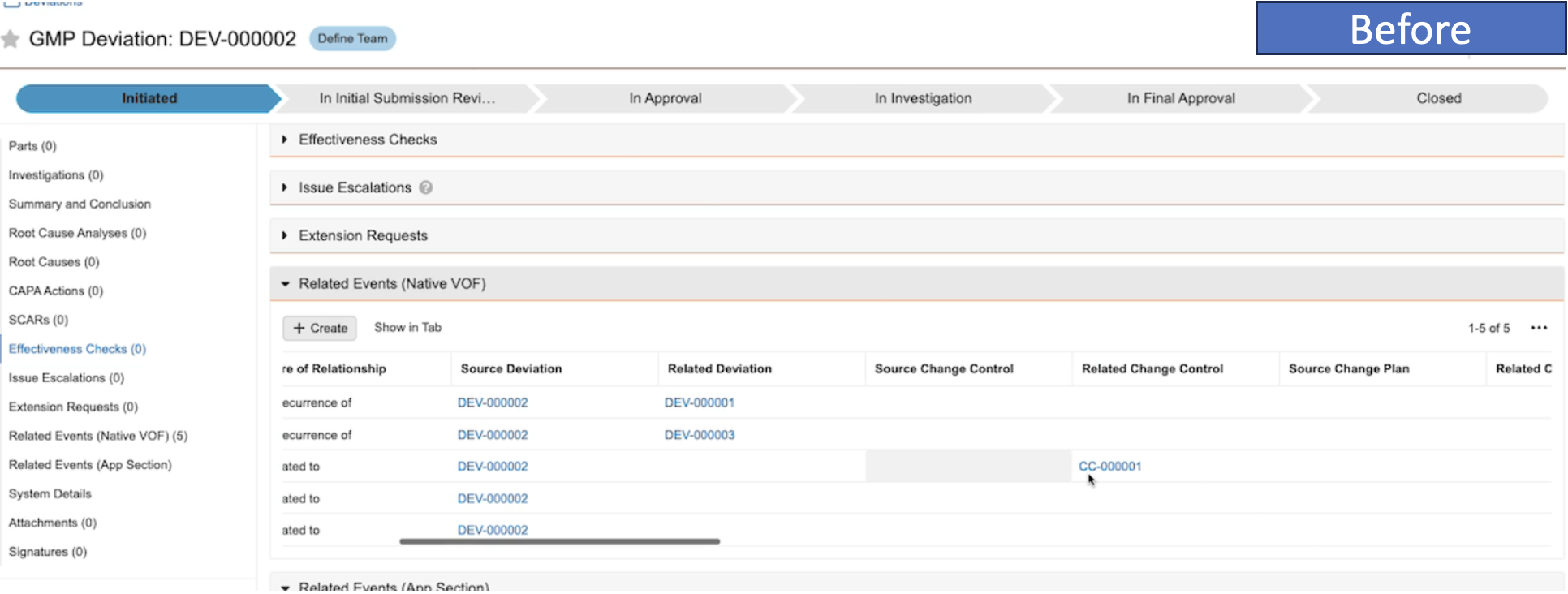
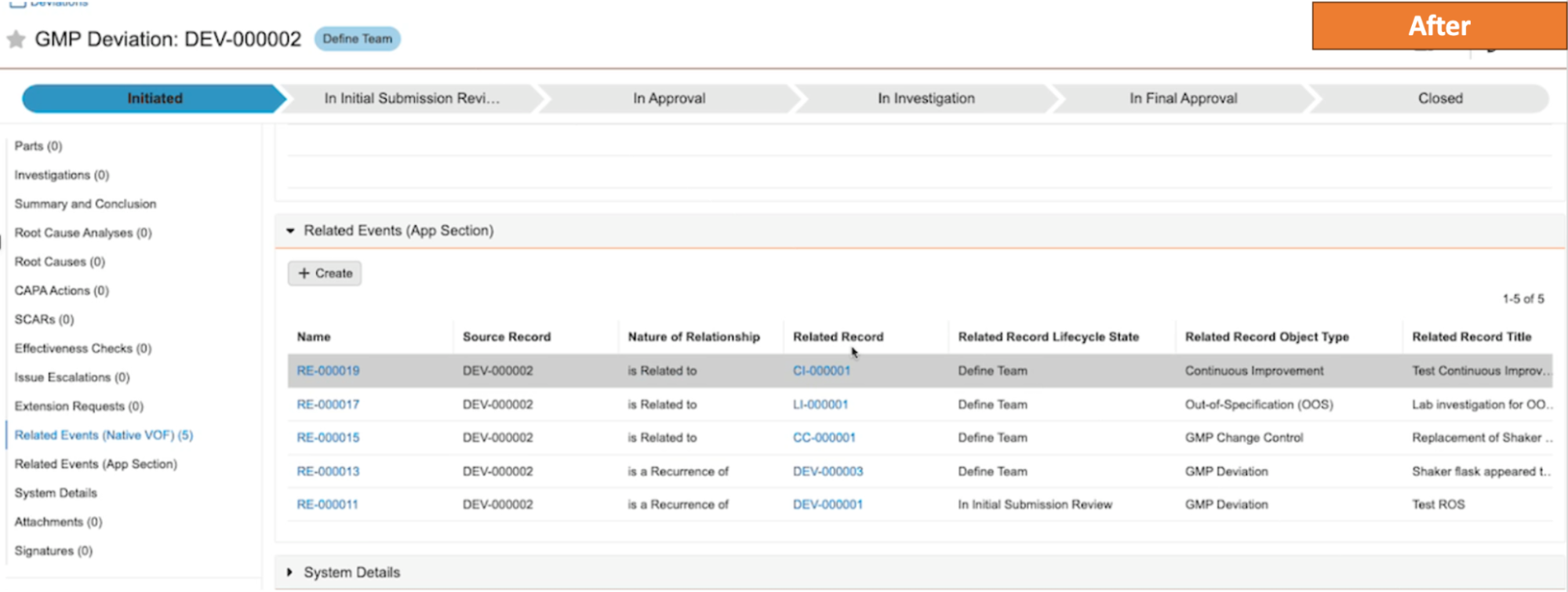
ネストされたバインダーに関するレポート作成の改善
コンポーネントバインダーを含むバインダーについてレポートを作成する場合、Vault はコンポーネントバインダーを親バインダー内のドキュメントとして表示するようになります。24R1 より前では、Vault はコンポーネントバインダー内のすべてのドキュメントを表示し、それらが親バインダーに直接存在しないことを示しませんでした。
例として、以下のシナリオでは、左側の構造にいくつかのコンポーネントバインダーが表示されています。24R1 より前は、これについてレポートを作成すると、ドキュメント YYZ1 がコンポーネントバインダーと親バインダーの両方に属していると示されていました。
24R1 以降、同じ構造が簡素化されます。親バインダーにはその中のコンポーネントバインダーが表示されますが、ドキュメント YYZ1 は直接属するコンポーネントバインダーにのみ表示されます。
この機能強化により、レポート結果が簡素化され、他のバインダー内でバインダーを使用する場合の関係をより正確に把握できるようになります。
詳細については、バインダーをご覧ください。
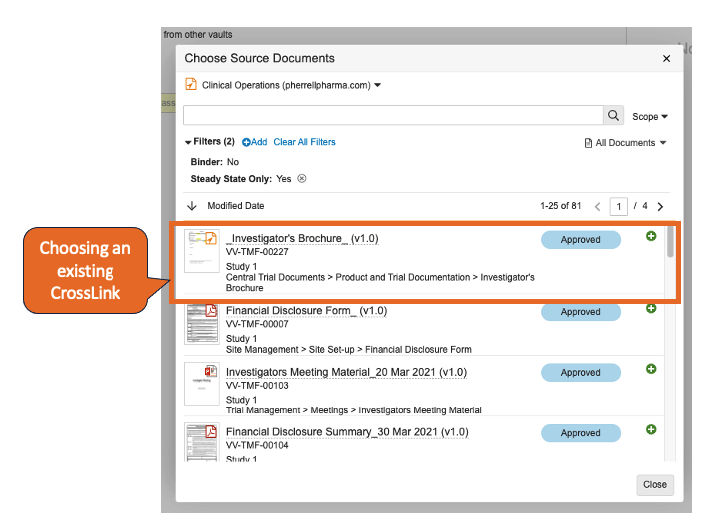
Crosslink a Crosslink
Vault は、単一ドメイン内で、ある Vault から別の Vault に既存の CrossLink をクロスリンクする機能をサポートするようになりました。この機能強化の主な使用例は、Vault Connections 周辺です。具体的には、最初にドキュメントをプロトコルなどの RIM で取得し、次に eTMF に (RIM から Clinical Operations への接続を介して) クロスリンクし、その後再び Study Training に (Study Training から Clinical Operations への接続を介して) クロスリンクすることが考えられます。
24R1 より前は、これを実現するには、Study Training と eTMF Vault の両方が RIM 内のソースを個別に参照する必要があったため、Study Training は RIM ドキュメントから直接ドキュメントを取得する必要がありました。
この機能強化により、複数の Vault 間でドキュメントをより包括的に参照できるようになり、製品化された Vault Connections が強化されます。
詳細については、CrossLink と Vault Connections をご覧ください。
管理者のクロスリンク設定
デフォルトで、クロスリンクにはソースドキュメントの署名ページとオーバーレイが含まれますが、製品サポートチケットを提出して、Admin > Settings > General Settings で次のオプションの表示を要求できます。
管理者が、24R1でこれらのオプションをデフォルトで利用できるようにすることで、これらのオプションを有効にするために、お客様がVeevaを経由する必要性を軽減します。チェックボックスはデフォルトで管理者に表示され、編集可能になりますが、以前に公開および編集されたことがないVaultでは、デフォルトでチェックが外されています。
File Staging Enhancements for Vault File Manager
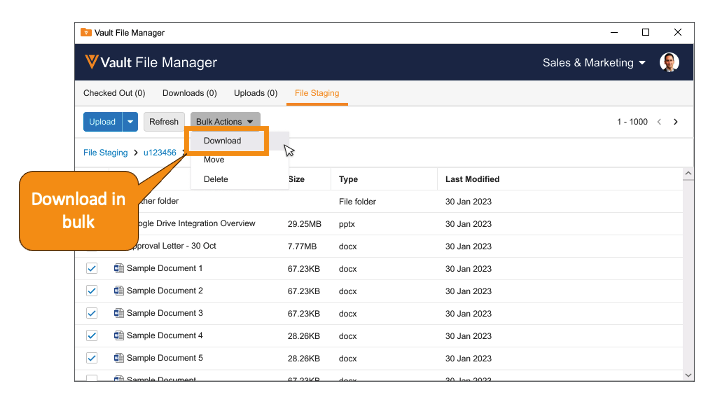
Vault File Manager は、ファイルステージングタブ内で直接フォルダを作成する機能、およびファイルステージングタブからドキュメントとフォルダを (個別または一括) ダウンロードする機能をサポートするようになりました。
23R2 では、Vault File Manager を新しい File Staging タブで強化しました。これを使用すると、管理者は別のファイル転送プロトコルセキュア (FTPS) クライアントを使用せずに、Vault File Manager でコンテンツをステージングして Vault にインポートできるようになりました。
23R3 では、Vault File Manager のファイルステージング機能が強化され、ユーザがフォルダ/ファイルの名前変更と削除、およびフォルダ間でのファイルの移動ができるようになりました。ただし、実際に新しいフォルダを作成するには、ユーザがデスクトップ上にフォルダを作成し、そのフォルダを Vault File Manager のファイルステージングタブに移動する必要がありました。
24R1 では、このプロセスでユーザが Vault File Manager の外部で作成を実行する必要がなくなりました。
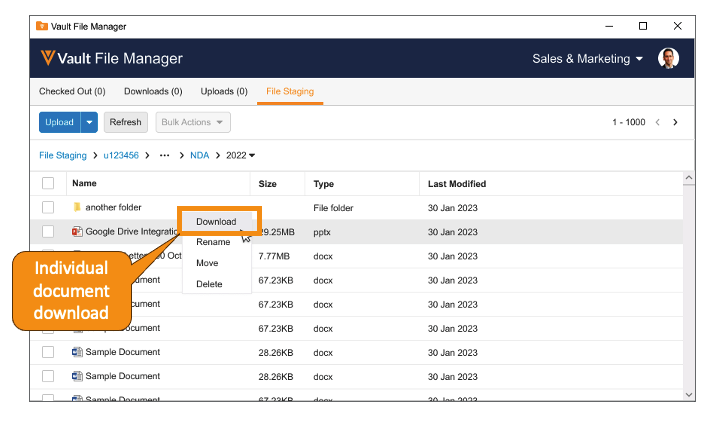
また、24R1 より前では、ファイルステージングからドキュメントをダウンロードする必要がある場合、サードパーティの FTP クライアントの使用が必要でした。24R1 では、Vault File Manager からドキュメントとフォルダをダウンロードできるようになりました。
多くの組織では、FTPS の使用と互換性のないファイアウォールやセキュリティポリシーが導入されている可能性があります。Vault File Manager 内にフォルダを作成する機能を追加することで、お客様は Vault File Manager をより適切に活用して、サードパーティの FTP クライアントを必要とせずにファイルステージングを管理できるようになります。
詳細については、ファイルステージングをご覧ください。
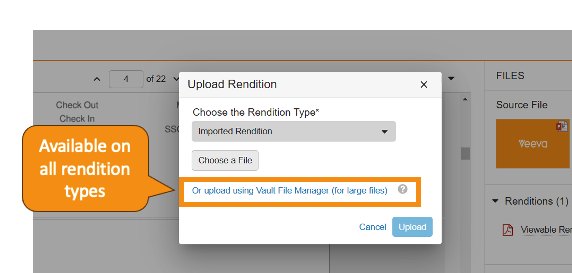
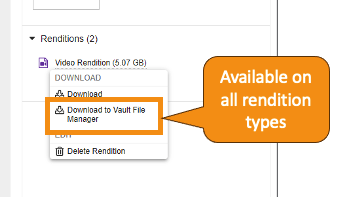
Vault File Manager でのレンディションタイプのサポートの拡張
Vault File Manager は、ダウンロードアクションとアップロードアクションの両方ですべての標準レンディションタイプをサポートするようになりました。24R1 より前は、Vault File Manager はアップロードについては大きなサイズのアセットレンディションのみをサポートし、ダウンロードについてはすべてのレンディションタイプをサポートしていませんでした。24R1 では、Vault File Manager 経由で次のレンディションタイプもダウンロードできます。
- ビデオレンディション
- オーディオレンディション
- Commercial Vault
- 配布パッケージ
- Veeva HTML
- Veeva プレビュー
- アセット
- eCTD 提出パッケージ
- Clinical Operations Vault
- インポート済みの監査証跡
- 履歴監査証跡
- Veeva eForm
- 施設レンディション
- SiteVault
- スポンサー/CRO レンディション
カスタムレンディションタイプは、これまではダウンロード時のみサポートされていましたが、今後はアップロード時にもサポートされるようになります。さらに、Vault File Manager では最大サイズ 500 GB のレンディションファイルのダウンロードおよびアップロードがサポートされるようになりました。
詳細については、Vault File Manager とレンディションタイプをご覧ください。
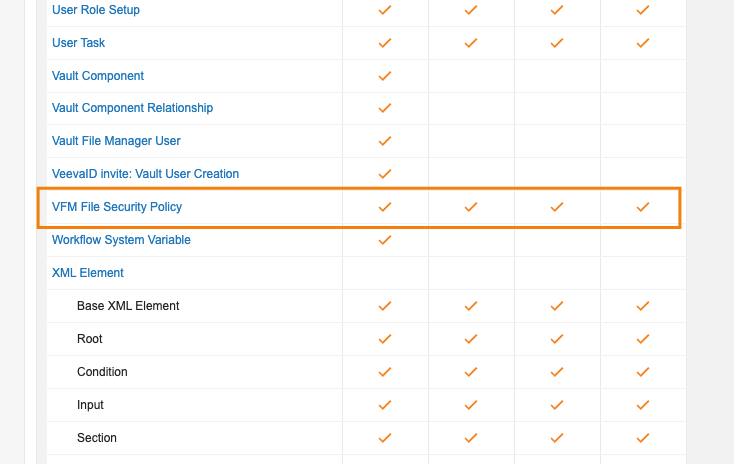
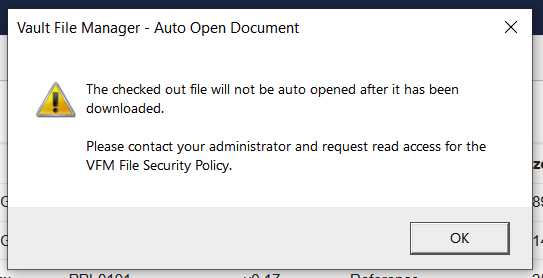
VFM File Security ポリシーが見つからない場合のエラーメッセージの改善
Vault File Manager (VFM) にドキュメントをチェックアウトする際、ユーザが権限セット内の VFM File Security Policy オブジェクトに対する Read アクセス権を持っていない場合に、VFM がファイルを自動で開こうとすると、より明確なエラーメッセージが表示されます。
24R1 より前には、管理者に連絡するための適切なコンテキストがユーザに提供されない一般的なエラーメッセージが存在していました。エラーメッセージに問題が明確に示されるようになりました。
詳しくは、Vault File Manager をご覧ください。
Reusable Labels for Document Field Layouts
Vault では、Document Field Layout ラベルが一意である必要がなくなりました。名前には引き続き一意性が適用されます。
この機能強化により、ドキュメントフィールドレイアウトの一意性が、他の Vault コンポーネントと一致した方法で処理されるようになります。
詳細については、ドキュメントフィールドレイアウトをご覧ください。
ライフサイクルとワークフロー
高度な開始ワークフロー
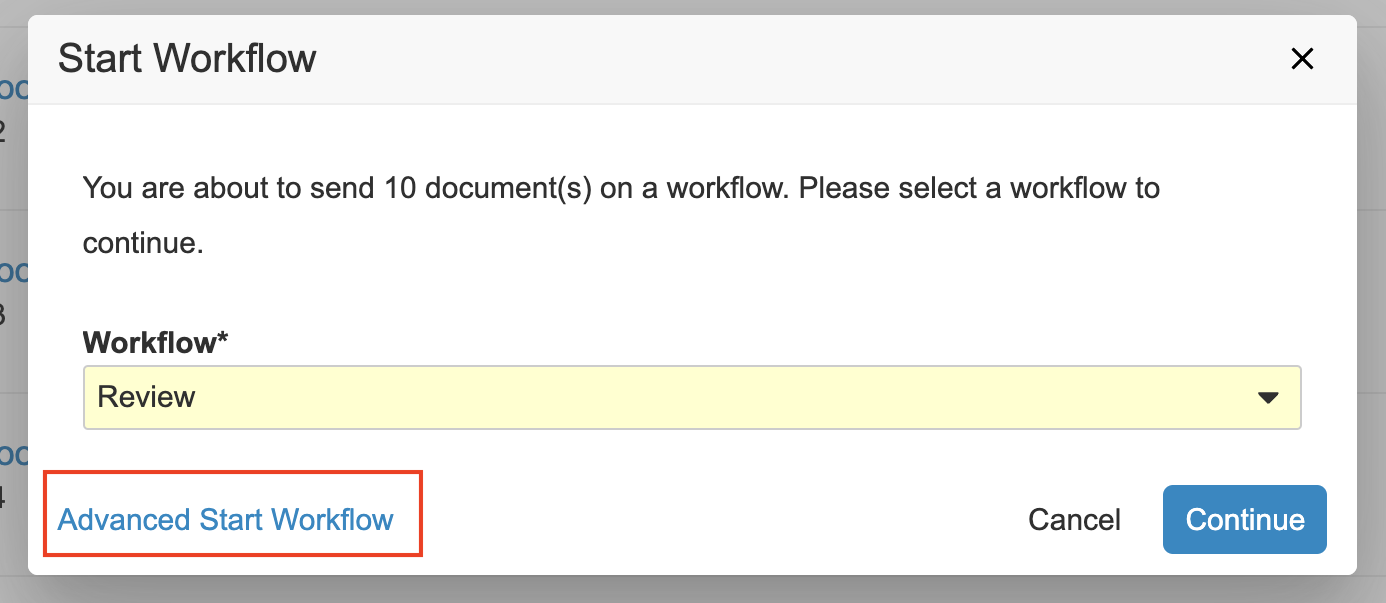
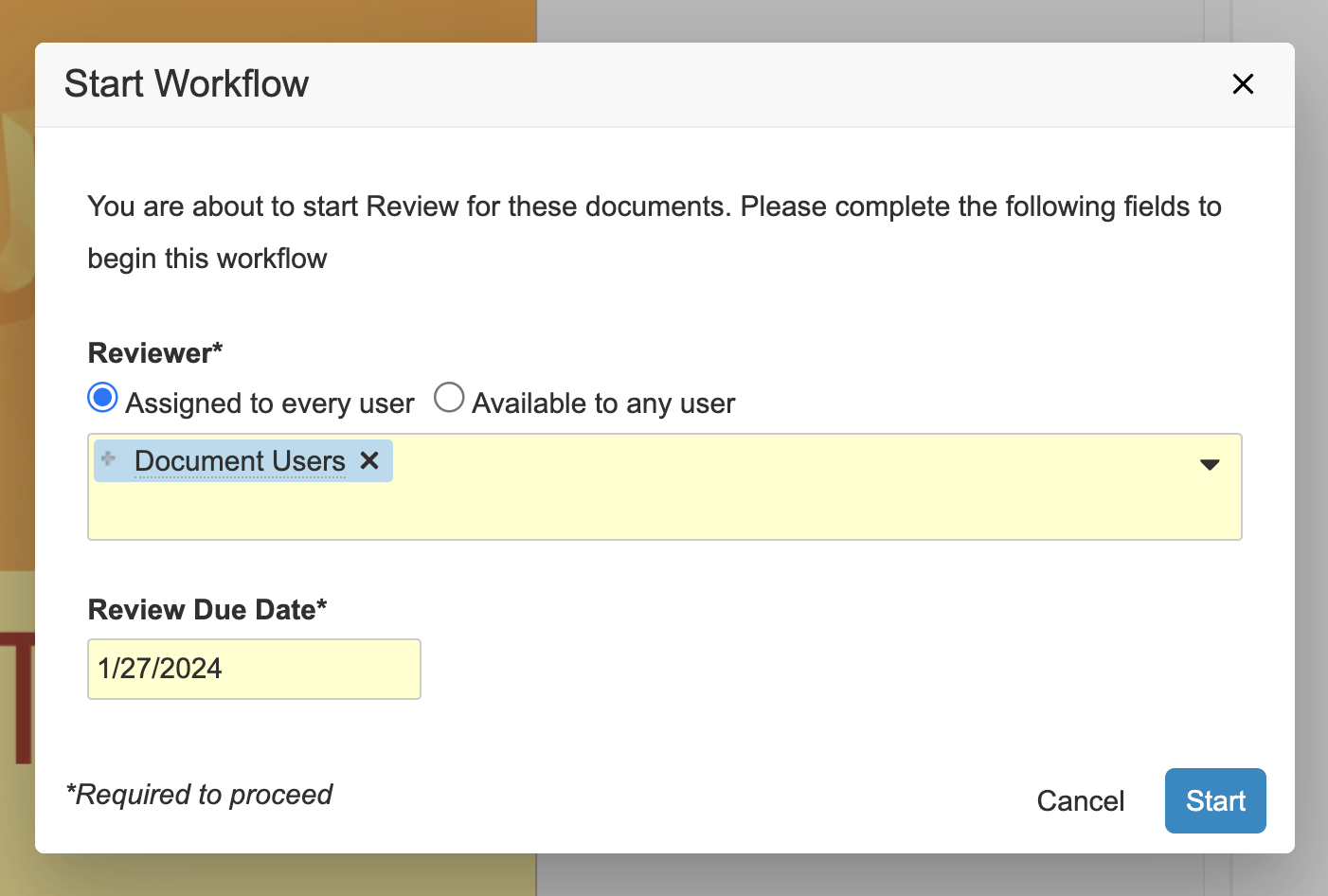
フィルタリングされた項目のリストから複数ドキュメントまたは複数レコードのワークフローを開始する場合、それらの項目をすべて同じ複数項目のワークフローで一緒に送信するのではなく、サブセットにグループ化する方法が必要になることがあります。これを行えるようにするために、複数項目のワークフローを開始するときに詳細オプションにアクセスできる新しいリンクが追加されました。ここで、最初に選択を絞り込み、開始するワークフローを選択し、ワークフロー参加者、フィールド、日付などのワークフローの Start 手順プロンプトを入力します。
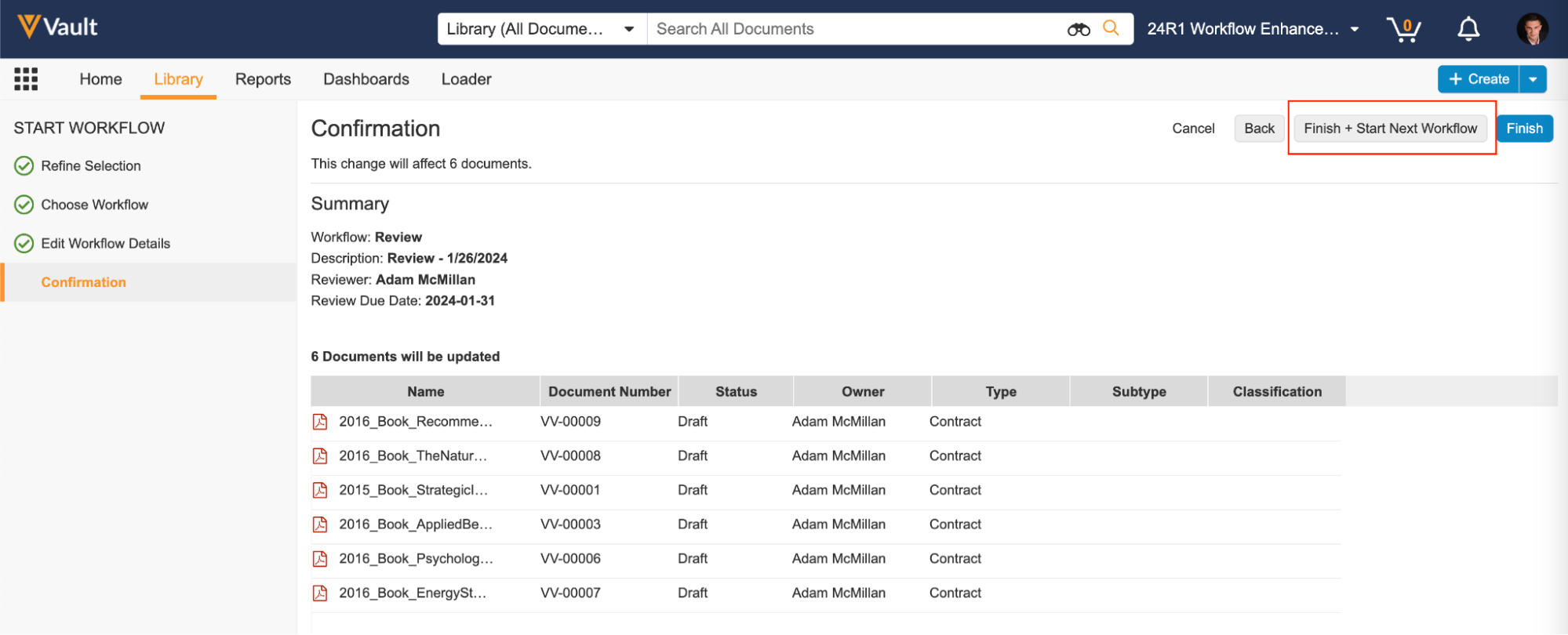
最後の手順で、Finish + Start New Workflow ボタンをクリックすると、ワークフローを開始した後、前に選択を解除した残りのアイテムに戻ってそれらのアイテムをワークフローに送信できます。
この方法は、タスクのないワークフローを実行する際に、代わりに複雑な論理手順を踏んでアイテムを送信することによりフィールドを更新して適宜進行させる場合にも適しています。タスクのない最初のワークフローが終了次第、続けて同じアイテムセットを別のワークフローに送信することができます。
あるいは、ワークフローを開始する前にいくつかの項目の選択を解除することが目的の場合は、最後の手順で Finish ボタンをクリックすることもできます。
Workflow Initiators Select Task Assignment Type
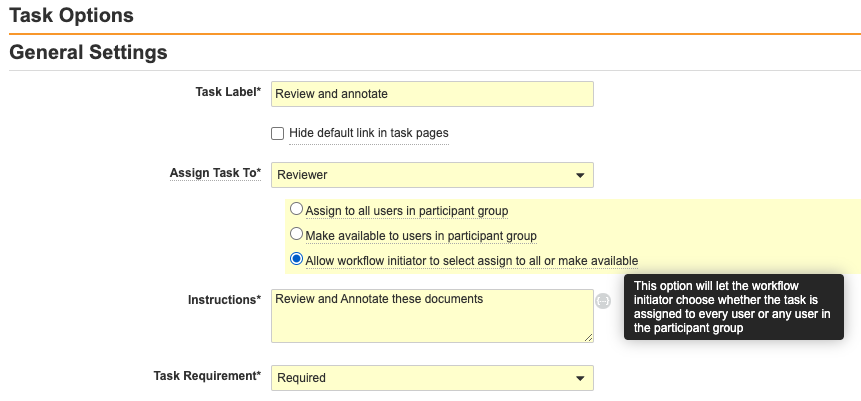
従来、管理者はタスクを設定するときに、そのタスクを参加者グループ内のすべてのユーザに割り当てるか、参加者グループ内のユーザが使用できるようにして、ワークフロー参加者グループ内のユーザがタスクを受け入れて制御できるようにするかを決定していました。ただし、シナリオによっては、この決定をワークフロー開始者に任せると便利です。このリリースでは、3 番目の設定オプションを提供します。これによりワークフロー開始者が、対応する参加者グループのワークフローの Start 手順で割り当てタイプを決定できるようになります。
上記の設定セットアップでは、ワークフロー開始者には、実行時にワークフロー参加者の選択の上に 2 つのオプションが表示されます。これらのオプションは、このワークフロー参加者グループに割り当てられた少なくとも 1 つのタスクに、新しい Allow workflow initiator to select オプションが設定されている場合に表示されます。ワークフロー開始者の選択は、ワークフロー開始者が選択できるように設定されたタスクにのみ適用され、このオプションで設定されていないタスクはオーバーライドされません。
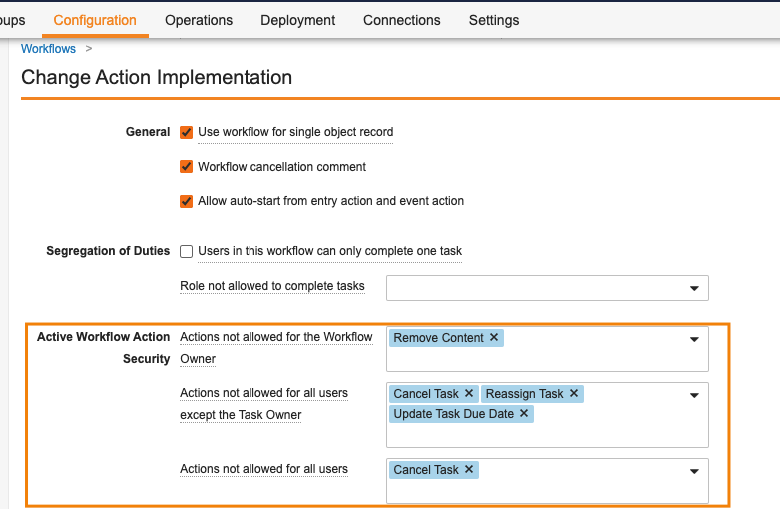
Workflow Action Security for Workflow & Task Owners
この機能により、ワークフロー所有者がワークフローのキャンセル、タスクの再割り当て、期日の更新などの特定アクションを自動的に実行できないようにワークフローを設定する新しいオプションが導入されます。ワークフローの設定時に、管理者が Active Workflow Action Security セクションを使用できるようになりました。このセクションでは、管理者は、ワークフロー所有者、すべての参加者、またはタスク所有者以外のすべての参加者から取り消されるアクションを定義できます。これらのアクションには、ワークフローのキャンセル、タスクのキャンセル、タスクの再割り当て、ワークフローの期日の更新、およびタスクの期日の更新が含まれます。
たとえば、ワークフロー所有者がワークフローをキャンセルできないようにしたり、「タスク所有者以外のすべての参加者」がタスクをキャンセルしたりできないようにすることができます (つまり、権限でワークフロータスク全般をキャンセルできるとされている場合でも、自分のタスクだけをキャンセルできるようにします)。
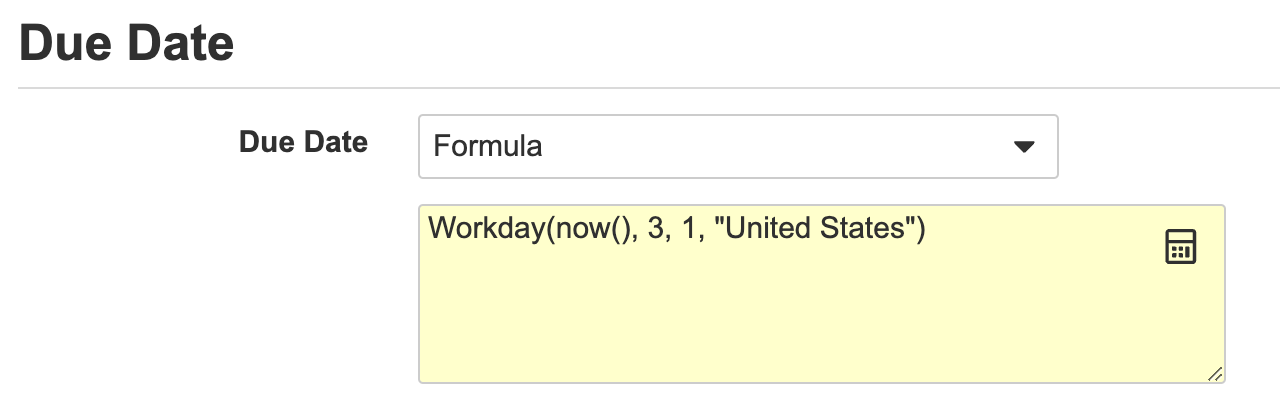
Formulas for Workflow Dates
Vault では、Vault の他の多くの領域と同様に、タスク期日の数式表現を使用できるようになりました。これにより、today() 関数や、オブジェクトレコードまたはドキュメント上の 1 つ以上の他のフィールドに基づいて日数を計算する機能など、期日の取得方法をより柔軟に制御できるようになります。
これは、ワークフロー開始手順の日付プロンプトにも適用されます。つまり、数式を使用してワークフローの期日を設定することもできます。
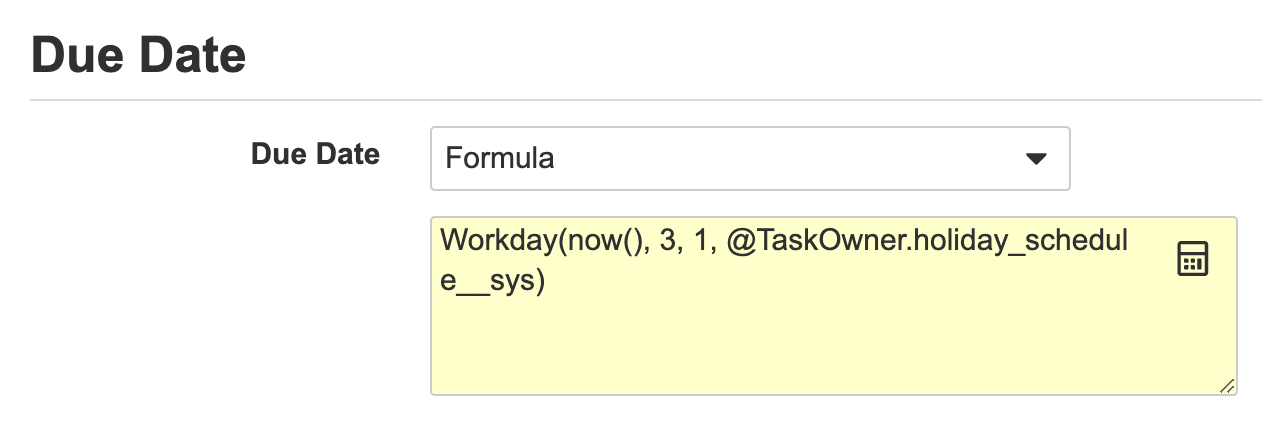
Workflow Owner & Task Owner Formula System Variables
タスク期日の新しい数式機能の一部として、ワークフロー所有者またはタスク所有者の休日スケジュールに基づいて期日を設定することもできるようになりました。たとえば、@WorkflowOwner 変数を次のように使用すると、タスク期日は、ワークフロー開始者の休日スケジュールに基づいて、今日から 10 営業日後に設定されます。
Workday(Today(),10, 1, @WorkflowOwner.holiday_schedule__sys)
これは、@TaskOwner 変数を使用する場合に特に興味深いものになります。Vault は、タスク担当者の休日スケジュールに基づいて各タスク担当者のタスク期限を設定するためです。
Document Workflow Task Due Dates Support Document Date Fields
管理者は、ドキュメントフィールドを使用してタスク期日を設定できるようになりました。ワークフロータスクの期日設定の一部として、ドキュメントフィールドが更新されたときに期日を自動的に更新するかどうかを選択できます。ドキュメントフィールドを空白に更新すると、Vault は期日を更新しません。この機能は、単一ドキュメントのワークフローでのみ使用できます。
Auto-Start Document Workflows
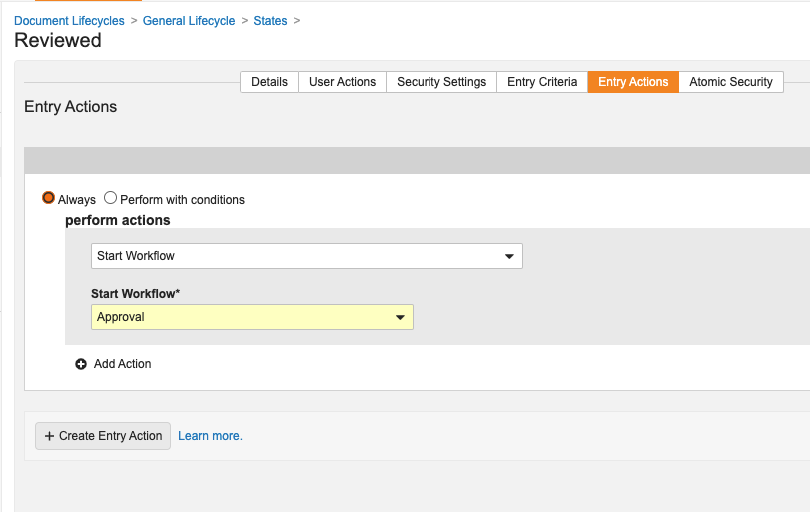
ユーザーは、ドキュメント ライフサイクル状態のエントリ アクションでドキュメント ワークフローを自動開始できるようになり、現在オブジェクト ワークフローで利用できるのと同じ種類のビジネス プロセスの自動化が可能になりました。
これにより、初期状態から安定状態への途切れることのないドキュメントフローが可能になり、クリック数や手動介入事例が削減されます。
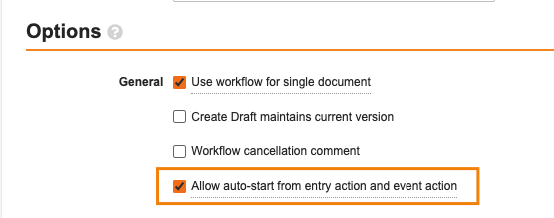
この機能強化は、特定のドキュメントライフサイクルに限定され、一度に 1 つのドキュメントに対してのみ実行するように設定されているドキュメントワークフローに適用されます。[単一ドキュメントにワークフローを使用] のチェックボックスをオンにすると、[エントリ アクションから自動開始を許可する] オプションが可能になります。
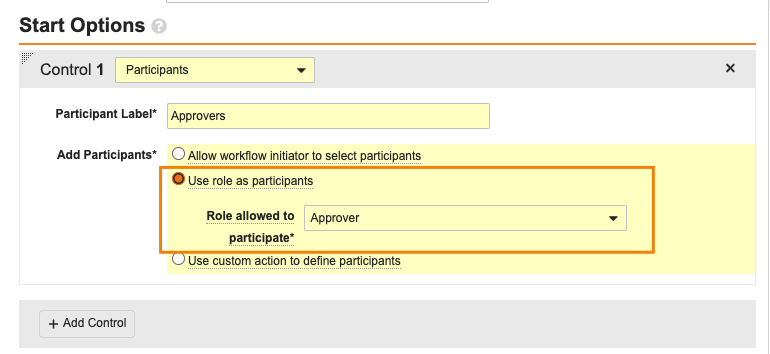
Vault がワークフローを自動開始するには、Participant コントロールが Use role as participant または Use custom action to define participants オプションのいずれかを使用する必要があります:
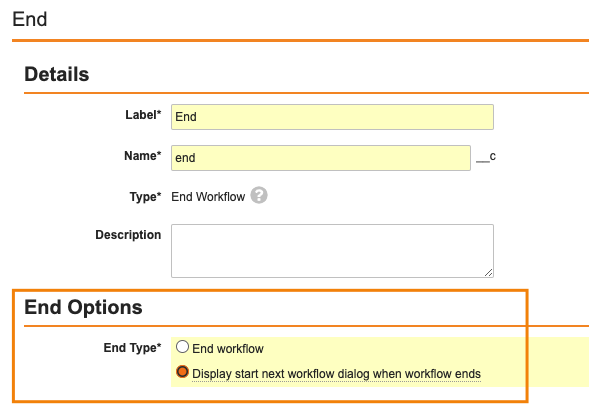
Start Next Workflow Prompt when a Multi-Record Workflow Ends
別のワークフローの終了時に、新しいオブジェクトワークフローを自動開始することが可能ですが、ユーザに複数のオプションを表示して、次の適切なワークフローを手動で選択できるようにする必要がある場合があります。
このプロセスを合理化するために、マルチレコードワークフロー機能を拡張し、ワークフローの最終タスクを完了したユーザに表示するように設定できる新しいワークフローの開始プロンプトを追加しました。
この機能は、他のワークフロータイプでは既に利用可能でしたが、マルチレコードワークフローでも使用できるようになりました。
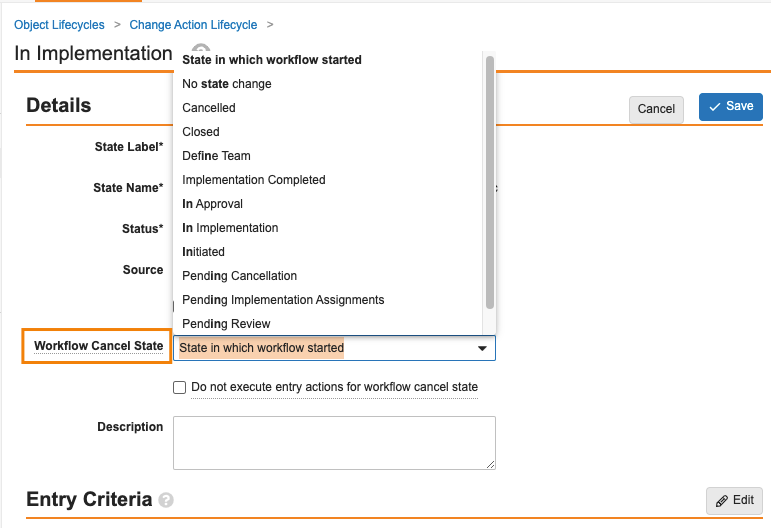
Object Lifecycle Workflow Cancel States
23R2 では、ライフサイクルエントリアクションに基づいて、オブジェクトワークフローをキャンセルする機能が導入されました。QMS などの Vaults の非常にパワフルな機能は、子レコード (つまり変更アクション) のワークフローをキャンセルするために、親レコード (つまり変更コントロール) をキャンセルする必要性が生じる可能性がありますが、キャンセル時に変更アクションがどのような状態になるかを管理者が定義できなかったという制限がありました。デフォルトの動作では、ワークフローが開始される前の状態に戻りました。
24R1では、ワークフローのキャンセル時にレコードがどの状態になるかを管理者が制御できるようになりました。これは、すべてのワークフローのキャンセルシナリオに適用されますが、特に、エントリアクションを使用して関連レコードの複数のワークフローをキャンセルする場合の一貫性が確保されます。この機能はドキュメントで可能でしたが、オブジェクトにも拡張されました。
これは、各オブジェクトのライフサイクル状態の新しいフィールドを介して制御されます。
デフォルトでこのオプションは、動作の一貫性を維持するために、既存のすべてのワークフローに対して State in which workflow started に設定されます。
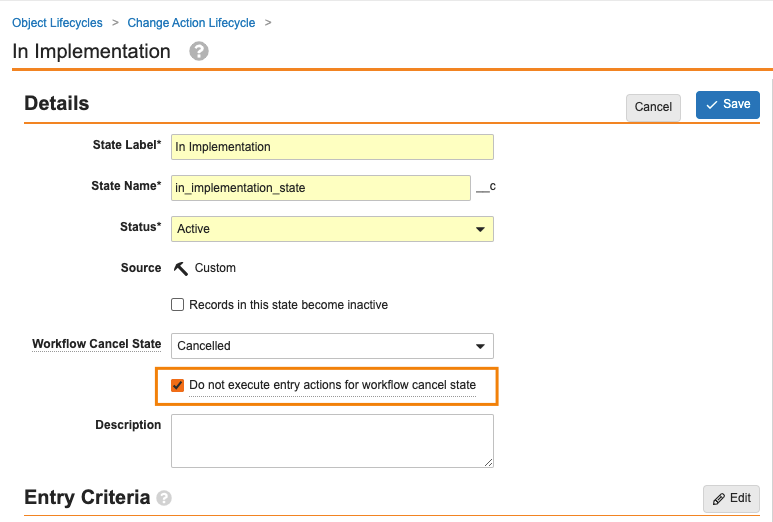
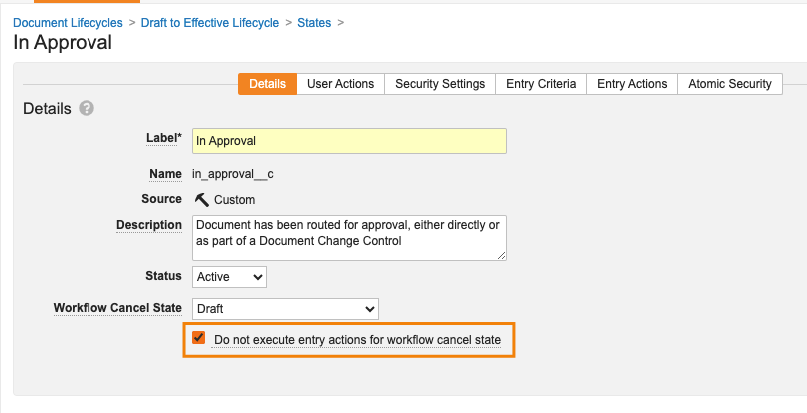
Workflow Cancel States Can Ignore Entry Actions
キャンセル状態を設定する新機能 (ドキュメントとオブジェクトの両方のワークフローで利用可能) に加えて、管理者はキャンセルに基づいてエントリアクションを実行するかどうかを制御することもできます。
このオプションが有効な場合、ドキュメントまたはオブジェクトレコードがキャンセル状態に指定されたライフサイクル状態に入ると、Vault はそのライフサイクル状態に設定されたエントリアクションを実行しません。ただし、Vault は引き続きエントリー基準を評価します。
この機能強化により、ドキュメントまたはオブジェクトがワークフローのキャンセルに基づく状態になったときに、不要なアクションが行われなくなります。
この変更は、オブジェクトワークフローとドキュメントワークフローをキャンセルする場合に適用されますが、レガシーワークフローには適用されません。
Reset Saved Task Information
ユーザがタスク完了ダイアログにフィールド情報を入力し、エラーによりタスクを正常に完了できない場合 (たとえば、入力した値が Lifecycle State Entry Criteria に準拠していない場合) でも、Vault はダイアログ内にその情報を保持するため、再度入力する必要はありません。ただし、試行している間に、ドキュメント情報またはレコードの詳細ページでドキュメントフィールドまたはオブジェクトレコードフィールドを直接編集し、変更を保存すると、Vault はそれらの以前の値を保持しなくなります。その間に行った意図的な変更を誤って上書きしないようにしてください。これは、ワークフロー内のすべての項目に適用されるタスクおよび判定のフィールドプロンプトにのみ適用されます (たとえば、項目ごとに異なる判定が許可されている場合には適用されません)。
Consistent Timeline View for Superseded Documents
ドキュメントのタイムラインビュー内で、Vault は過去版ドキュメントのイベントを一貫して表示するようになりました。24R1 より前は、状態の変化ではなくワークフローの結果としてバージョンが過去版になった場合、タイムラインビューには以前のバージョンではなく最新バージョンが過去版として表示されていました。
状態の変化によってドキュメントが過去版になった場合には、正しいバージョンのタイムラインビューに適切に反映されていました。
この機能強化により、どのようなアクションによってバージョンが過去版になったかに関係なく、タイムラインビューが正確で一貫性を保持することが保証されます。
詳細については、ドキュメントタイムラインビューをご覧ください。
Objects
Merge Records API
Vault 内では、移行、統合、または日常のアクティビティによって重複レコードが発生する可能性があります。オブジェクトレコードはさまざまな方法で参照できるため、これらの重複レコードを修正するのは困難な場合があります。たとえば、オブジェクトレコードは、設定とドキュメントおよびレコードの関係の両方で参照される場合があります。この問題は、Main レコードを Duplicate レコードとマージできる新しい Merge Records API によって大幅に単純化されました。マージプロセスでは、Duplicate レコードを指す他のオブジェクトからのすべての受信参照 (添付ファイルを含む) が更新され、Main レコードに移動されます。Main レコードのフィールド値は変更されず、プロセスが完了すると Duplicate レコードが削除されます。
Merge Records API は、Enable Merges が設定されており、API 呼び出しを実行するユーザがアプリケーション: オブジェクト: Merge Records 権限を持っているオブジェクトでのみ機能します。Enable Merges 機能は、カスタムオブジェクトに対してのみオンに設定できます。
Pre-default Object Reference
オブジェクトレコードを作成またはコピーする際に、選択できる値が 1 つしかないオブジェクト参照フィールドが存在する場合がよくあります。これは、参照制限や (親子や兄弟関係などの) 制御フィールドなどといった設定がされているため、あるいは実際にその参照オブジェクト内にレコードが 1 つしか存在しないためです。より効率的なオブジェクトレコードの作成を可能にするため、オブジェクト参照フィールドに、Pre-default on non-required field when only one reference record is available という新しい属性が追加されました。
この属性を有効化すると、オブジェクトレコードの作成時またはコピー時に、選択可能なレコードが 1 つしかない場合は Vault でオブジェクト参照フィールドが事前入力されます。もちろんですが、選択可能なレコードが 1 つしかない場合は、(この新規属性が有効化されているか否かにかかわらず) Required のオブジェクト参照フィールドは常に事前入力されます。
注: この事前デフォルト入力動作は、レコード詳細ページ UI からレコードを作成した場合にのみ適用されるものであり、API やその他のバックグラウンドプロセスの初期化動作ではありません。
Related Record Audit Limits
この機能は、関連オブジェクトの数に制限を追加し、デフォルトの日付範囲を変更することで、関連レコード監査証跡のパフォーマンスを向上させます。ユーザは最大 10 個の関連オブジェクトを選択できるようになり、デフォルトの日付範囲は過去 1 か月に設定されます。
Expression Support for Long & Rich Text Fields
オブジェクトのロングテキストフィールドとリッチテキストフィールドを、Left() 関数、IsBlank() 関数、If() 関数と Case() 関数を用いた式で使用できるようになりました。
たとえば、MedInquiry Vault では、オブジェクトライフサイクルエントリアクションの式を使用して、Standard Response の既存のロングテキストまたはリッチテキストフィールドの値を Case Response にコピーできるようになりました。
24R1 より前は、一部の表現 (レポート式フィールドなど) でロングテキストフィールドとリッチテキストフィールドがサポートされていましたが、この機能強化により、表現を利用するすべての領域で確実にサポートされるようになりました。Left() でロングテキストフィールドを使用した場合は、テキストは最初の 1500 文字までしか表示されない点に注意してください。
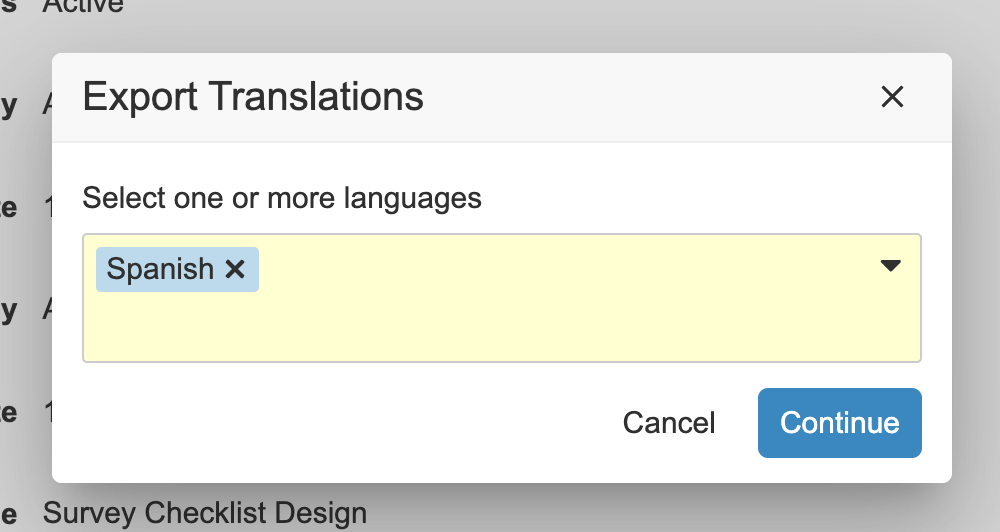
Checklist Design Translation
Vault はチェックリストデザインの翻訳をサポートするようになりました。管理者は、言語ごとに 1 つのチェックリストデザインを作成するのではなく、チェックリストデザインごとに、セクション、質問、回答の翻訳を含む複数の翻訳をインポートできるようになりました。
この機能強化により、チェックリストを活用するお客様は、作成/保守が必要な個別のチェックリストデザインの数が減り、グローバル組織でチェックリストデザインをより効率的に管理できるようになります。これにより、言語全体ですべての内容が単一のチェックリストデザインで維持されるため、レポート作成のトレーサビリティも向上します。
管理者は、言語ごとに CSV ファイルを 1 つ管理し、その言語の複数のチェックリストデザイン全体の翻訳をそこに入れることができます。ユーザアクションがチェックリストザインオブジェクトに追加され、管理者が翻訳ファイルをインポートおよびエクスポートできるようになりました。
これらのアクションはチェックリストデザインオブジェクトのライフサイクルでユーザアクションとして設定する必要があり、ユーザは権限セットでこれらのアクションにアクセスする必要があることに注意してください。
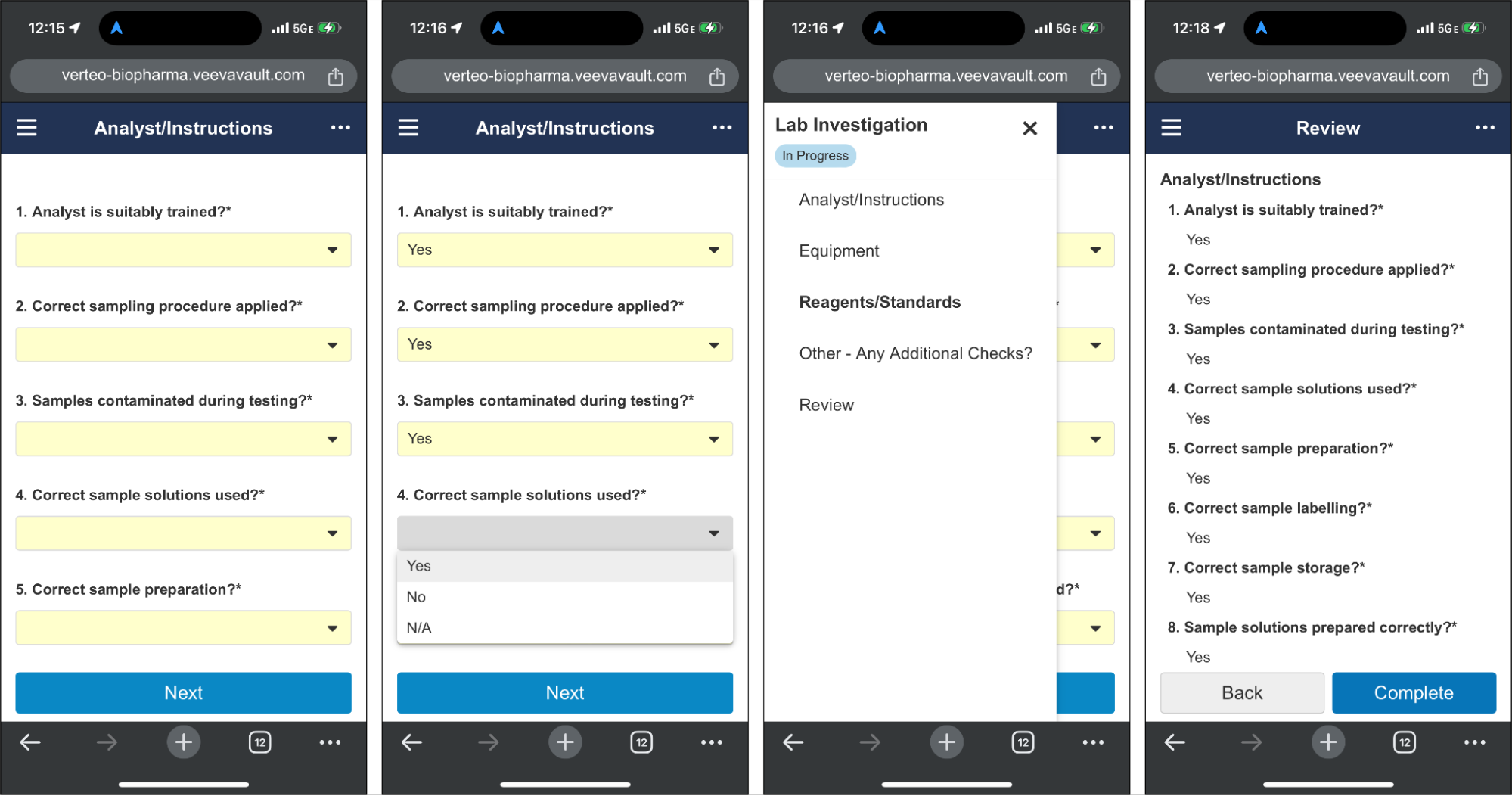
Mobile-Optimized Checklist UI for Respondents
コンピュータから離れた場所にいるチェックリストの回答者は、携帯電話のウェブブラウザを介してチェックリストにアクセスできるようになりました。この新しいモバイル向けに最適化されたビューを使用すると、ユーザはあらゆる種類の質問への回答、添付ファイルのアップロード、関連する Vault ドキュメントの追加、完了前の回答の確認を簡単に行えます。
次のチェックリストは、このリリースではモバイル最適化ビューをサポートしていません。
- QualityOne:
- Quality Event チェックリスト
- 監査チェックリスト
- クイズ
- Quality
- 所見を使用した監査チェックリスト
注: この機能が影響するのは、モバイル版ウェブブラウザで表示されるチェックリストです。このリリースでは、Vault Mobile アプリはチェックリストをサポートしていません。
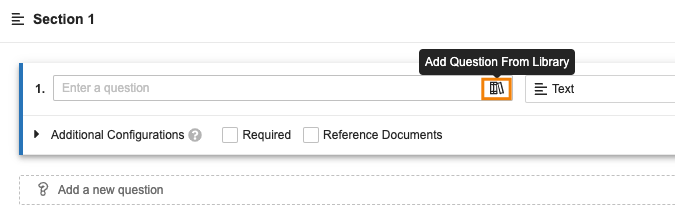
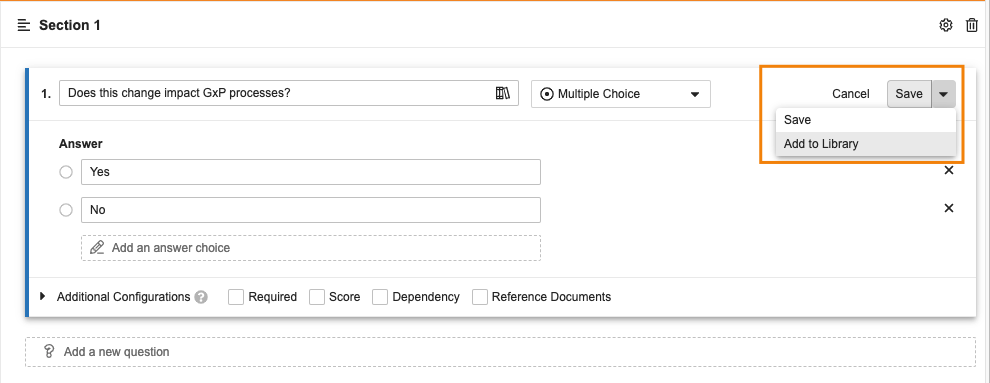
視覚的チェックリストデザイナーでのライブラリ質問のサポート
管理者は、ライブラリに追加された Questions から、新しい視覚的チェックリストデザイナーで質問を作成できるようになりました。
管理者は、視覚的チェックリストデザイナーで作成された Question からライブラリに質問を直接追加することもできるようになりました。
この機能強化により、お客様は視覚的チェックリストデザイナーの使いやすさを活用できるようになり、複数の Checklist Designs に適用できる質問を手動で再作成する必要がなくなります。
注: クイズ (Vault Training および Study Training で使用される) は、ライブラリ質問をサポートしていません。
詳細については、視覚的チェックリストデザイナーおよびライブラリ質問をご覧ください。
Checklists: Increased Character Limit for Answer Text in Answer Library Design
Answer Text フィールド (Answer Library Design オブジェクトと Available Answer オブジェクト) の文字数制限は、Available Answer Design オブジェクトの文字数制限との一貫性を保つために、128 文字から 255 文字に増加されました。
New Object Audit Fields in CSV Exports & API
オブジェクト監査 API (CSV および JSON) と Vault UI からのデータのエクスポート (CSV) に、フィールド、オブジェクト、新しい値、古い値の API 名を表示する新しいフィールドが含まれるようになりました。
これらの新しいフィールドにより、監査データと他の Vault データとの結合、外部システムでの監査データの追加分析が容易になります。
High Volume Object (HVO) Renamed to Raw Object
大容量オブジェクトは、Raw オブジェクトと呼ばれるようになりました。標準オブジェクトのスケーラビリティ向上を目指すため、大容量オブジェクトを生データの保存に使用される通常のデータベーステーブルのように扱うようになります。
これらのオブジェクトの目的をより明確にするために、ユーザーおよび開発者のドキュメント全体、Vault UI 自体の用語を更新し、このクラスのオブジェクトを未加工オブジェクトとして参照します。
現在、未加工オブジェクトには 10 億のレコード制限があります。さらに、オブジェクト MDL コンポーネントの data_store 属性に、raw 値が含まれるようになりました。この変更には下位互換性があり、high_volume は入力値として引き続きサポートされます。
共通 UI と検索
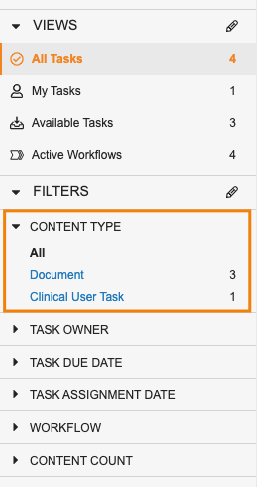
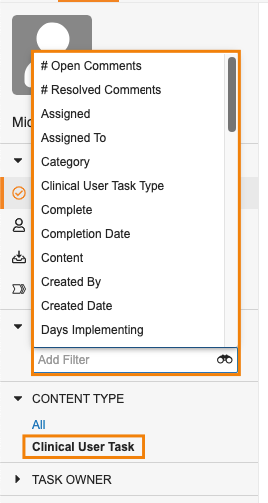
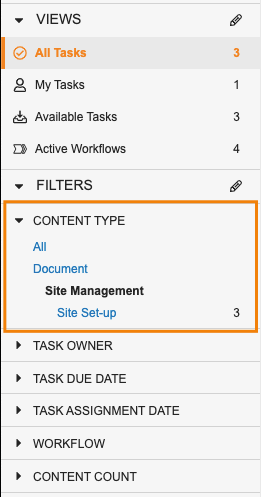
Task Filtering Enhancements
ホームタブで、マルチレコードワークフロータスクおよびマルチドキュメントワークフロータスクがより閲覧しやすくなりました。新しい Content Type フィルタを使用すると、各ワークフローにある複数の項目を含むタスクを、レガシータスクや単一項目タスクと並べて表示できます。新しい Content Count フィールドを使用すると、単一項目タスクと複数項目のタスクを区別できます。Content Type フィルタを使用すると、ドキュメントタスクのみ限定して表示したり、特定オブジェクトのタスクのみを表示したりできます。
コンテンツタイプで特定のオブジェクトまたはドキュメントタイプにフィルタリングした際に、ユーザはそのコンテンツタイプからさらに別のフィールドを選択して、タスクのフィルタリングをさらに続けることもできます。
Content Type でドキュメントをフィルタリングする場合、Vault では特定のドキュメントタイプに基づいてさらにフィルタリングすることもできます。
さらに、ユーザはワークフロー名でフィルタリングすることができ、Workflow Name フィールドおよび Content Count フィールドが、フィルタもしくはタスクグリッドビューの列として利用できるようになりました。
Column Header Filters
ユーザは、列の見出しからドキュメントとレコードを直接フィルタリングできるようになりました。
列の見出しフィルタは、選択リスト、オブジェクト参照、数値、日付、はい/いいえ、ユーザ、および標準フィールド (ドキュメントタイプ、ステータス、ライフサイクルなど) に対して使用でき、次の場所で使用できます。
- オブジェクトタブ
- ドキュメントタブ (グリッドビュー)
- 企業管理者内のオブジェクト
- 拡張検索セクション
この機能強化は、Vault の左側にあるフィルタオプションと連携して機能します。ユーザは、表示された列に基づいてより簡単にフィルタを適用できるようになりました。一方、左側のフィルタは、列として表示されない情報にフィルタを適用したり、実際には表示できる列ではない特殊なフィルタ (Role および Steady State Only など) を使用したりする場合に便利です。
Vault でのフィルタリングの詳細については、Vault ヘルプのフィールドおよびロールのフィルタリングをご覧ください。
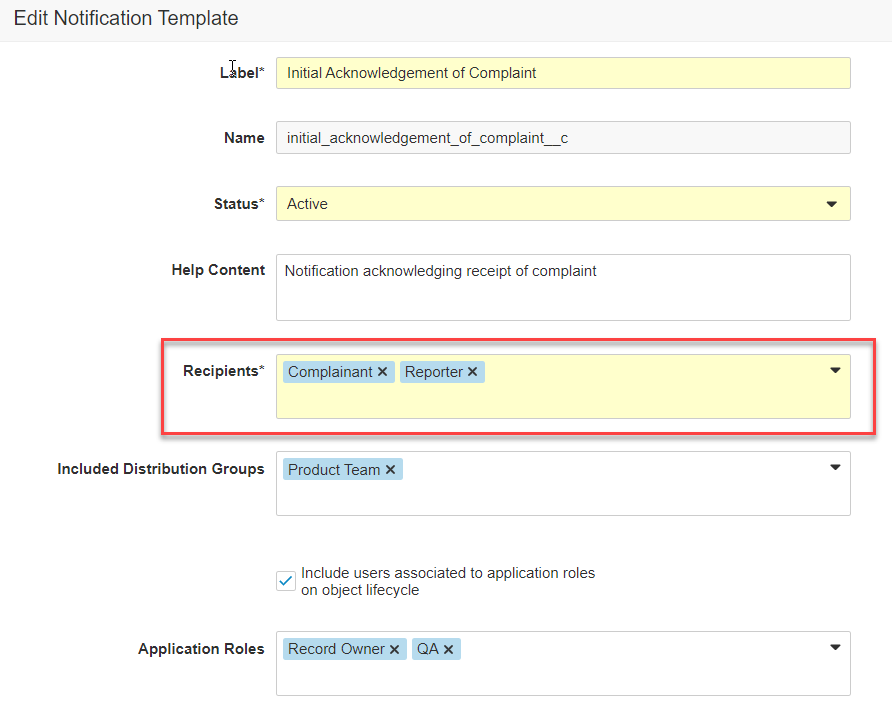
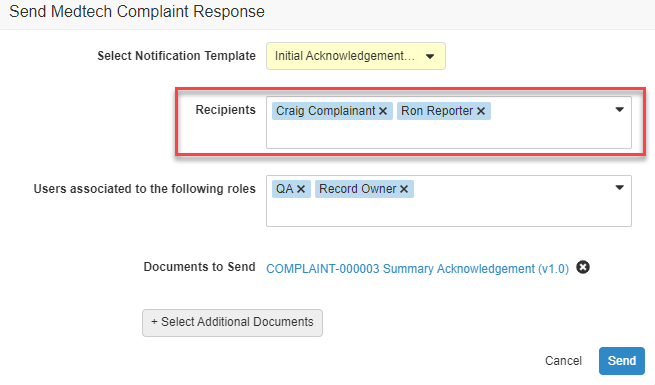
Notification Categories & Email Preferences
通知テンプレート設定の一環として、管理者はこれらの通知を電子メールで送信する頻度を具体的に定義できるようになりました。通知は、[すべての発生時]、[概要]として、または [送信しない] (つまりアプリ内通知のみのように扱われる) の頻度で電子メールを送信するように設定できます。
これはまた、通知カテゴリを許可する Vault 通知の機能強化でもあります。通知カテゴリでは、Vault の通知ページで通知をグループ化する方法や、概要メール内で通知をグループ化する方法を制御できます。管理者は、独自の通知カテゴリを定義し、通知テンプレートにカテゴリを割り当てることができるようになりました。
これらの変更により、ユーザーは、重要で時間的制約のある通知を見逃すことなく、通知ノイズを減らしながら、適切な方法と適切なタイミングで適切な通知を受け取ることができます。
この機能のリリース時に電子メールの頻度の継続性を確保するために、すべての既存の通知テンプレートの電子メール設定値は、ユーザー オブジェクトの対応するフィールドに設定されているデフォルト値と一致します。デフォルトとは異なる値を持つユーザー レコードについては、その値が維持され、尊重されます (これらのユーザーには頻度の変更は表示されません)。
My Vaults Card Layout Enhancements
マイ Vaults ページを利用して 1 つのアカウントから複数の Vault にアクセスするユーザには、カードレイアウトが、スクロール可能な 1 ページで最大 50 個の Vault をサポートするようになったことがわかります。24R1 より前は、ユーザが 12 個を超える Vault にアクセスできる場合でも、マイ Vaults ページには 1 ページあたり 12 個の Vault しか表示されませんでした。
1 ページに最大 50 個の Vault を表示できることで、ユーザは複数のページを移動することなく、必要な Vault に簡単に移動できるようになります。
さらに、Vault はユーザのウィンドウのサイズに基づいて 1 行にできるだけ多くのカードを自動的に表示し、カードの幅を広げて画面全体が活用されるようにします。
Additional Supported Time Zones
Vault は、最新の標準に従ってサポートするタイムゾーンを 229 個追加しました。世界中のユーザは、ユーザインターフェースに日付と時間が正しく表示されるように、自分の場所に最適なタイムゾーンを選択できます。
最新の標準に準拠するために 8 つのタイムゾーンラベルが更新されました。
Outbound Email Domains: Support Multiple Root Domains
Vault では、medinfo@verteo.com のような Veeva 以外のメールアドレスを使用して、送信メールを外部受信者に送信することができます。この機能は、MedInquiry Vaults (症例回答メール用)、QMS Vault (外部通知の送信用)、および Study Startup Vaults (実行可能性調査送信用) 内で利用および設定が可能です。
このリリースでは、管理者は最大 3 つのルートドメイン (verteo.comなど) と 5 つの電子メールドメインを作成できます。多くの企業(特に同じ組織の傘内に子会社がある場合)では、組織内の複数のドメインを活用しています。この機能強化により、お客様はこれらの異なるドメインの送信メールを使用できます。
この変更を容易にするために、送信メールアドレスには新しい Root Domains セクションがあり、企業は必要に応じて追加のサブドメインを設定できます。
Version Created By & Date Filters
既存の (元のドキュメントの作成者と作成日でフィルタリングする) Created By フィルタと Created Date フィルタに加えて、ユーザは最新バージョンの作成者、または最新バージョンの作成日でフィルタリングできるようになりました。
Vault は、ユーザにアクセス権限がある最新バージョンのみを返すため、これらのフィルタでは条件を満たす過去バージョンが結果表示されない点に注意してください。
この機能は、現在ドキュメントを所有し新しいバージョンの作成を担当するユーザが、ドキュメントの元の作成者と異なる場合に特に役立ちます。
Rename All Tabs to All
Tab Collections を設定した管理者は、ユーザがすべてのタブを表示するオプションを選択するのではなく、これらのオプションに最初にアクセスすることを望んでいる可能性があります。
そのため、そのオプションをタブコレクションのリストの一番下に移動し、単に All とラベル付けし直し、左側に global アイコンを付けて、ユーザがあなたの設定したタブコレクションと区別できるようにしました。
Business Admin および Admin オプション (一部のユーザにのみ表示されます) は分割線の下に表示され、All オプションの下に表示されるようになりました。
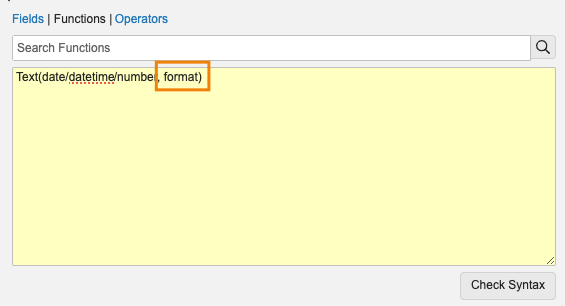
Add Spaces and Characters to Text with Text() Function
24R1 では、Text() 関数でテキストデータタイプを受け入れ、文字の挿入やスペースの追加などの単純なフォーマット設定を適用できるようにすることで、Vault 式を使用してテキストフィールドをフォーマット化する方法を強化しています。
現在、テキストフィールドが Text() 関数にパスされると、常に同じテキストが返されます。24R1 では、管理者はオプションの 2 番目のパラメータを Text() に追加できます。たとえば、Text("1800FLOWERS", "A-AAA-AAAAAAA") は、"1-800-FLOWERS" を返すようになりました。
レポートと式
機能強化されたダッシュボードタブ
ユーザは、ダッシュボードタブの検索とフィルタ、カスタムビューの作成と保存、並べ替え可能な表形式レイアウトの活用にアクセスできるようになります。この機能は、Vault の他の領域でダッシュボードの検索や整理に使用できる使い慣れたオプションをユーザに提供することで、ダッシュボードへのアクセスのエクスペリエンスを向上させます。
お客様がダッシュボードを識別して並べ替えるのに役立ついくつかの新しいフィールドがダッシュボードに追加されました。これには、ダッシュボードをより適切に整理するためのタグを追加する機能も含まれます。
この変更を有効にするために、既存のすべてのダッシュボードがオブジェクトに移行されます。ユーザエクスペリエンスの観点からは、既存のダッシュボードに変更はありません。
この機能強化により、標準の共有設定セクション、標準のダッシュボード管理者グループとダッシュボード所有者グループを使用して、レポート作成と一致するダッシュボードのセキュリティ/共有も実現します。
ダッシュボード管理者グループには、システム管理者、企業管理者、およびダッシュボードの管理権限を持つカスタムセキュリティプロファイルが含まれています。ダッシュボード所有者グループには、Vault 所有者が含まれています。
ダッシュボードの Read、Create、Edit、Delete を制御する新しいオブジェクト権限も権限セットで利用可能になります。
今後は、ユーザにダッシュボードの Read、Create、Edit、Delete アクセス権を付与するときに、これらのオブジェクト権限を新しい権限セットに使用する必要があります。
既存のダッシュボード権限はすべて維持されます。たとえば、権限セットのアプリケーションタブで、ダッシュボードおよびレポートの Read のチェックボックスがオンになっている場合、その権限セットでは、Dashboard オブジェクトの Read のチェックボックスが自動的にオンになります。
詳細については、ダッシュボードの作成と編集およびダッシュボードの表示と共有をご覧ください。
Table Chart
Vault では、ダッシュボードのコンポーネントタイプとして新たに表チャートがサポートされるようになりました。グループ化されていないレポート、または 1 レベルのみのグループ化が適用されているレポートは、すべて表チャートに変換できるようになりました。
この機能強化により、お客様が Vault ダッシュボードで情報を視覚化するために使用できる選択肢が広がります。特に表チャートを使用すると、各レポートをクリックして詳細を確認する手間なく、元のレポートからより多くのデータポイントをダッシュボード上に表示できます。
表チャートの並べ替えとフォーマットは、元のレポートの並べ替え順序とフォーマットによって決まります。
表チャートは、最大 200 行 (200 グループ) をサポートしています。表チャート作成時に、ユーザは最大 10 個のフィールドを表の列として含めることができます。また、閲覧者が元のレポートにアクセスしやすいよう、クリック可能なレポート名を追加することもできます。
詳細に関しては、ダッシュボードをご覧ください。
Report Edit Columns Modal Enhancement
レポート編集中、「列の編集」ダイアログボックス使用時に表示されるユーザインターフェースが、次のように改善されました。
- ダイアログボックスの大きさを拡張
- 長いフィールド名はテキストを折り返し表示
- マウスポインターをフィールド名の上に置くと背景が暗転
24R1 より前はダイアログボックスが小さかったため、フィールド名が長いと、ユーザは含めたい情報を特定しづらい状態でした。この機能強化によりユーザは、適切な情報をより簡単かつ迅速に特定し、レポート内に適用できるようになりました。
Union Report Type Enhancement
Union レポートを使用する場合、Vault は結合されているオブジェクトのすべてのオブジェクトフィールドへのアクセスをサポートするようになりました。これにより、管理者は、すべてのフィールドを結合することなく、Union レポートタイプを構築することで、ユーザに関連するすべてのデータへのアクセスを可能にすることができます。また、フィールドを結合されたものとして扱う場合と、一部のフィールドをオブジェクトごとに一意として扱う場合の両方により、柔軟性が得られます。
Union レポートビューで数式フィールドがサポートされるようになりました。これがサポートされたことで、お客様はさまざまなデータ型のフィールドを組み合わせて、最終レポート用の新しい列を作成できます。これにより、管理者はテキストと選択リスト、さまざまな選択リスト、さまざまなオブジェクトを組み合わせることができます。単一の列で異なる ID またはレコード名を組み合わせると、それらをマルチパスレポートタイプの別のビューと結合できるようになります。
24R1 での Union レポートタイプの追加の機能強化には、次のようなものもあります。
- 結合されたオブジェクト・ドキュメント参照フィールドがハイパーリンクとして表示されるようになりました。
- どのオブジェクトに属しているかを理解しやすくするために、オブジェクトラベルがフィールドとして追加されました。
- グループ化を含む Union レポートビューを、マルチパスレポートの他のビューと組み合わせることができるようになりました。
- Union レポートビューで結果を Excel にエクスポートできるようになりました。
- Union レポートを Excel にエクスポートするときに、ロングテキストフィールドとリッチテキストフィールドがサポートされるようになりました。
詳細については、Union レポートタイプをご覧ください。
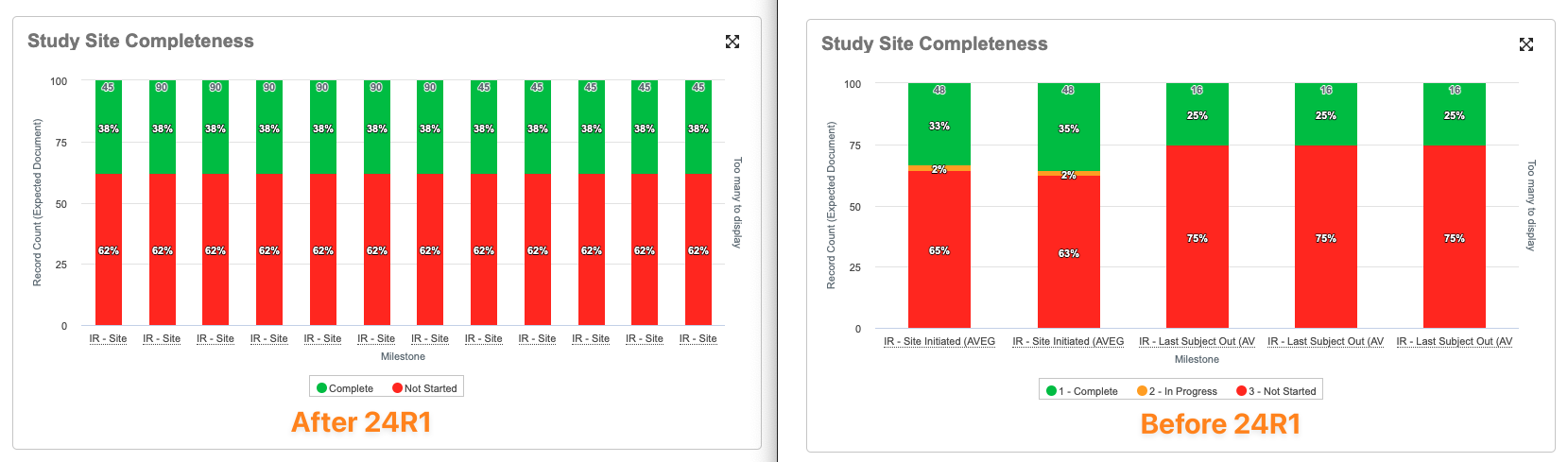
Expand Chart Enhancement
棒グラフ、折れ線グラフ、管理図では、ダッシュボードセルごとに 1 つのデータポイントが表示されるようになり、ダッシュボードコンポーネントのサイズに基づいてより多くの情報をユーザに簡単に表示できるようになります。たとえば、7 セル幅に拡張されたグラフには 7 つのデータポイントが表示され、11 セルに拡張されたグラフには 11 個のデータポイントが表示されます。
24R1 より前は、これらのグラフでは、グラフの対象となるセルの数に関係なく、ページの幅全体に拡張しない限り、常に 5 つのデータポイントが表示されていました。
ページの幅全体に拡張されたグラフの動作に変更はありません。
Limit Increases for Reports
Vault では、使用できる全体的なフラッシュレポート数と、マルチパスレポートで使用できる数式フィールド数が増えました。
フラッシュレポートの合計上限数は、(200 から) 400 に引き上げられました。
マルチパスレポートで使用可能な数式フィールドの合計上限数は、(3 から) 10 に引き上げられました。
詳細に関しては、フラッシュレポートおよびマルチパスレポートをご覧ください。
セキュリティ
Increased Field Limit for User Role Setup Objects
「ユーザロール設定」クラスのオブジェクト (動的アクセスコントロールによって、オブジェクトのロールに対してユーザを自動的に割り当てるために使用するタイプのオブジェクト) については、使用できる一致フィールド最大数が 5 から 6 に引き上げられました。この実装は、お客様から多く寄せられたリクエストに応え、ユーザとロールの自動マッチングをより複雑にする必要がある場合や、異なる一致フィールドのある多数のオブジェクト間で同じユーザロール設定を使用する場合に対応したものです。
注: この制限数には、標準フィールドはカウントされません。
ツール
External URL Reverse IP Lookup Change
外部 URL ジョブは、Veeva Vault に関連付けられた IP アドレスから送信されるようになりました。現在ドメインによるリストを許可しているお客様は、*.veevavault.com ドメインをサポートするようにルールを更新する必要があります。
詳細に関しては、外部 URL 呼び出しをご覧ください。
Sandbox Allowance Warning
Sandbox の制限数を超えて一時的な Sandbox の追加が許可されている本番環境 Vault (多くの場合、特定の期間のみ Vault を必要とするプロジェクト) に関しては、利用規約の更新に伴い、2024 年 9 月 以降、Veeva 社は有効期限を超過した Sandbox を削除する権利を有します。
24R1 リリース以降、お客様各位にこのような変更への対応をご検討いただくための機会として、管理者ユーザが Sandbox 管理ページにアクセスした際に、今後のより積極的な Sandbox クリーンアップ作業に関するメッセージが表示されます。
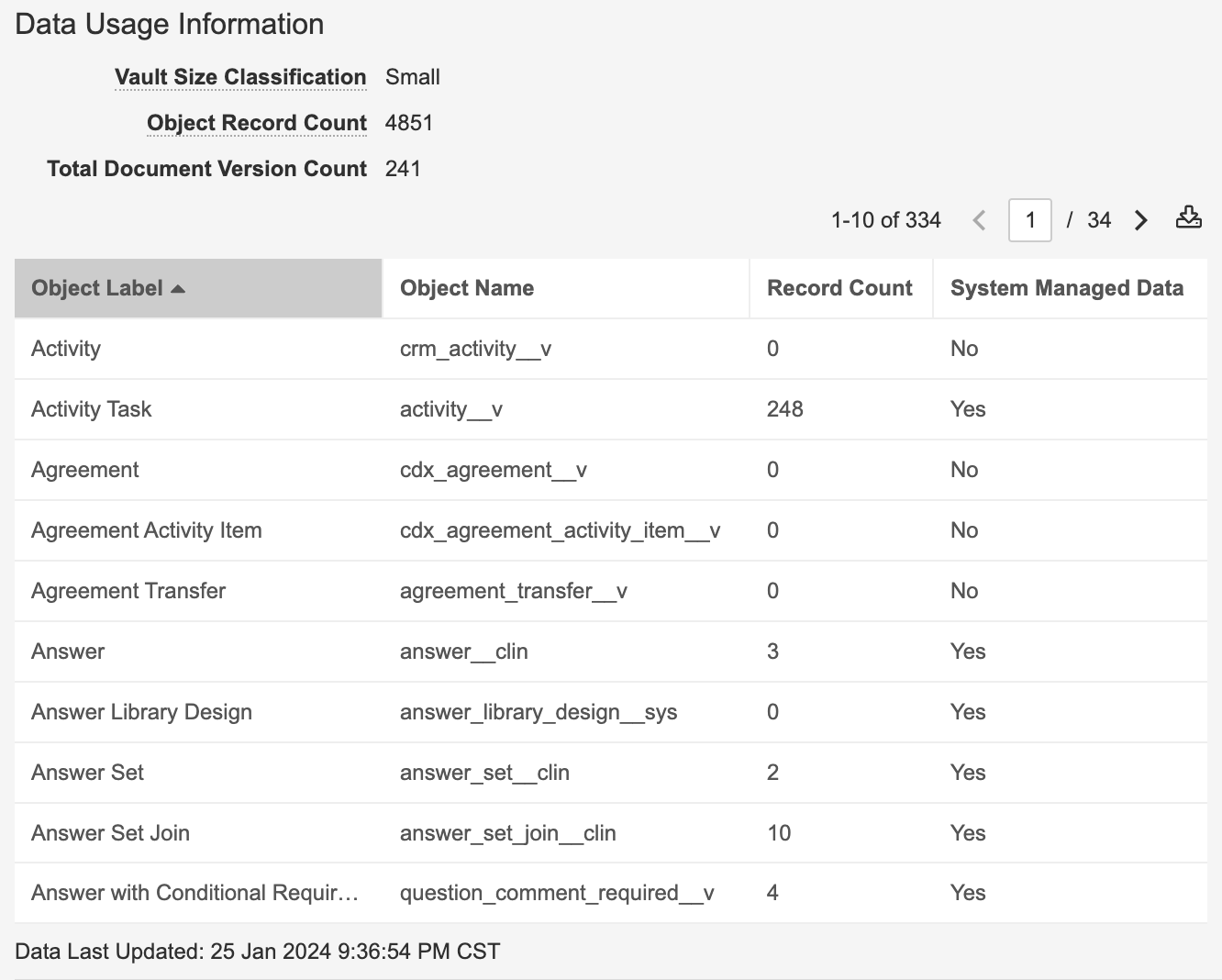
Vault Data Usage Information
Vault では、Admin > Settings > General Settings に Data Usage Information セクションが表示されるようになりました。
これにより、管理者は、エクスポート可能なオブジェクトレコードの内訳など、Vault のサイズを簡単に確認できるようになります。Vault には、システム管理データを除く Object Record Count と Total Document Version Count も表示されます。Data Usage Information に表示される情報は、オブジェクトレコード数に対して継続的に更新され、Total Document Version Count は毎日更新されます。
System Managed Data とは、Sandbox サイズなどの制限にカウントされないデータを示す分類であり、ユーザ管理データを Vault クローン作成に含まれる設定データと区別します。たとえば、RIM では、Controlled Vocabulary は RIM のコア機能の一部であるため、設定データとみなされます (つまり、Sandbox サイズにはカウントされません)。23R3 リリース以降、Vault では設定データをカウントしなくなりましたが、ユーザ管理とみなされるものと、みなされないものを簡単に確認できませんでした。
Data Usage Information には、ドキュメントとデータの量に基づいて Vault のサイズを分類する Vault Size Classification も表示されます。ここでのサイズは、Sandbox サイズに使用されるものと同じです。
Sandbox サイズの調整が必要なシナリオでは、管理者はこの機能強化により、対処が必要な内容の詳細を簡単に特定できるようになります。
詳細については、Sandbox サイズをご覧ください。
Vault Loader のドキュメントのダウンロード制限が 10,000 に増加
Vault Loader を使用してドキュメントソースファイルとレンディションファイルをエクスポートする時の制限が、2,000 ファイルから 10,000 ファイルに引き上げられました。この変更はすべての Vault に適用されており、その他の機能の変更は含まれていません。
Migration Packages Support for Record Migration Mode Update & Upsert
弊社のお客様は、Vault アプリケーションを段階的に導入することが多く、これは Vault と外部システムの両方に存在する重要な共有データが一時的に存在することを意味します。これらのシステムが Vault アプリケーションに統合された後の実装で、その共有データに設定とデータ変更を行う必要があります。このプロセスはデータ移行として処理され、これをサポートするために、移行モード機能がオブジェクトレコードの作成のみから、アップデートとアップサートにも適用されるようになりました。
Scheduled Data Export Support for AWS Bucket Policy
Scheduled Data Export 機能では、24R1 以前のアクセスコントロールリスト (ACL) が使用されていましたが、代わりに S3 バケットに推奨される AWS バケットポリシーアプローチに対応するようになりました。
ACL を無効にすると、バケット内のオブジェクトを誰がアップロードしたかに関わらず、ポリシーを使用してバケット内のすべてのオブジェクトへのアクセスをより簡単に制御できます。バケット所有権についての詳細に関しては、AWS S3 ドキュメンテーションを参照してください。
Enhancements to Query Field Type
フィールド規則で、フィールド規則のクエリフィールドとは異なる SELECT フィールドを使用できるようになりました。この機能を使用すると、アプリ開発者/顧客は、クエリフィールドをオーバーロードせずに必要なフィールド値を取得できます。
これを可能にするために、新しい Query Field Select 属性 (query_field_select) を導入しています。
この属性は、Vault Connectionsで使用されるフィールド規則では推奨されますが、ローカル接続または外部接続のフィールド規則には必要ありません。
クエリオブジェクト規則の詳細については、こちらを参照してください。
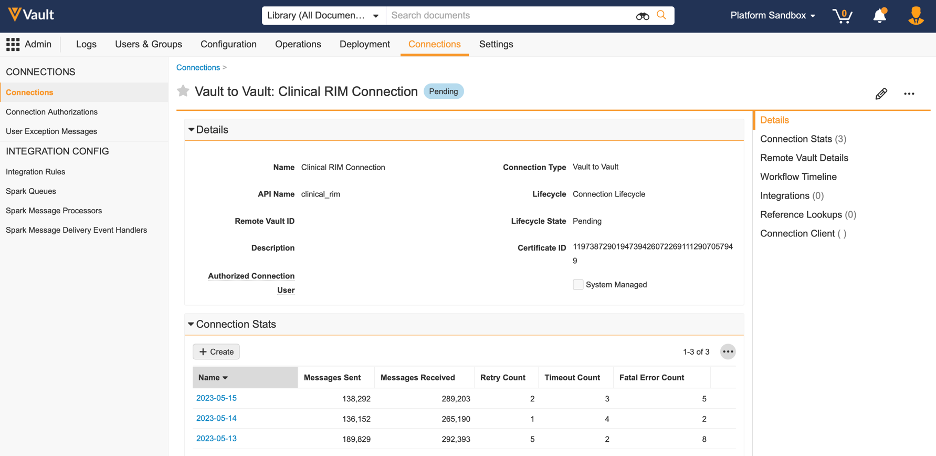
Connection Stats
管理者は、送受信されたメッセージ、失敗、再試行、経過時間を含む、接続 (Vault 間または外部) の毎日のパフォーマンス指標にアクセスできるようになりました。
以前のリリースでは、Vault は既に、毎日のパフォーマンス統計の一部として接続メッセージ (受信または送信) の合計数を提供していましたが、接続と統合のトラブルシューティングと監視を行うには、接続ごとにさらに詳細な内容が必要になることがよくあります。
このリリースでは、接続ごとおよび統合ごとに毎日の統計を保存する新しいオブジェクトが導入されました。このオブジェクトは、一般にカスタムフィールド、トリガー、カスタマイズをサポートしません。
このオブジェクトはデフォルトで企業管理者で使用できますが、お客様は新しいレポートタイプを作成して接続の詳細を監視することもできます。
Connection Stats の新しい関連セクションは、コネクションでもデフォルトで利用できるため、これらの統計情報をコネクションごとに簡単に表示できます。
Vault Connections の詳細については、Vault ヘルプ (接続の作成および管理) をご覧ください。
Vault Java SDK: Record Triggers Return Correct Fields for New Object Type
この機能の説明については、「開発者ポータル」をご覧ください。
Vault Mobile
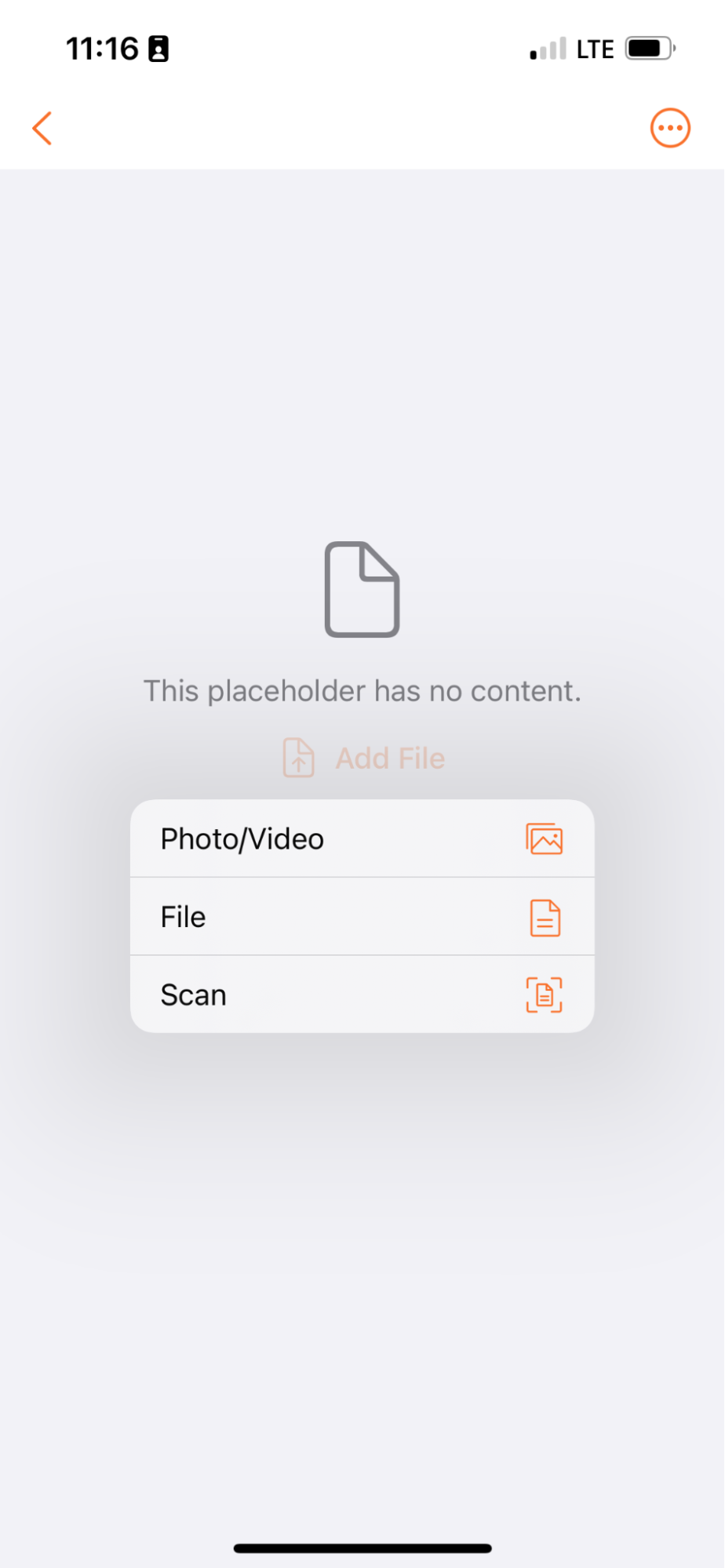
Add Content to Placeholders
Vault Mobile では、ユーザーがドキュメントをスキャンしたり、Vault の既存のプレースホルダーにアップロードしたりできるようになり、外出先でのコンテンツ投稿機能がさらに拡張されました。
たとえば、ユーザーはプレースホルダーを使用してドキュメントを必要とする別のユーザーにタスクを送信することができます。タスクを受け取ったユーザーは、ドキュメントをスキャンしてそのプレースホルダーに置き、タスクを完了することができます。
これはまた、ユーザーがコンテンツを分類済みとして投稿するメカニズムも提供します (プレースホルダーには既に分類が割り当てられているため)。
Apply Study Information to New Documents (Clinical Vaults Only)
Clinical Operations Vault のユーザは、Vault Mobile でスキャンされたドキュメントに Study、Study Country、Study Site の各フィールドを適用するオプションが利用できるようになりました。
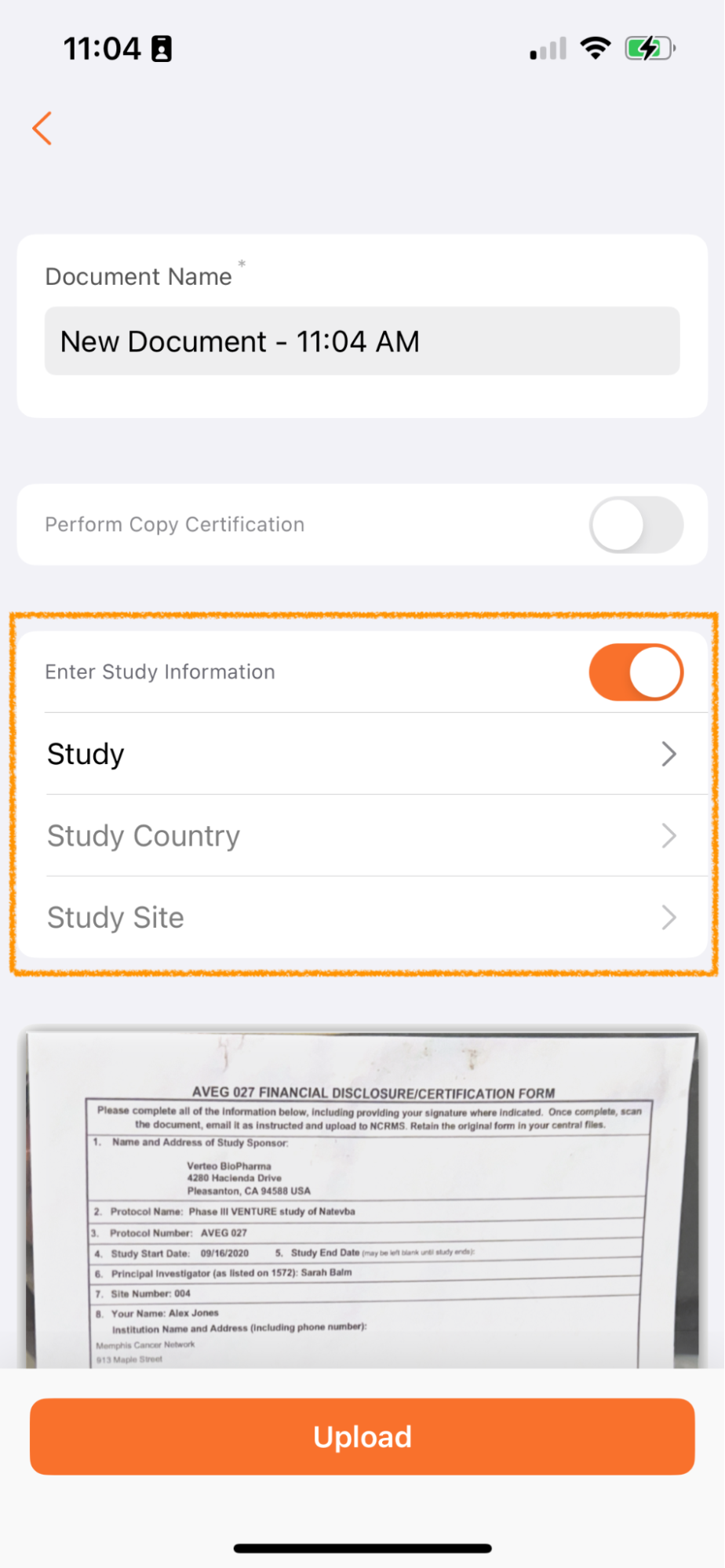
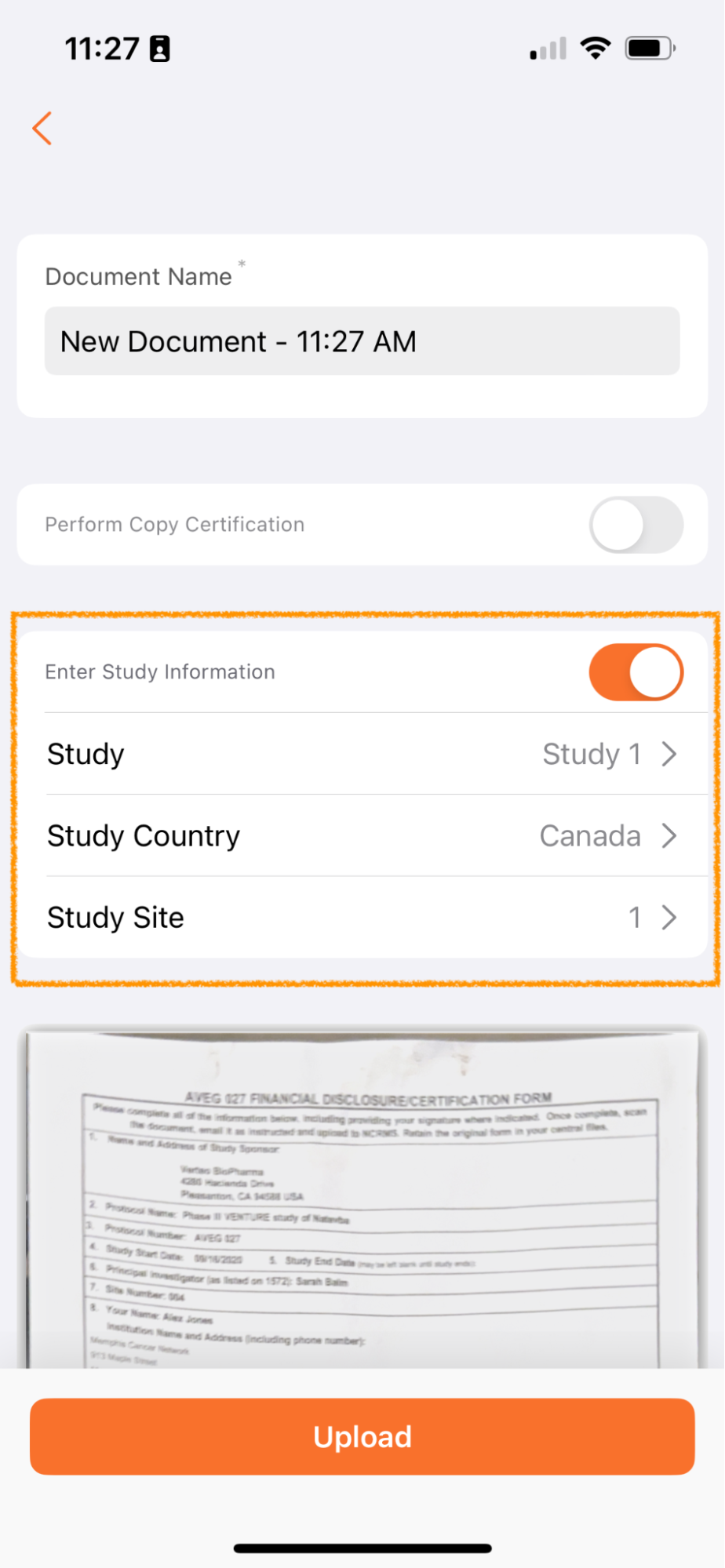
Vault Mobile 経由でアップロードされたドキュメントは未分類になり、ドキュメント受信箱に追加されます。多くのお客様は、治験ごとに受信箱のドキュメント処理を管理しています - たとえば、治験 ABC のドキュメント処理に重点を置いたチームと、治験 XYZ のドキュメント処理に重点を置いた別のチームが存在する場合があります。
アップロード時にこのメタデータを追加できるようにすることで、ドキュメント受信箱を管理するユーザーは、職務をより簡単に分割し、自分の役割と担当する治験に適したドキュメントに集中することができます。
この機能は、スキャン機能を使用している場合にのみ適用され、デバイスを介して Vault Mobile にアップロードされたファイルには適用されません。
治験、治験実施国、および 治験実施施設 フィールドは、未分類 ドキュメント タイプ (または ベース ドキュメントタイプ) に共有フィールドとして存在する場合に使用できます。これらのフィールドの表示は、Vault の構成に応じて、フィールドの依存関係によっても異なります。
アップロードするファイルの選択
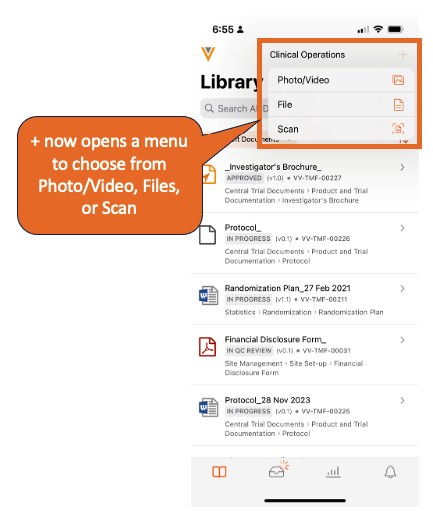
Vault Mobile で新しいドキュメントを作成する場合、ユーザは新しいドキュメントをスキャンするだけでなく、デバイスから既存のファイル/写真を選択できるようになりました。iOS デバイスの場合、オプションはファイル、写真/ビデオ、スキャンになります。Android デバイスの場合、オプションは ファイル、スキャンになります。
24R1 より前は、Vault Mobile ユーザは通常、モバイルアプリ内からドキュメントページをスキャンして、新しい未分類のドキュメントを作成していました。携帯電話に既にファイルがある (何もスキャンする必要がない) 一部のユーザにとって、アプリ内からアップロードする既存のファイルを選択する簡単な方法はありません。
別のアプリから Vault にファイルを共有できる既存の Vault への共有機能がありますが、別のアプリから共有する前に Vault Mobile にログインする必要があるため、シームレスなユーザエクスペリエンスが低下します。
携帯電話からファイルを選択できるようにすることで、Vault Mobile でのドキュメント作成エクスペリエンスが完成し、ウェブユーザエクスペリエンスに匹敵するユーザエクスペリエンスが提供されます。
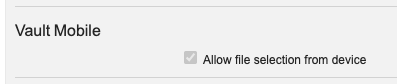
この機能強化は自動的にオンになりますが、管理者は Admin > General Settings > Settings でこれを無効にできます。
これを無効にすると、写真やファイルからのアップロードはブロックされますが、ユーザは新しいファイルをスキャンできます。
Language Support
Vault Mobile アプリのテキストは、常に英語で表示されるのではなく、ユーザの Vault 言語とロケール設定を反映するようになりました。
この機能強化により、世界中のユーザが Vault Mobile をより適切に活用できるようになり、モバイルアプリのユーザエクスペリエンスがウェブユーザエクスペリエンスと一致するようになります。
詳細に関しては、言語 & 地域設定をご覧ください。
Platform Data Model Changes
24R1 Platform データモデルの変更をご覧ください。
Vault Connections
Clinical Operations と CDMS の接続
Clinical Operations-EDC Connection: Complex Clinical Trials
この機能によって、Vault CTMS での複雑な臨床試験の導入に対応するために、Clinical Operations と CDMS との接続が強化されます。複雑な臨床試験で、お客様は、マイルストーン、メトリクス、被験者登録の追跡に使用できるさまざまな被験者グループ (アーム、コホート、サブスタディなど) を作成できます。
新しいフィールド規則を使用して、サブジェクトグループの割り当てが CDMS から Vault CTMS に転送されます。2つのシステム間で被験者グループを手動リンクすると、CDMS の被験者に被験者グループのアサインメント、またはアーム、コホート、またはサブスタディがある場合、その接続で被験者グループは Vault CTMS の同じ治験の被験者グループアサインメントと照合されます。一致するものがある場合、Vault CTMS レコードの被験者グループアサインメントが更新されます。一致するものがない場合、接続はユーザー例外レコードを作成し、被験者グループアサインメントのリファレンスを変更せずに Vault CTMS レコードに残します。その他の新しい Clinical Operations の機能については、こちらをご覧ください。
Clinical Operations-EDC Connection: Subject Visit Method
治験の分散化が進んだことに対応して、23R2では Clinical Operations Vaults に被験者来院方法フィールドが追加され、来院方法 (オンサイトまたはリモート) に関する追加情報が追加されました。被験者来院方法は、さまざまな支払い額に対応するために料金表で使用され、監視対象の被験者来院でも追跡され、監視対象となる予想されるドキュメントに関する見解を CRA に提供します。
この機能により、Clinical Operations から CDMS への接続が強化され、CDMS から Vault Clinical への被験者来院方法の転送がサポートされます。
詳細に関しては、Clinical Operations to CDMS Connection をご覧ください。その他の新しい Clinical Operations の機能については、こちらをご覧ください。
Clinical Operations-ePRO Connection
Clinical Operations-ePRO Connection
この機能により、Clinical Operations Vault と ePRO Vault を接続できるようになりました。Clinical Operations Vault で作成され、かつ ePRO への転送フラグが設定されている治験は、関連するすべての治験実施国および治験実施施設を含め、ePRO Vault 上に自動作成されます。この接続により、Clinical Operations と ePRO で 2 重にデータを入力する手間が削減され、治験依頼者ならびに CRO にとってデータ作成が効率化されます。
LIMS と Quality の接続
LIMS-Quality Connection: Document Exchange
この機能によって、QualityDocs と LIMS Vault 間に新しい接続・統合ポイントが追加され、指定したドキュメントタイプがメジャー バージョン固定状態に達した時のクロスリンク自動作成がサポートされるようになりました。この接続の一環として、ドキュメントのクロスリンクは QualityDocs から LIMS へ、もしくは LIMS から QualityDocs へのいずれの方向でも送信できます。この接続により、定常状態ドキュメントのクロスリンクバージョン管理の自動化、および両 Vault 間でのメタデータ更新の自動化がサポートされます。
Quality と RIM の接続
Quality と RIM の接続: ドキュメント交換
この機能により、指定したタイプ/サブタイプ/分類のドキュメントがメジャーバージョン固定状態に達した時に、Quality Vault と Regulatory Vault 間のドキュメントが自動クロスリンク処理されるようになります。これにより、Quality Vault 上で作成された提出に含める必要があるドキュメントを、CrossLink として RIM Vault に自動転送できるようになります。さらにこの接続により、たとえば製品仕様書や、ラベル表示/アートワークドキュメント、CMC ドキュメントなどの RIM Vault で作成されたドキュメントを、CrossLink として Quality Vault に自動転送することもできます。これにより、信頼できるデータソースの一元化が保証され、クロスリンクの手動作成が不要となります。CrossLink は、ドキュメントの初回バージョンが固定状態になった時にシステムによって自動生成され、そのドキュメントの新しいメジャーバージョンが固定状態に達するたびに自動的にバージョンアップされます。新しいメジャーバージョンが作成されると、それ以前の CrossLink バージョンは過去版になります。既存のドキュメントを廃版にすると、既存のクロスリンクも自動的に廃版になります。
Quality-RIM Connection: Updated Error Message when Executing UpdateRimChangeDetails Action
Update RIM Change Details レコードアクションが Impact Assessment レコードのユーザアクションとして設定されている場合、そのアクションが常に表示されるようになりました。以前、このレコードアクションは、Impact Assessment レコードの RIM Event ID フィールドが入力されている場合にのみ表示されていました。RIM Event ID と Quality Event の両方、または Change Control フィールドが入力されていない場合、ユーザはエラーを受け取るようになりました。その他の新しい Quality 機能 についてはこちら、Regulatory 機能についてはこちらをご覧ください。
RIM と PromoMats の接続
RIM-PromoMats Connection: CrossLink Document Transfer Integration
RIM-PromoMats Connection で、AdPromo サブミッションをサポートするために、固定状態ドキュメントを転送できるようになりました。設定が完了すると、接続を使用するお客様は、PromoMats にアップロードするために RIM の現在使用中のラベル付けドキュメントをダウンロードする必要がなくなります。これにより、エンドユーザの効率が向上し、AdPromo サブミッションに古いラベル付けドキュメントが含まれるリスクが軽減されます。
管理者は、ドキュメントタイプグループと統合ルール (クエリオブジェクトルール、フィールドルール、および参照ルックアップ) に基づいて転送するドキュメントタイプを設定できます。デフォルトでは、機能の設定後に次の固定状態に達した場合にのみ、統合によって固定状態のドキュメント転送が開始されます。その他の新しい Regulatory 機能についてはこちら、Commercial 機能についてはこちらをご覧ください。
RIM-PromoMats Connection Performance Improvements
この機能により、RIM - PromoMats Connection パフォーマンスが向上し、あるサブミッションでの検証エラーで他のサブミッションがブロックされることがなくなります。その他の新しい Regulatory 機能についてはこちら、Commercial 機能についてはこちらをご覧ください。
Medical と Safety の接続
Medical-Safety Vault Connection: Adverse Event Reports
この機能により、お客様は Vault Medical - MedInquiry で発生した潜在的な有害事象を、Vault 間接続を介して Vault Safety と共有できるようになります。
このコネクションで、MedInquiry の Adverse Events レコードは、必要な患者、報告者、製品、および事象情報とともに Vault Safety と共有され、安全性ケースの処理とレポート作成用の Inbox Items レコードとして作成されます。インボックスアイテムの作成後、調整情報は Vault Safety から MedInquiry に共有されます。Vault Safety のインボックスアイテムに対するさらなるアクションにより、Safety から MedInquiry への症例情報 (Case Number および Safety のインボックスアイテムの結果を含む) の共有がトリガーされます。その他の新しい Medical の機能についてはこちら、Safety の機能についてはこちらをご覧ください。
詳細については、Medical と Safety の Vault Connection、Medical と Safety の Vault Connection の使用方法、およびこの Vault Connection の有効化をご覧ください。
PromoMats と Medical の接続
PromoMats と Medical の接続
この機能により、固定状態のドキュメントとそれに関連するアンカーを Medical Vault と PromoMats Vault の間で転送できるようになります。たとえば、Medical Vault に保存されている医学文献の参考資料を PromoMats Vault に転送して、販促/マーケティング資料で行われた主張の裏付けに役立てることができます。
リリース時には、この双方向接続には、お客様が追加、変更、または無効化できるデフォルト設定が含まれます。設定可能な要素には、PromoMats と Medical の接続ドキュメントタイプグループを含むドキュメントタイプと統合ルール (クエリオブジェクトルール、フィールドルール、および参照ルックアップ) が含まれます。
Medical Vault と PromoMats Vault の間でこれらのドキュメントとデータを転送することにより、エラーやコンプライアンスのリスクにつながるドキュメントをある Vault からダウンロードして別の Vault にインポートする必要がなくなります。
参照ドキュメントの作成はクロスリンクによって自動化され、主要なドキュメントが組織内で唯一の信頼できる情報源を持つことが可能になり、重複ドキュメントが作成される可能性が減ります。その他の新しい Commercial の機能についてはこちら、Medical の機能についてはこちらをご覧ください。
Safety と Clinical Operations の接続
Safety-Clinical Operations Connection: Safety Letters
Safety から Clinical Operations への接続では、Vault Safety から Vault Clinical Operations への安全性レターの配布がサポートされるようになりました。これ以前は、医薬品安全性監視から Clinical Operations システムへの安全性レターの配布は手動で行われ、ミスも起こりがちでした。この機能により、安全性レターの配布が自動的かつ準拠した方法で行われます。
この機能は、Safety から Clinical Operations への接続が有効になっている Vault で自動的にオンになります。
その他の新しい Safety の機能についてはこちらをご覧ください。
詳細については、Safety から Clinical Operations への Vault Connection、およびこの Vault Connection の有効化をご覧ください。
Safety と EDC の接続
Safety-EDC Connection: Serious Adverse Event Reports
Safety と EDC の Vault Connection は、重篤な有害事象 (SAE) と、関連する被験者のケース情報の Vault EDC から Vault Safety への自動転送をサポートします。SAE は治験実施施設によって発見され、施設ユーザによって SAE 評価として EDC Vault に入力されます。これにより、症例処理とレポートのためにインボックスアイテムが Vault Safety に自動的に生成されます。この機能により、ユーザは遅延やコンプライアンスの問題を回避できるようになります。その他の新しい Safety の機能についてはこちらをご覧ください。
詳細については、Safety と EDC の Vault Connection、およびこの Vault Connection の有効化をご覧ください。
Safety と RIM の接続
Safety-RIM Vault Connection: Product Trade Names
Safety-RIM 製品接続により、関連する製品の商標名の詳細が Vault RIM から Vault Safety に転送されるようになりました。商標名の詳細が表示されるため、ユーザは採用プロセス中に正しい製品を選択することができます。この機能は、RIM と Safety の間で商標名の整合性を確保することにより、Safety と RIM の Vault Connection を強化します。
この機能は、Safety と RIM の接続が有効になっている Vault で自動的にオンになります。その他の新しい Safety の機能についてはこちら、Regulatory の機能についてはこちらをご覧ください。
詳細については、Safety-RIM Vault Connection をご覧ください。
Clinical Operations
以下の Release Notes に加えて、Vault CTMS、Vault eTMF、Veeva Site Connect、Vault Study Startup、および Vault Study Training Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモが提供されています。
Vault Connections セクションにリストされているいくつかの機能も、Clinical Operations アプリケーションファミリーに影響を与えます。
Clinical Operations の全アプリケーション
Complex Trials: Tracking Country & Site Participation
近年、臨床試験のデザインはますます複雑になり、施設、治験責任医師、スタッフの共通の中核を使用して、単一のマスタープロトコルの下で、複数の治療法、異なる用量、および多くの患者集団を試験するための柔軟性が組み込まれています。これらのデザインでは運用の時間とコストが効率化されますが、より複雑な事前計画プロセスも必要になります。この機能により、Clinical Operations Vault でより複雑な臨床試験を管理するためのサポートが追加されます。
主要な要素は次のとおりです。
- 既存の Study Arm オブジェクトに、Study Arm、Cohort、Study Part、Substudy、Study Element のオブジェクトタイプが含まれるようになりました。
- Subject、Monitored Subject、Issues オブジェクトのすべての Study Arm オブジェクトタイプに対する新しいオブジェクト参照フィールド
- Study Arm と Study Country、Study Site と Study Product 間の新しい結合オブジェクトが、以下の Study デザイン要素の使用状況を追跡するために追加されました
- 治験実施国の被験者グループ
- 治験実施施設の被験者グループ
- 治験製品の被験者グループ
- 治験実施国、治験実施施設、治験製品を追跡するために必要なレコードを作成する新しいアクション、Seed Subject Group Records。
Seed Subject Group Records アクションは、アクションが行われた時点の治験でのアクティブな治験実施国、治験実施施設、治験製品ごとのレコードを作成します。一度実施すると、新しいStudy Country、Study Site、Study Product レコードが追加されたときに、Vault は対応する Study Group 結合レコードを自動的に作成します。結合レコードには、関連レコード (Study Country、Study Site、Study Product) の標準ライフサイクル状態と一致する標準ライフサイクル状態があります。
これらの更新により、お客様は Clinical Operations Vault で治験対象グループを定義し、調査対象の製品や各グループに参加している国や施設を追跡できるようになりました。
Clinical Operations License Tracking by Application
この新しい自動オン機能により、Clinical Operations Vault での既存の eTMF ライセンス追跡が拡張され、追加の Clinical Operations アプリケーション (CTMS、Payments、SSU) のライセンス追跡が含まれるようになります。
アプリケーション全体のライセンス消費は、Vault 設定と、Vault データに基づいて Clinical Operations ライセンスを追跡する新しいオブジェクト (Study Site License) 内で管理者に表示されます。本番環境の Vault で消費がライセンス量を超えると、警告メッセージバナーが表示されます。
この新機能は、Sandbox 環境での消費数超過の警告を無視しますが、追跡は表示されます。
さらに、データのクリーンさを保証するために、この機能は、治験実施国の治験実施施設のレコードのステータスがアクティブ、移行中、またはアーカイブ済みの場合、治験実施国のステータスが 非アクティブのライフサイクル状態に移行するのを防ぎます。治験実施国を非アクティブのライフサイクル状態に移動するには、すべての治験実施国のライフサイクル状態が非アクティブである必要があります。この機能は今回のリリースから削除されました。
注: 契約した組織のライセンスに関する質問、ライセンスのニーズの変更、またはライセンスに関する矛盾については、Veeva アカウントパートナーにお問い合わせください。
Re-trigger EDL Automation
場合によっては (たとえば、新しい EDL テンプレートアイテムが追加されたときなど)、お客様は既存のStudy Person、Study Product、および Study Organization のレコードのエクスペクテッドドキュメントを再作成する必要があります。以前は、これを行えるのは、ライフサイクル状態の変更に対するエントリアクションを通じてのみでした。この機能では、状態変更を必要とせずに EDL 自動化作成ジョブをトリガーする新しいレコードアクションが導入されています。このアクションは、個々のレコードに対して実行することも、一括して実行することもできます。
Email to Vault: Subaddress Case Insensitive
この機能を使用すると、クリニカルメールプロセッサは、受信メールからサブアドレスを解析し、治験フィールドを設定するときに大文字と小文字を区別しません。メールからドキュメントを作成するために使用されるサブアドレスはすべて小文字として処理されます。Study を作成すると、Email Subaddress フィールドは、Study Number からすべて小文字としてコピーされます。さらに、リリース後に Study を編集すると、Email Subaddress フィールドは更新され、小文字になります。
Disable Related Document Requiredness on Quality Issues
場合によっては、ユーザエラー (機密コンテンツや PHI を TMF にアップロードするなど) により、Vault 内のドキュメントを「強制削除」する必要がある場合があります。この機能は、Related Document フィールドを必須ではなくオプションにすることで、ユーザが Quality Issues (QI) を記録する方法の柔軟性を高めます。以前は、ドキュメントを削除するには、ユーザは QI を TMF プランなどのドキュメントにリダイレクトする必要がありました。機能フラグを有効にすると、ユーザはドキュメントを完全に削除する前に Related Document フィールドを再ポイントする必要がなくなり、Quality Issues がドキュメントに存在する場合のドキュメント削除プロセスが合理化されます。
この機能は、元に戻すことのできない一方向機能フラグによって有効になります。このフラグを有効にすると、すべての Quality Issues のインデックスが再作成されるため、この機能は業務時間後に有効にすることをお勧めします。
Milestone Workspace & EDL Template Scalability Enhancements
23R2 リリースの一環として、Vault Clinical に 新たなテンプレート構造であるEDL オーバーライドテンプレートが導入されました。この新たなテンプレートを使用することにより、柔軟性を高め、国別 EDL テンプレート (カントリーインテリジェンス) の柔軟性を高め、全体的なメンテナンスにかかる手間を軽減できます。24R1 リリースで実装されたこの機能には、既存機能の強化や新機能の導入など、複数のコンポーネントが含まれています。
この機能には、次の機能強化が含まれています。
- EDL 階層ロジックの更新により、EDL 自動化、個人、組織または製品固有のエクスペクテッドドキュメントのサポートが強化されました。
- 国固有のエクスペクテッドドキュメントで追加のマイルストーンタイプが必要ない場合は、マイルストーンタイプ値が空白になっている Template Expected Document (
edl_item__v) レコードを使用して、オーバーライドテンプレートを設定できるようになりました。 - マイルストーンアイテムが削除された時に、そのアイテムに関連するマイルストーンドキュメントのみが削除されるようになりました。
この機能には、次の新機能が含まれています。
- 移行用途として、API を使用してマイルストーンドキュメントを一括作成できるようになりました。
- 治験削除のサポートとして、管理者はアーカイブ済み治験に含まれるマイルストーンドキュメントを削除できるようになりました。
この機能の PSA は次のとおりです。
- 24R1 より前では、カントリーインテリジェンスを使用した場合に一部の Clinical Vault で EDL オーバーライドが無効化される場合がありました。今回のリリースで、EDL オーバーライドはすべての Clinical Vault で有効化されるようになりました。
- 1 つのテンプレート階層内に、オーバーライドを使用する (すなわち、国オーバーライドフィールドが「はい」に設定されている) EDL テンプレートが 1 つでもある場合、Vault はカントリーインテリジェンスおよび EDL 自動化の EDL オーバーライドロジックに従います。
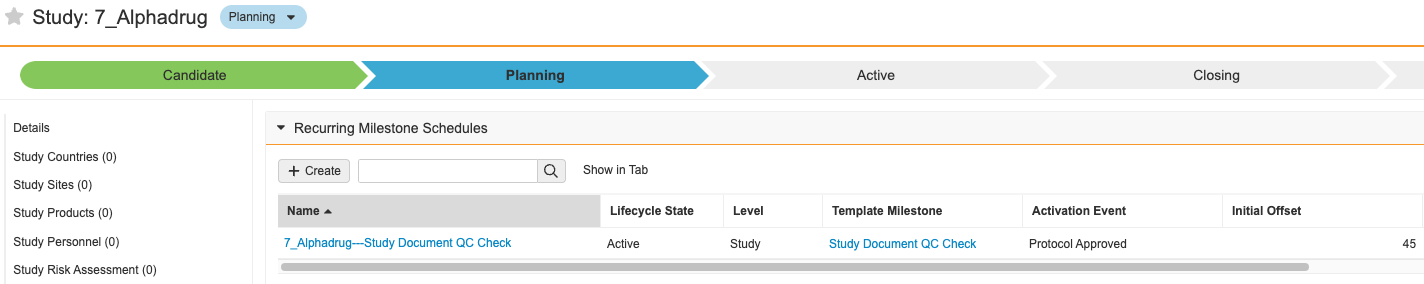
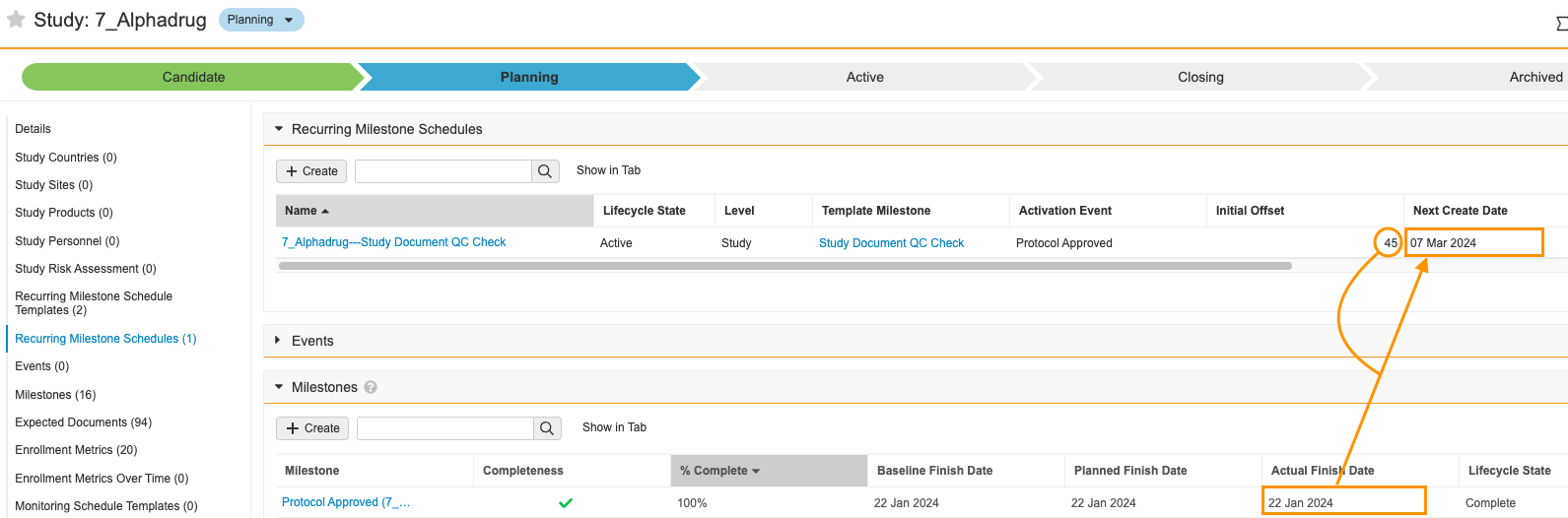
Recurring Milestone Schedules
既存の Milestones 機能は、1 回限りのアクティビティを効果的に管理しますが、現在、Vault には、治験の特定の期間中に定期的に繰り返し計画および実行する必要があるプロセスのマイルストーンを作成する簡単なオプションはありません。一般的な使用例は、長期にわたる治験中に「x」日ごとにドキュメント QC を実行することです。定期的なマイルストーンスケジュール機能は、このギャップを埋めます。
この設定可能な機能では、Recurring Milestone Schedules と Recurring Milestone Schedule Templates という 2 つの新しいオブジェクトが導入されています。これらにより、定期的な日付ベースの間隔でマイルストーンの作成を管理できます。
ユーザはイベント (たとえば、治験の 1 回限りのマイルストーンの完了) を定義して、これらのスケジュールをアクティブまたは非アクティブにできます。スケジュールがアクティブである間、毎日のジョブにより、スケジュールの次回作成日に指定した治験、治験実施国、または施設のマイルストーンが作成されます。
たとえば、ドキュメント QC は治験レベルで追跡されるものの、治験実施計画書承認済みマイルストーンが完了するまで実施されない治験があるとします。この場合、ドキュメント QC の定期的なマイルストーンスケジュールを設定して、治験実施計画書承認済みマイルストーンをアクティベーションイベントにすることができます。
治験実施計画書承認済みマイルストーンが完了すると、システムアクションにより定期的なマイルストーンスケジュールがアクティブ化され、ドキュメント QC マイルストーンが作成される次回作成日が設定されます。
定期的なマイルストーンにより、治験チームはこれらの繰り返しのプロセスを計画および管理できます。
CTMS
CTMS Transfer
臨床研究に携わる治験依頼者は、受託臨床試験実施機関 (CRO) の専門知識と契約して試験を管理することがよくあります。このような状況において、CRO が推奨する運用モデルには、独自の臨床試験管理システム (CTMS) 内で試験データを追跡および管理し、確立されたプロセスを活用して治験の効率を高めることが含まれます。
このように責任を委任しても、治験依頼者は被験者の安全性と試験データの品質と完全性に対する最終的な責任を負います。治験依頼者は、治験への登録、ステータス、コンプライアンス、問題解決などの CRO の活動を綿密に監督する必要があります。この監視の証拠を文書化すること (データレビューと監視の問題の記録) は、規制当局の検査にとって非常に重要です。そのため、治験依頼者は CRO パートナーと協力してコストのかかるカスタムソリューションを実装し、CTMS から監視データにアクセスする必要があります。
CRO と治験依頼者システム間のデータ転送を簡素化し、治験依頼者が治験データにシームレスにアクセスできるようにするために、CTMS 転送を導入します。この機能は、両方の組織が Vault CTMS を利用している場合、CRO から治験依頼者への日々の試験データの転送を自動化します。転送には、治験実施国、施設、治験群、登録メトリクス、マイルストーン、問題、モニタリング報告書のドキュメントなどの重要な要素が含まれます。転送は一方向であり、指定されたソース Vault からターゲット Vault にしか行われないことに注意することが重要です。転送は特定の標準フィールドと値に限定されます。ソース Vault 内のカスタム値はすべて、臨床標準フィールドにマッピングする必要があります。さらに、転送されたデータはターゲット Vault 内で読み取り専用のままになります。
Oversight Issue Tracking for Outsourced Studies
近年、臨床試験のアウトソーシング市場は大幅な成長を遂げており、この傾向は今後数年間続くと予想されます。フルサービスアウトソーシング (FSO)、特定サービスアウトソーシング (FSP)、ハイブリッドモデルなどのさまざまな外部委託モデルが、業界の多様なニーズに対応します。
ICH E6 (R2) では、治験依頼者はいかなる外部委託モデルを選択した場合でも、外部委託した治験に対する最終的な責任を負い、適切な監督を維持する必要があると強調されています。したがって、治験依頼者は、治験参加者の安全と健康、および治験中に開発業務委託機関 (CRO) が収集したデータの正確性と品質を確保する責任を負います。経験上、治験依頼者は検査中、特に監督プロセスを通じて特定された問題や CRO への伝達に関して、監視アクティビティの証拠を検査官に提供する必要があることがわかっています。
監視アクティビティの文書化を標準化するために、Vault CTMS に新しい Oversight Issue オブジェクトを導入します。この機能により、Monitoring Events レコード、Milestones レコードおよび Issue レコードの監視問題の追跡が可能になります。さらに、定期的な監視マイルストーンと臨床ユーザタスクにより、CTMS ユーザは監視アクティビティを通じてガイドを得ることができます。この場合、監視問題は、これらの臨床ユーザタスクから直接追跡でき、これらの臨床ユーザタスクに関連付けることができます。これらの活動を Vault CTMS で文書化すると、監視問題の追跡に包括的なコンテキストが提供され、エンドツーエンドのストーリーを説明するのに役立ちます。
Complex Trials: Subject Metrics and Recruitment Planning
今回のリリースでは、Clinical Operations データモデルの更新により、Vault Clinical で被験者グループを使用して複雑な治験デザインを追跡できるようになりました。さらにこの機能によって被験者グループが拡張し、被験者指標と被験者募集計画が必要な箇所をグループ別に指定しやすくなりました。
新たに追加された Create Recruitment Plan と Track Enrollment Metrics の 2 つのフィールドでは、特定の被験者グループに対して、それぞれ経時指標および被験者指標を必要とするかどうかを選択できます。指標タイプは、治験レベルで設定された値に基づいて生成されます。たとえば、使用しない指標リストに登録されている指標は、治験内にも各被験者グループ内にも作成されません。
指標および経時指標の計算動作が更新され、被験者グループ内の件数と治験内の件数を独立してカウントするようになりました。たとえば、ある被験者が特定の被験者グループに登録されている場合、その被験者はそれぞれ登録先被験者グループの登録済み合計指標と、治験の登録済み合計指標に別々にカウントされます。被験者が被験者グループに登録されていない場合は、その被験者は治験レベルでのみカウントされます。被験者グループの指標計算は、日付ベースの指標計算が選択されている場合にのみ適用されます。
ロールアップ動作も、同様に独立して機能します。被験者グループが指定されている場合、施設レベルの指標は、対応する被験者グループの国別指標にロールアップされ、その国別値が対応する被験者グループの治験指標にロールアップされます。同様に、被験者グループが指定されていない指標も、施設レベルの値は国レベルにロールアップされ、その国別値が治験レベルにロールアップされます。
Complex Trials: Subject Milestones
自動化された症例登録マイルストーン機能が、複雑な臨床試験設計をサポートできるよう強化されました。この機能を設定すると、被験者グループ登録マイルストーン (すなわち、下位試験の最初の被験者) に、被験者グループの指定されている関連する被験者レコードに入力された該当の日付に基づいて、実際の完了日が自動入力されます。治験登録マイルストーンおよび被験者グループ登録マイルストーンは、別々に入力されます。
Last Subject Milestones: Lost to Follow-Up Dates Evaluated
この機能により Vault では、最終被験者登録マイルストーン (すなわち、最終症例完了日) に実際の完了日を入力した際に、被験者レコードのフォローアップ喪失日が自動評価されるようになりました。
Japanese Clinical Trial Notification Validation Review
Vault CTMS は、日本での臨床試験をサポートするために設計された堅牢な機能セットを提供します。これには、日本の医薬品医療機器総合機構 (PMDA) 向けの治験届 (CTN) を特定の XML 形式で生成する機能が含まれます。コンプライアンスを維持するには、これらの CTN を定期的に、一定の期間内に提出する必要があります。
規制要件がダイナミックに変化する状況では、CTN の XML 形式やコンテンツルールが急遽変更されることは珍しくありません。このような場合、お客様にとっては、同じ時間制約内でこれらの新しい要件に迅速に適応し、準拠することが依然として不可欠です。お客様のプロセスの機敏性を維持し、CTN の生成を妨げないことが重要です。
お客様が最新の規制基準に迅速に対応できるようにするために、CTN 検証レビュー機能を導入します。Vault CTMS ユーザは、現在実施されている検証チェックを確認した後、CTN ドキュメントと XML ファイルの生成を続行することができます。これにより、柔軟で即応性の高いアプローチが保証され、お客様は効率を損なうことなく規制の変更に迅速に適応できるようになります。
Vault Payments
Payments Adjustment Reasons
支払調整を使用すると、対応する Visits、Procedures、Site Fees、Fee Schedules に変更が加えられたときに、Vault で自動的に Payable Items が更新されます。この機能により、Vault は事前定義された選択リストから Payment Adjustment Reason を自動的に入力し、ユーザに追加情報を提供して、調整に関する施設とのコミュニケーションを合理化します。
詳細については、Vault 支払調整をご覧ください。
Payment Adjustment Override
ユーザは自動更新から Payable Items を除外できるようになりました。これにより、既存のお客様が以前は手動で管理していた項目を分離する必要がある場合にも、簡単に導入できるようになります。また、個々の Payable Items の逸脱を承認しながら、Vault Payments の対応する Fee に関連付けたままにすることもできます。
詳細については、Vault 支払調整をご覧ください。
eTMF
TMF Standard Document Fields
業界標準の概念をサポートするために、Clinical Operations Vault に 6 つの新しい標準ドキュメントフィールドとトリガーを導入します。この機能により、新しいフィールドと 2 つの既存フィールド (Primary および Secondary QC Status) にトリガーが追加されます。新しいフィールドは共有フィールドとしてプロビジョニングされ、使用するドキュメントタイプに設定する必要があります。以下は、新しいフィールド、その動作、およびその使用目的の説明を示したものです。
|
フィールドラベル |
フィールド名 |
編集可能 |
フィールドタイプ |
フィールドの説明 |
フィールドの動作 |
|
Effective Date |
|
はい |
日付 |
このドキュメントが使用可能になったとみなされる日付。 |
|
|
Collected Date |
|
はい |
日付 |
このドキュメントバージョンをソースから受け取った日付。 |
|
|
System Created Date |
|
いいえ |
日付 |
このドキュメントバージョンが Vault にアップロードされた日付。 |
|
|
Inspection Ready Date |
|
はい |
日付 |
このドキュメントバージョンが検査準備完了とみなされ、すべての品質管理チェックに合格した日付。 |
|
|
System Approved Date |
|
いいえ |
日付 |
このドキュメントバージョンが Vault で承認された日付。 |
|
|
External Document Version |
|
はい |
数字 |
Vault のバージョンとは無関係の、Vault 外部で追跡されるこのドキュメントのバージョン番号。 |
該当なし |
|
Primary QC Status |
|
はい |
既に存在している |
既に存在している |
|
|
Secondary QC Status |
|
はい |
既に存在している |
既に存在している |
|
Study Country & Site Metadata Extraction
TMF Bot の機能は引き続き拡張されています。ドキュメント分類と治験フィールドのほか、治験実施国と治験実施施設の各メタデータもサポートされるようになりました。これらの値は、Vault のドキュメントインボックスにドキュメントをアップロードした時点で自動入力されます。この機能強化は治験メタデータの抽出と緊密に連携し、治験の一致が見つかった治験実施施設が優先されます。
この拡張機能により、eTMF 内のドキュメントの処理がさらに合理化されます。弊社の調査と研究では、TMF Bot によって治験、治験実施国および施設の情報を自動抽出することで、ドキュメント 1 件あたり平均 1 分の時間削減になると見込まれています。
Study Startup
Survey Reminder Notifications
Vault Study Startup が調査リマインダー通知をサポートするようになったため、フィージビリティチームは、未完了の調査を完了するよう施設に通知するために、電子メールや電話でのコミュニケーションに依存する必要がなくなりました。この機能を使用すると、フィージビリティチームは Vault を利用して、設定した頻度でリマインダーをトリガーできます。ユーザは、Vault が調査回答者に個別または一括で通知を送信できる、アドホック通知の新しいアクションを活用することもできます。施設とアウトリーチターゲットへの招待状は、最終リマインダー日、次回リマインダー日、通知頻度、リマインダー配信ステータスを追跡します。この機能により、PAL リンクの有効期限も 30 日から 60 日に延長されます。
詳細については、フィージビリティ調査とチェックリストのデザインをご覧ください。
Cycle Time: Omission of Study Part Milestones
マスタープロトコルをサポートするために、Vault では治験群関連のマイルストーンのサイクルタイムの計算が省略されます。マスタープロトコルのデータモデルの一部として、マイルストーンにより治験対象グループによる登録追跡が可能になります。治験群 (arm__v) の値を含むマイルストーンは、夜間サイクルタイムジョブから除外され、サイクルタイム指標を歪めません。
Site Connect
Send Investigator Brochure with Safety Distribution
この自動オンの機能強化により、Site Connect をご利用のお客様は、Site Connect の Safety Distribution 機能を利用して治験薬概要書を各治験実施施設に送信できるようになりました。治験薬概要書は、メインの安全性ドキュメントとして Safety Distribution に関連付けるか、サポート文書として追加することができます。
Mark Document Recalled Tracking
この自動オンの機能強化により、Site Connect をご利用のお客様は、どの治験実施施設でドキュメントがリコールとしてマークされたかを追跡し、報告できるようになりました。
この機能が導入される前は、治験依頼者/CRO は、措置を講じた後で、どの治験実施施設からドキュメントがリコールされたのかについて報告できませんでした。
注意: 新しい [治験依頼者/CRO によるリコール] フィールドが、[配布タスク] オブジェクトにチェックボックスとして追加されます。これにより、既存のすべての配布タスクレコードに FALSE の値が設定され、[最終変更日] フィールドが更新され、この更新のレコード監査にイベントは記録されません。
Additional Vault Clinical Docs Support
Site Connect のお客様は、次の種類のドキュメントを施設と交換できるようになりました。
- 治療復号ドキュメント
- IP ラベル変更ドキュメント
- 募集計画
- システムアカウント管理
- 安全性報告書の裏付け文書
関連するドキュメントタイプの設定を更新して、新しい Vault Clinical ドキュメント階層にマッピングする必要があります。
Site Connect Data Model Update on Distribution Task
このリリースでは、次の新しいフィールドを Distribution Task オブジェクトに導入します。
- Last Downloaded Date (
last_downloaded_date__v) - SiteVault Document Type (
sitevault_document_type__v) - Last Filed to SiteVault Date (
last_filed_to_sitevault_date__v) - SiteVault Document Number (
sitevault_document_number__v) - Recalled by Sponsor/CRO (
recalled_by_sponsorcro__v) - Reason for Recall (
reason_for_recall__v)
この自動オン機能はエンドユーザに新しい機能を提供するものではなく、24R2 で予定されている大規模な変更 (Site Connect の施設のエクスペリエンスを Clinical Operations Vault にもたらす) への準備です。
eConsent エディタ
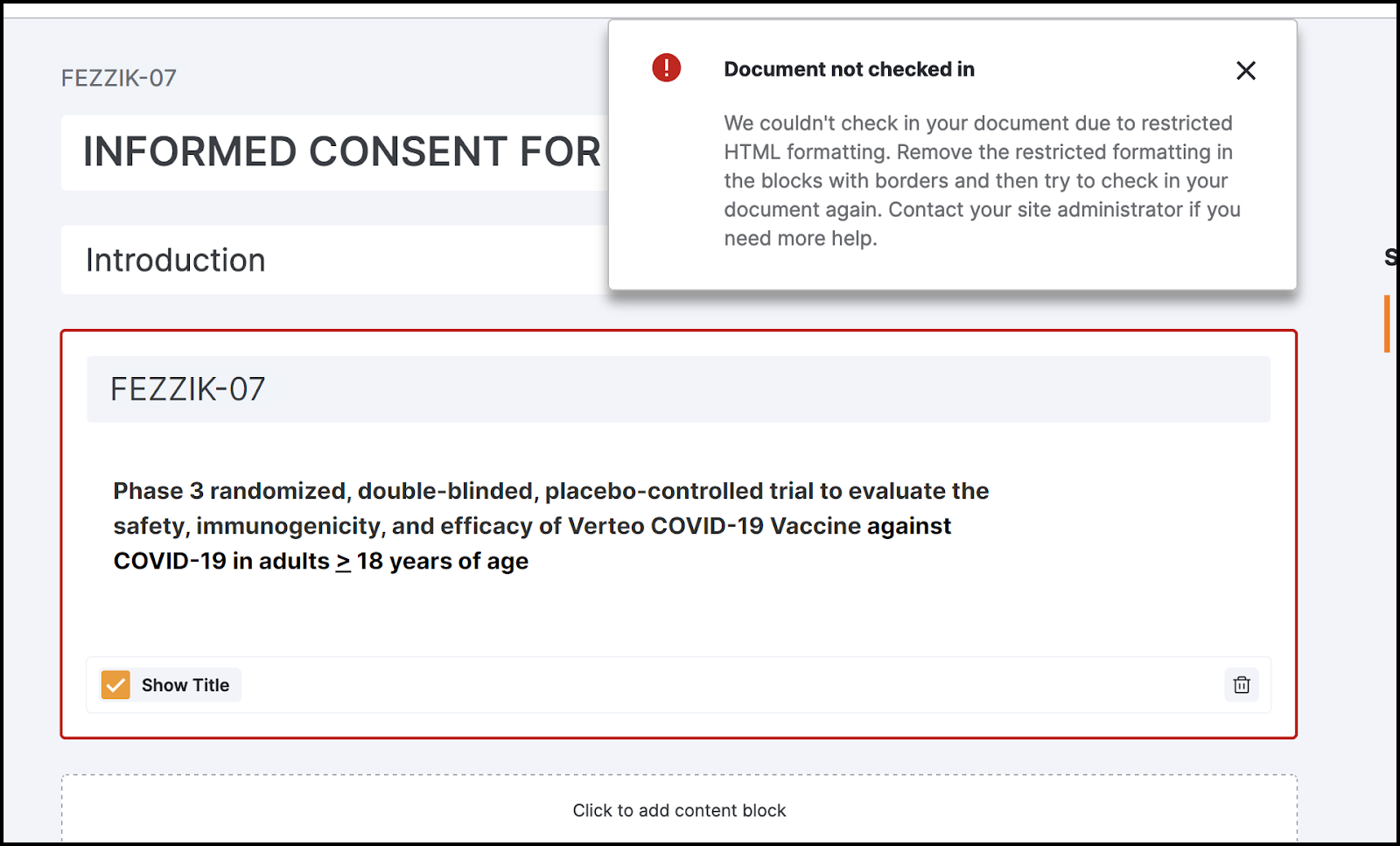
バックエンドエラー処理ガイド
この機能により、eConsent エディタではエラーのあるブロックとコンテンツが強調表示されるようになり、ユーザが問題をより迅速に特定して修正できるようになります。
ヘッダーとフッターの Merge フィールド
この機能により、eConsent のヘッダー部分とフッター部分で Vault Merge フィールドを使用できるようになりました。これにより、Word ドキュメントのアップロード時に、テキストに Vault 変数が含まれている場合も、変数で表示される代わりにヘッダーとフッターに入力されたテキストが表示されるようになり、コンテンツのアップロードが簡素化されます。
eConsent ビューア
eConsent プレビュータイムアウトの機能強化
この機能により、治験依頼者/CRO スタッフならびに施設スタッフは、システムがタイムアウトすることなく、比較的大きなファイルのプレビューを閲覧できるようになります。
eConsent: SiteVault インテグレーション
署名フォームの新しい回答フィールド
この機能により、治験依頼者/CRO スタッフは同意書への署名日時を確認できるようになったほか、署名レコード内のドキュメントの説明がより識別しやすくなりました。
Clinical Operations Data Model Changes
詳細に関しては、24R1 Clinical Operations データモデルの変更をご覧ください。
ePRO
患者調査
Single Select Optional Answer Shown as a Radio Button
この機能により、患者調査におけるオプション回答の表示がチェックボックスからラジオボタンへ更新されました。この表示変更により、患者がオプションの回答を選択した場合も選択できる回答は 1 つだけであるため、ボタンの機能がより正確に反映されました。
Studio
Translation Export by Survey
この機能により翻訳ファイルを調査またはグループ別に分けられるようになったため、翻訳ベンダーが翻訳プロセスを管理しやすくなりました。
FTP エクスポート設定アクセスの拡張
この機能により、Studio アクセス権限のある治験依頼者/CRO スタッフは誰でも FTP エクスポートを設定できるようになりました。
ePRO Vault
24R1 では、ePRO が Clinical Operations Vault および SiteVault から分離されました。新規治験ではすべて ePRO Vault が使用されます。今回リリースで、新しい ePRO Vault が入手可能になりました。
ePRO Vault
ePRO Vault を使用すると、必要な権限を持つ治験依頼者/CRO スタッフは、治験データの作成、および社内スタッフと施設スタッフの Veeva ePRO アクセス権限管理を行えます。Studio と Study Home には、ePRO Vault からアクセスします。また、ユーザは標準のユーザアクセスレポートを実行することもできます。
Create and Assign Site Users in the ePRO Vault
この機能により、治験依頼者/CRO スタッフは ePRO Vault 上で施設スタッフメンバーを治験に追加できるようになりました。施設スタッフには、Veeva ePRO で治験業務を行うよう招待する電子メールが送信されます。
治験実施施設のサポート対象患者言語の管理
この機能により、治験依頼者または CRO スタッフは、各施設で利用できるサポート対象の患者言語を管理できるようになります。各施設は、当該施設内でサポートされている患者言語の治験バージョンのみを受信し、サポート対象の患者言語でのみ参加者を設定できます。
Study Home
Study Home
この機能により、治験依頼者/CRO スタッフによる治験監視が向上します。さらに、治験、施設および参加者の各レベルでのコンプライアンス指標の監視に役立つほか、各施設でどの治験バージョンがアクティブであるかなどを示す、新しい指標を複数利用できます。
Veeva ePRO (施設)
このリリースでは、新しい ePRO Vault 外でホストされる治験に対応するため、各施設で参加者への ePRO の実施、eClinRO 評価の入力、および治験データへのアクセスを行うための新しいインターフェイスが追加されました。施設スタッフは、Veeva ID でログインして ePRO にアクセスできます。
参加者アクティビティと事象アクティビティ
この機能により、施設スタッフは、新しい Veeva ePRO を使用して参加者アクティビティや事象アクティビティを管理できるようになりました。以下の機能を利用できます。
- 参加者詳細の管理
- MyVeeva for Patients アカウントの登録/リセットに使用するアクティベーションコードの生成
- コンプライアンス指標と、参加者が送信したヘルプリクエストの閲覧
- スタディ事象発生時の入力
- スタディ調査の閲覧または入力
- 対面環境における参加者による調査入力の許可
- データ変更の入力
Study Details and Reports
この機能により、施設スタッフは新しい Veeva ePRO を使用して、スタディバージョンの管理およびレポートの実行を行えるようになりました。スタディ依頼者/CRO が各施設に適用されるスタディビルドを承認すると、その対象施設は有効化する必要のある新しいスタディバージョンを受信します。施設スタッフは ZIP ファイル 1 つをダウンロードするだけで、すべての患者向けコンテンツが入った ePRO コレクションドキュメントと、すべての施設向けスタディコンテンツが入った eClinRO コレクションドキュメントを入手できます。また、施設スタッフは担当施設でサポートされている患者言語の一覧も閲覧できます。
利用できるレポートの種類は、調査データ、データ変更、コンプライアンスおよび監査証跡です。
SiteVault に接続する
この機能を使用すると、施設スタッフは担当している ePRO スタディを SiteVault 上のスタディに接続できます。これにより、SiteVault で作成した参加者を ePRO に追加することができます。ePRO で作成された参加者は全員、SiteVault 上にも作成されます。すでに eConsent 用の MyVeeva アカウント (SiteVault から登録する) を持っている参加者は、引き続き同じアカウントで ePRO を使用できます。これにより、参加者に一貫したスタディエクスペリエンスを提供することができるとともに、施設スタッフが 2 重にデータ入力する手間が削減されます。SiteVault に接続できるのは、SiteVault スタディへのアクセス権限を持つ施設スタッフのみです。
新しい施設翻訳 - 日本語
この機能により、日本国内施設で Veeva ePRO インターフェイスを日本語表示にできます。
Reporting
レポートに調査のインスタンス番号と追加のスケジュール詳細を含める
この機能によりスタディ依頼者/CRO スタッフは、調査回答レポートとコンプライアンスレポートで、繰り返し行われる調査のインスタンス番号、およびトリガースケジュールのエクスポート詳細を確認できるようになりました。この情報は、データ分析用の入力データとして使用できます。
MyVeeva for Patients
施設側および患者に求められる登録プロセスを簡素化することによりエクスペリエンス向上を図るため、複数の更新を行いました。さらに、システムの個人を特定できる情報に対する依存度も軽減しました。
Streamlined MyVeeva Registration: Activation Code
この機能により施設スタッフは、MyVeeva ユーザが登録に使用できる Study Connect または Veeva ePRO からアクティベーションコードを生成できるようになります。施設が参加者または介護者の生年月日をすでに施設側システムに入力済みである場合、MyVeeva ユーザは登録時にその値を入力することによって、施設側で対象者にアクティベーションコードを発行済みであることを確定できます。
MyVeeva 登録の効率化: PIN アクセス
この機能により、全ユーザが iOS MyVeeva アプリまたは Android MyVeeva アプリで PIN ログインを設定できるようになりました。これまでは、電子メールアドレスまたは電話番号を持たないユーザのみが PIN を設定できました。
Streamlined MyVeeva Registration: Account Settings
この機能により MyVeeva ユーザは、iOS、Android とウェブアプリにあるアカウント設定ページから、自分のユーザ名、パスワード、メールアドレス、電話番号および PIN を更新できるようになりました。
Streamlined MyVeeva Registration: Conversion and Account Recovery
この機能により MyVeeva ユーザは、自分のアカウントにログインできない場合に、担当のスタディチームに連絡して回復アクティベーションコードを提供してもらうことができます。また、ユーザはアカウントページで新しいスタディのアクティベーションコードを入力することにより、既存のアカウントを使って新しいスタディをアクティブ化することもできます。
MyVeeva 登録の効率化: ログイン、署名と監査証跡の更新
この機能に関して以下の更新が行われました。
- Veeva ePRO を SiteVault に接続済みの場合、MyVeeva ユーザは、最初は eConsent 用途でのみ使用していたがその後 ePRO に追加された際に、同じログインアカウントを引き続き使用できます。
- 規制遵守を保証するため、アプリでは、ユーザが電話での 2 要素認証手順を行えない場合に手書きの署名を収集するようになりました。
- 監査対象の事象およびアクションを確実に追跡するため、監査証跡が更新されました。
翻訳
右から左方向に書く言語のサポート
この機能により、スタディ依頼者、CRO および施設スタッフは、スタディテキストをヘブライ語やアラビア語などの右から左方向の書式構造をもつ言語で閲覧するため、最適化できるようになりました。またこれにより、MyVeeva ユーザはアプリおよび翻訳されたコンテンツすべてを、右から左方向に書く言語で表示できるようになりました。
24R1 翻訳と新たな言語の追加
この機能により MyVeeva ユーザは、アプリケーションのテキスト、メール、通知、テキストメッセージ、利用規約およびプライバシーポリシーを、アラビア語 (イスラエル)、カタロニア語 (スペイン)、英語 (オーストラリア)、ドイツ語 (ベルギー)、ヘブライ語 (イスラエル)、ならびにハンガリー語 (ハンガリー) で表示できるようになりました。
全般的な UI
30-Day Offline Experience in the Mobile App
この機能により MyVeeva ユーザは、ネット接続を求めるメッセージが表示されるまで、最大 30 日間にわたってインターネットまたはネットワークにアクセスせずに MyVeeva for Patients モバイルアプリの操作を続けることができます。オフライン利用中は、アプリ内にユーザの送信したデータがすべて保存され、デバイスが再びネットに接続された時に保存済みのデータが送信されます。オフライン利用中は、ユーザは必要な調査を 1 回のみ送信でき、調査に再度アクセスするにはインターネットに接続する必要があります。
Account Deletion Instructions
この機能により MyVeeva ユーザは、Android アプリと iOS アプリの設定ページから、プライバシーポリシーに記載されたアカウント削除のリクエスト方法に関する説明を開くことができるようになりました。
Commercial
以下の Release Notes に加えて、Vault PromoMats Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモを提供しています。
Vault Connections セクションにリストされているいくつかの機能も、Commercial アプリケーションファミリーに影響を与えます。
PromoMats
Improved OCR for Images
Vault の光学文字認識 (OCR) 機能が改善され、画像からテキストをより正確に検出できるようになったため、より多くの読み取りテキストを他の Vault 機能でも使用できるようになりました。これには、検索や注釈、自動リンクなどが含まれます。
24R1 より前は、Vault の OCR 機能はスキャンされた PDF を主な読み取り対象として最適化されていたため、画像ファイルが頻繁に用いられる PromoMats などのアプリケーションが影響を受けていました。このような画像ファイルの OCR を改善することにより、より一貫したユーザエクスペリエンスで文字認識テキストに依存する機能を活用いただけます。
詳細については、光学文字認識をご覧ください。
Commercial Action Layouts
PromoMats では、コピーしてカスタムアクションレイアウトのベースとして使用できる、Commercial 標準アクションレイアウトが利用できるようになりました。オブジェクトとオブジェクトタイプには多数のフィールドを使用できますが、特定のライフサイクル状態や特定のユーザにおいては、そのフィールドすべてが関連しているわけではありません。アクションレイアウトを使用すると、特定のライフサイクル状態やユーザセキュリティプロファイルに合わせてオブジェクトレコード入力画面をカスタマイズすることで、効率を高めることができます。
Auto-On Auto-Linking: Match Text Variations Maximum
24R1 では自動リンク付けの一環として、すべての PromoMats Vault に 9 回の一致テキスト最大変更数が割り当てられています。
Automated Image Tagging
Vault PromoMats は、アップロードされた画像コンテンツの自動タグ付けをサポートするようになりました。この機能は、管理者が設定した事前定義タグをドキュメントのメタデータフィールドに入力します。Automated Image Tagging は、プロセスの効率化をサポートしタグ付けを標準化するうえで重要であり、検索性の向上によりエンドユーザがデジタル資産を見つけやすくなります。
Automated Image Tagging は、Vault によって作成された表示可能なレンディションを活用し、次のファイル形式をサポートします。
- 画像形式: .eps、.ai、.indd、.psd、.avif、.bmp、.jpg、.jpeg、.png、.svg、.tif、.tiff、.webP、.heic
- Creative Cloud: .ai、.indd、.psd、および InDesign パッケージ (zip)
- Powerpoint プレゼンテーション: .ppt、.pptx
- その他: .pdf、.html、.htm
Automated Image Tagging は、音声ファイルや動画ファイルをサポートしません。
この機能は AWS Rekognition サービスを活用しており、お客様は Amazon の強力な Rekognition エンジンの恩恵を受けてタグ付けの一貫性を向上させることができます。
Automated Image Tagging は PromoMats ユーザライセンスに含まれており、Vault ごとに毎月最大 250,000 件のタグ付けリクエストが可能です。
Modular Content Combinations
Vault では、ユーザがコンテンツモジュールの組み合わせを作成できるようになりました。組み合わせにより、ユーザは特定のチャネル、メッセージング、またはペルソナのコンテンツモジュール内のアセットをグループ化できます。
コンテンツモジュールの組み合わせを CRM メールモジュール内で使用する場合、その組み合わせは、CRM メールビルダーの許可されたアセットのビジネスルールとして動作します。
Modular Content: CRM Email Builder
今回のリリースで、モジュールタイプ「CRM メール」が導入されました。このモジュールタイプにより、ユーザは CRM メールモジュールでパーソナライズされたメールを作成できるようになります。Vault PromoMats が新たにホストするメールアセットを使用すると、現場担当者は CRM メールモジュールを使って、社内設定されたルールの範囲内でパーソナライズされたメールを作成できます。この機能を使用するには、CRM メールモジュールタイプおよびその他のデータモデルの変更を有効化設定する必要があります。
Commercial Application Settings Optimization
PromoMats アプリケーション設定が更新され、不要になったフラグが削除され、設定ラベルが機能名に揃えられました。機能の起動時に必要なフラグは、関連性がなくなったり、設定で管理できるようになったりする場合があります。
eCTD: Annotate Submission Ready References
このリリースでは、eCTD コンプライアンスパッケージに取り込まれた注釈付き参照および注釈付きラベル付けドキュメントで、関連する実証コンテンツを強調表示することができます。含まれている PDF リンクに関連するアンカーは、提出準備完了コピーでは赤いボックスで囲まれています。
Claims Linking: Auto-On Auto-Linking
よりターゲットを絞ったクレームのリンク付けのエクスペリエンスを提供するために、自動リンク付けがすべての PromoMats Vault のデフォルトになりました。ユーザがリンクの推奨アイコンをクリックすると、新しい自動リンク付け機能がバックグラウンドで実行され、柔軟性と提案が向上して、より優れたマッチングエクスペリエンスが実現します。
Claims Linking: Lightbulb Icon for Auto-Linking
対応するライフサイクルのどの状態にも推奨リンクのユーザアクションが追加されていない場合、すべてのドキュメントの状態に電球アイコンが表示されます。いずれかの状態に推奨リンクのユーザアクションが設定されている場合、該当する状態のみに電球アイコンが表示されます。設定は必要ありません。電球アイコンは、ドキュメントタイプの詳細にある有効化アクションに基づいてドキュメントタイプに使用できます。
Claims Linking: Language Filter Behavior Change
このリリースでは、Language フィールドの自動リンク付けフィルタリングが変更されました。自動リンク付けの実行時にドキュメントで Language が使用可能な場合、システムはドキュメントの Language 値と一致するすべてのテキストアセットとの一致を試みますが、Language 値が設定されていないすべてのテキストアセットとの一致も試みます。
Text Assets: Functionality Changes
すべての PromoMats Vault で自動リンク付けが有効になっているため、500 件のレコード作成制限が削除され、テキストアセットには 9 つを超えるカスタムフィールドを含めることができるようになり、一致テキストバリエーションには最大 9 つ含めることができます。
Text Assets: Duplicate Record Change
同じフィールド値のあるテキストアセットが複数存在して、テキスト値と一致することができるようになりました。ただし、自動リンク付けまたは手動テキストアセットリンク付けを使用する場合、同じフィールドと一致するテキスト値を持つテキストアセットを 1 つだけ承認済み状態にして識別できます。
PromoMats Text Asset Object: Remove Custom Field Limit
テキストアセットオブジェクトには、各オブジェクト標準の最大数のカスタムフィールドを設定できるようになりました。
PromoMats Text Asset Library: Record Limit Increase
テキストアセットオブジェクトには、フィールドに同じ値を持つレコードを 500 件以上入れることができます。製品、国、任意言語の組み合わせ最大数は 5,000 です。
Portals: Layouts Available in the Portal Library
ポータルのエンドユーザは、ポータルライブラリのコンテンツを表示するときに追加のオプションを利用できるようになります。ドキュメントライブラリの標準レイアウトがポータルライブラリで利用できるようになります。これにより、エンドユーザには、日々使用する Vault に慣れているのと同じように、ポータルでの一貫したユーザエクスペリエンスが提供されます。
現在利用できるレイアウトには、コンパクト、サムネイル、詳細、グリッドがあります。グリッドビューには、24R1 より前にポータルライブラリで使用されていた既存のレイアウトが反映されます。
コンパクトレイアウトは、ポータルライブラリのコンテンツを表示するためのデフォルトビューになります。
Allow Multi-Select for Standard Audience Field
管理者は、professional_consumer__v ドキュメントフィールドを複数選択にすることができるようになりました。コンシューマとプロフェッショナルの両方が選択された販促用資料の場合、eCTD 一括生成機能がコンシューマコンプライアンスパッケージを生成し、2253 のコメントフィールドに「この提出にはプロフェッショナルとコンシューマの両方の対象者向けの資料が含まれています」という注記が追加されます。
Best Effort Mapping for Standard Metrics Durations
PromoMats は、標準メトリクス状態マッピング設定ページで状態が設定されていない Vault に対してベストエフォートマッピングを実行します。Vault に少なくとも 1 つの状態が既にマッピングされている場合、PromoMats はマッピング設定を変更しません。実行されるベストエフォートマッピングは、ここにあります。
Vault Digital Publishing: AWS Bucket Policy Support
Vault デジタル出版は、Vault とお客様のアマゾンウェブサービス S3 バケットとの間の統合を設定するためのバケットポリシーの使用をサポートするようになりました。
eCTD: Improved Error Messaging
eCTD コンプライアンスパッケージのエラー処理が改善され、アクション可能なエラーメッセージをより迅速にサポートできるようになりました。また、eCTD コンプライアンスパッケージを生成しようとしたときに、提出パッケージのレンディションが壊れていることをユーザに通知するエラーメッセージも導入しました。新しい通知テンプレートは、リリースの夜以降に eCTD コンプライアンスパッケージの生成に使用されることに注意してください。
eCTD: Create as System
eCTD コンプライアンスパッケージとその中で作成されるドキュメントがシステムによって生成されるようになりました。以前は、ドキュメントはユーザによって生成されたものとして表示されていました。この機能強化により、eCTD コンプライアンスパッケージを作成するユーザの権限要件が簡素化されます。現在、ユーザに必要なのは、提出準備完了コンプライアンスパッケージドキュメントタイプに対するバインダーの作成権限と、センターオブジェクトおよび販促用資料のドキュメントタイプオブジェクトに対する表示権限のみです。
eCTD: Submission Package Enhancements
eCTD コンプライアンス パッケージでは、Deflate64、Deflate、および Stored の圧縮方法で作成された提出パッケージに対応するようになりました。また、提出パッケージのコンテンツから作成されたドキュメントは、バインダー内の他の提出可能なコピーと同じ命名方法によって名前が変更されます。以前は、提出パッケージから作成されたドキュメントの名前は変更されませんでした。
eCTD: Update Correspondence Letter on Regenerate
プレクリアランス eCTD コンプライアンスパッケージの場合、パッケージの再生成時に通信レターが自動的に再生成されるようになりました。
Auto-On Multichannel Features: Enable CLM Integration with Auto-Publishing
CLM 機能は、Multichannel アプリでの使用ライセンスを有する Commercial Vault で自動的に有効化されます。
Set State for Previous Email Fragment Versions
管理者は、電子メールフラグメントが最新バージョンでない場合でも、新しいエントリアクションを、電子メールフラグメントが選択された状態に自動設定されるように設定できます。
Auto-On Multichannel Features: Enable Engage Integration
Engage ポータル機能は、Multichannel アプリでの使用ライセンスを有する Commercial Vault で自動的に有効化されます。
Auto-On Multichannel Features: Enable Approved Email
承認済みメール機能は、Multichannel アプリでの使用ライセンスを有する Commercial Vault で自動的に有効化されます。これには、メールフラグメントの自動公開機能が含まれます。
Multichannel Performance Improvements
この機能によりパフォーマンスが改善され、Multichannel コンテンツの配信速度が向上します。
Commercial Data Model Changes
詳細に関しては、24R1 Commercial データモデルの変更をご覧ください。
Medical
以下の Release Notes に加えて、Vault MedComms および Vault MedInquiry Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモを提供しています。
Vault Connections セクションの一覧に記載されている、新たに実装された Medical - Safety Connection や PromoMats - Medical Connection などの一部の機能は、Medical アプリケーションファミリーにも影響します。
MedComms
Auto-On Multichannel Features: Enable CLM Integration with Auto-Publishing
CLM 機能は、Multichannel アプリでの使用ライセンスを有する Medical Vault で自動的に有効化されます。
Set State for Previous Email Fragment Versions
管理者は、電子メールフラグメントが最新バージョンでない場合でも、新しいエントリアクションを、電子メールフラグメントが選択された状態に自動設定されるように設定できます。
Auto-On Multichannel Features: Enable Approved Email
承認済みメール機能は、Multichannel アプリでの使用ライセンスを有する Medical Vault で自動的に有効化されます。これには、メールフラグメントの自動公開機能が含まれます。
Multichannel Performance Improvements
この機能によりパフォーマンスが改善され、Multichannel コンテンツの配信速度が向上します。
Auto-On Multichannel Features: Enable Engage Integration
Engage ポータル機能は、Multichannel アプリでの使用ライセンスを有する Medical Vault で自動的に有効化されます。
Portals: Layouts Available in the Portal Library
ポータルのエンドユーザは、ポータルライブラリのコンテンツを表示するときに追加のオプションを利用できるようになります。ドキュメントライブラリの標準レイアウトがポータルライブラリで利用できるようになります。これにより、エンドユーザには、日々使用する Vault に慣れているのと同じように、ポータルでの一貫したユーザエクスペリエンスが提供されます。
現在利用できるレイアウトには、コンパクト、サムネイル、詳細、グリッドがあります。グリッドビューには、24R1 より前にポータルライブラリで使用されていた既存のレイアウトが反映されます。
コンパクトレイアウトは、ポータルライブラリのコンテンツを表示するためのデフォルトビューになります。
Vault Digital Publishing: AWS Bucket Policy Support
Vault デジタル出版は、Vault とお客様のアマゾンウェブサービス S3 バケットとの間の統合を設定するためのバケットポリシーの使用をサポートするようになりました。
Modular Content: CRM Email Builder
今回のリリースで、モジュールタイプ「CRM メール」が導入されました。このモジュールタイプにより、ユーザは CRM メールモジュールでパーソナライズされたメールを作成できるようになります。Vault Medical が新たにホストするメールアセットを使用すると、現場担当チームは CRM メールモジュールを使って、社内設定されたルールの範囲内でパーソナライズされたメールを作成できます。この機能を使用するには、CRM メールモジュールタイプおよびその他のデータモデルの変更を有効化設定する必要があります。
MedInquiry
Standard Case Owner & Enhanced Escalation for MedInquiry
この機能により、新しい Owner フィールドが Case オブジェクト、Case Request オブジェクト、Event オブジェクトに追加され、レコードの所有者と継続して同期できます。Owner フィールドのデフォルトは、医学的照会 UI の現在のユーザです。
ワークフローアクションを使用すると、Owner フィールドの値に、割り当てられたタスクを完了するユーザを入力できます。
この機能により、ユーザは作業に割り当てられた Cases を表示するビューを作成できます。
Automatically Add Email Attachments onto MedInquiry Object Records
現在、添付ファイル付きのメールを取り込むと、ユーザは添付ファイルをローカルマシンにダウンロードし、オブジェクトレコードの添付ファイルとして再アップロードする必要があります。この設定可能な機能により、取り込んだメールから症例リクエスト、症例回答、またはイベントオブジェクトレコードの添付セクションへの添付ファイルの自動転送が可能になります。
管理者は、「メール添付ファイルをレコードに引き込み」アクションを、症例リクエストおよび症例回答、またはイベント、症例リクエスト、症例回答のユーザアクションのいずれかに追加できます。
Allow Case Responses to be Created Automatically
この機能は、症例および症例リクエストオブジェクトのライフサイクルにアクションを追加して、症例回答を自動的に作成できるようにします。
「関連症例回答の作成」アクションを使用すると、症例確認メールとして使用できるレコードを自動的に作成できます。レコードは自動的に作成され、エントリアクションを介して HCP に送信されます。
Allow Case Response Emails to be Sent Automatically
「回答パッケージの送信」アクションは、ユーザアクションだけでなくエントリアクションとしても設定できるようになりました。この機能を使用すると、レコードが特定のライフサイクル状態に入ったときに症例回答を HCP に自動的に送信できます。
設定すると、自動的に発行される症例確認メール、症例ステータス更新メール、および症例終了メールの要件に対処するために使用できます。
詳細については、症例への回答をご覧ください。
Automatic Case Contact Creation for Email Ingestion
この機能を使用すると、メールが取り込まれ、症例連絡先の詳細に一致するメールアドレスが存在しない場合に、新しい症例連絡先が作成されます。この自動化により、最初の問い合わせの取り込み時に症例連絡先フィールドが空白のままになることがなくなります。
現在、そのメールアドレスの症例連絡先が存在しないメールが取り込まれた場合はその症例の症例連絡先フィールドは空白のままになるため、ユーザは元の問い合わせに症例回答をメールに返信する前に手動で症例連絡先を作成する必要があります。
詳細については、新しい症例連絡先の作成をご覧ください。
MedInquiry OpenData Search: Pull HCP Country & Show Only Local HCPs
この機能を使用すると、MedInquiry OpenData 接続経由で作成された症例連絡先に国が自動的に入力されます。
さらに、管理者設定により、MedInquiry ユーザが活動している国の HCP レコードのみを表示するように OpenData 検索を制限できます。ユーザが使用している国は、ユーザレコードのロケールフィールドによって定義されます。
詳細については、OpenData を使用して症例連絡先を追加するをご覧ください。
Support for Criteria VQL on OpenData Search Case Contact field
OpenData の症例連絡先フィールドが基準 VQL をサポートするようになりました。この機能により、国や連絡先のソースなどの基準の依存関係を処理できるようになります。
CRM Data Sharing: Pull Custom Response Details
MedInquiry の CRM データ共有機能が変更され、Veeva CRM 医療照会レコードで指定されたメールアドレスに症例回答を送信できるようになりました。
CRM データ共有機能では、以前は Vault の症例連絡先レコードで定義されたメールアドレスへのメール回答の送信のみがサポートされていました。このレコードは通常、Veeva CRM のアカウントレコードから同期されます。
たとえば、HCP が外出中で、アカウントレコードにあるメールアドレスとは異なるメールアドレスに症例回答を送信することを希望する場合、CRM ユーザは CRM 医療照会レコードで新しいメールアドレスを指定できます。CRM 医療照会レコードのメールフィールドが MedInquiry の症例のメールアドレスフィールドにマッピングされている場合、症例回答では、指定されたメールアドレスが回答受信者として使用されます。.
MedComms、MedInquiry
Medical Provision Action Layouts
コピーしてカスタムアクションレイアウトのベースとして使用できる、標準の医療アクションレイアウトが利用できるようになりました。オブジェクトとオブジェクトタイプには多数のフィールドを使用できますが、特定のライフサイクル状態や特定のユーザにおいては、そのフィールドすべてが関連しているわけではありません。アクションレイアウトを使用すると、特定のライフサイクル状態やユーザセキュリティプロファイルに合わせてオブジェクトレコード入力画面をカスタマイズすることで、効率を高めることができます。
MedComms、PromoMats
Modular Content: CRM Email & Combinations Data Model in Vault Medcomms
MedComms が Modular Content をサポートするようになりました。サポートされている使用例は、CRM Email Builder で使用する CRM メールモジュールの作成です。
Medical Data Model Changes
24R1 Medical データモデルの変更をご覧ください。
Training
Study Training は、Quality Suite: Vault Training アプリケーションと並行して機能とデータモデルの更新を受け取ります。
Study Training
以下の Release Notes に加えて、Vault Study Training Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモを提供しています。
Study Training: Training Matrix Automation
自動トレーニングマトリックスビルダーは、トレーニングマトリックスビルダーの新しいバージョンです。Vault では、臨床データおよびトレーニングマトリックスビルダーを使用して、Training Materials、Learner Roles および Studies に関連するレコードが自動作成されるようになりました。自動ビルダーでは、Learner Roles に割り当てられた Training Requirements に基づいて Curricula が作成されます。この機能により、1 件のスタディ内でトレーニングマトリックス作成にかかるクリック数が全体で 70% 削減されます。
Auto-Create Security Records for Study Country & Site
この機能は、User Role Setup レコードの作成に Study Country および Study Site が含まれるように拡張することにより、既存の自動化に基づいて構築されています。この機能は、このプロセスを自動化することにより、各ユーザおよびスタディの User Role Setup レコードを手動で作成する必要がなくなります。これにより、IT 部門の管理負担が軽減されるだけでなく、Clinical Operations Vault でのユーザエラーや不正なドキュメントアクセスのリスクも軽減されます。
Quality
以下の Release Notes に加えて、Vault QMS、Vault QualityDocs、Vault Training、Vault LIMS、および Validation Management Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモが提供されています。
Vault Connections セクションにリストされているいくつかの機能も、Quality アプリケーションファミリーに影響を与えます。
QualityDocs
Enable Launching Document Workflows from the Document Change Control
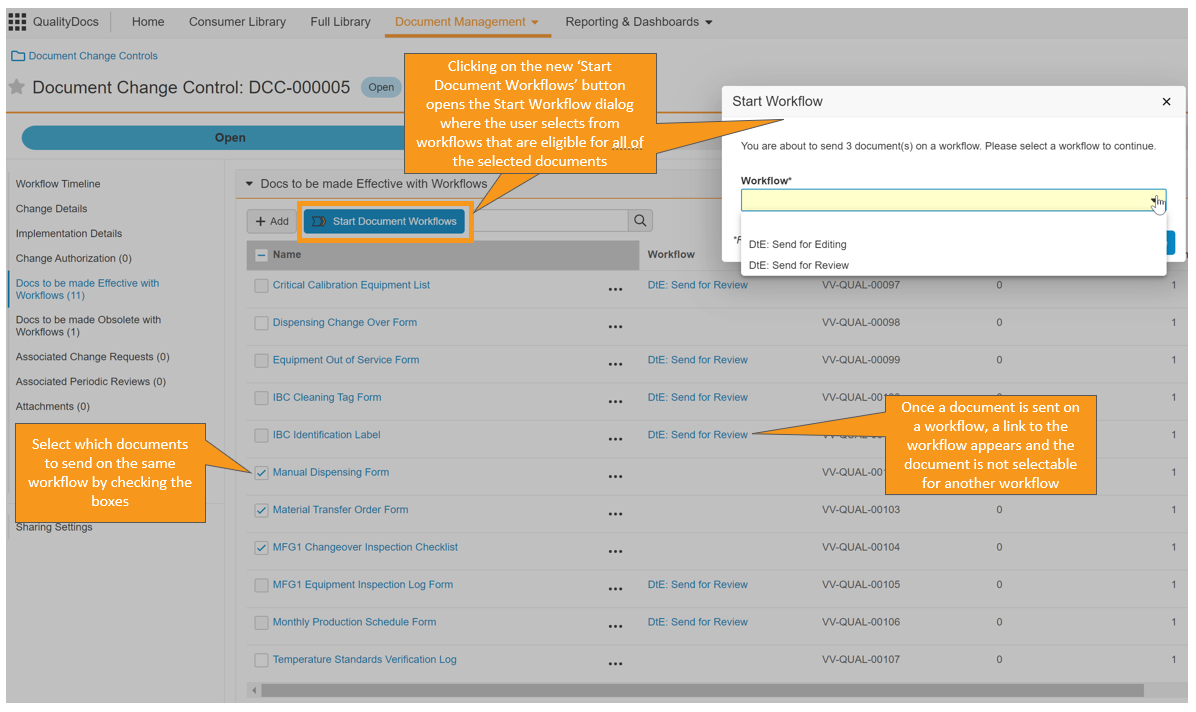
この機能によりユーザは、ドキュメント変更管理 (DCC) レコードから、DCC ページレイアウトに追加可能な新しい有効にするドキュメントセクションおよび廃止するドキュメントセクションを使用して直接、ドキュメントのワークフローを開始できます。ユーザーは、ワークフローの状態の表示や DCC からワークフローエンベロープのリンクをクリックできるようになりました。この機能は、レガシーワークフローと互換性がありません。
Document Change Control: Document Association Governance Limit
ガバナンスの制限により、1 つのDocument Change Control (DCC) に追加できるドキュメントの総数が Change Authorization、Documents to be made Effective および Documents to be Made Obsolete の 3 つのセクション全体で 100 までに制限されるようになりました。この制限は、24R1 Release 以降に作成されたすべての Document Change Control レコードに適用されます。リリース前に作成され、100 件以上の関連ドキュメントを含む Document Change Control レコードは、引き続き処理することが可能で、影響を受けません。
Station Manager
Android Station Manager 用のドキュメントページのサムネイルビュー
Station Manager の Android アプリケーションのユーザは、サムネイルビューアでドキュメントのすべてのページを表示するオプションを利用できるようになりました。これにより、ユーザはドキュメント内の目的のページにすばやく簡単に移動できるようになります。この機能は、23R3 リリースで Station Manager iOS アプリケーションのユーザ向けにリリースされました。
Training
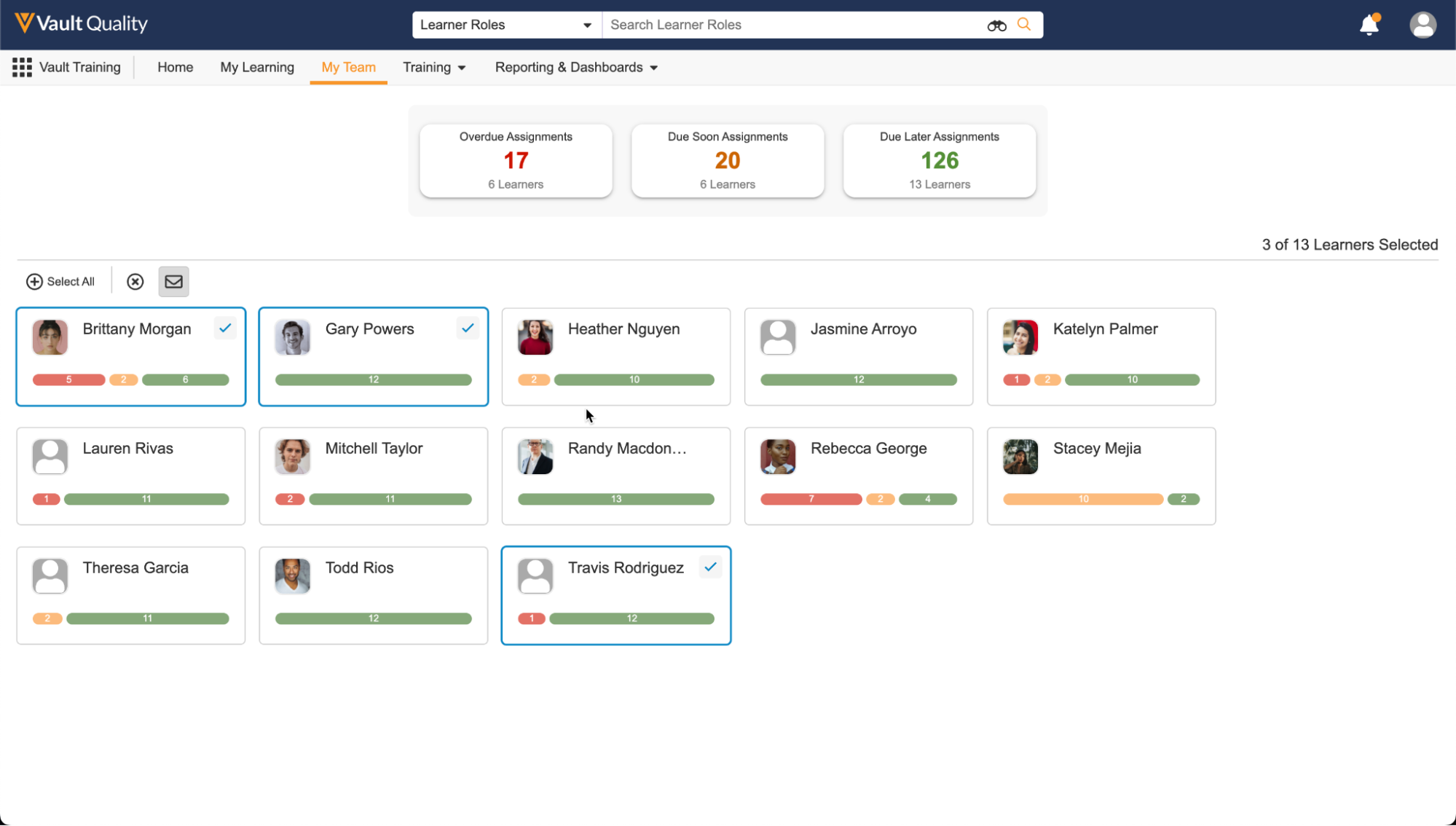
My Team Page (Manager Homepage)
マネージャは、直属のチームのトレーニングの進捗に関するリアルタイムの洞察にすぐにアクセスできるようになりました。マネージャは、中央のトレーニング管理者チームに頼ることなく、個人とチーム全体のパフォーマンスを一目で評価できます。この機能強化は、組織の規模を拡大するのに役立つだけでなく、トレーニング管理者の支援や介入なしでトレーニングステータスをアクティブに追跡して推奨事項や割り当てを行うことができるようにすることで、マネージャがチームのトレーニングで果たす重要な役割にも対応します。
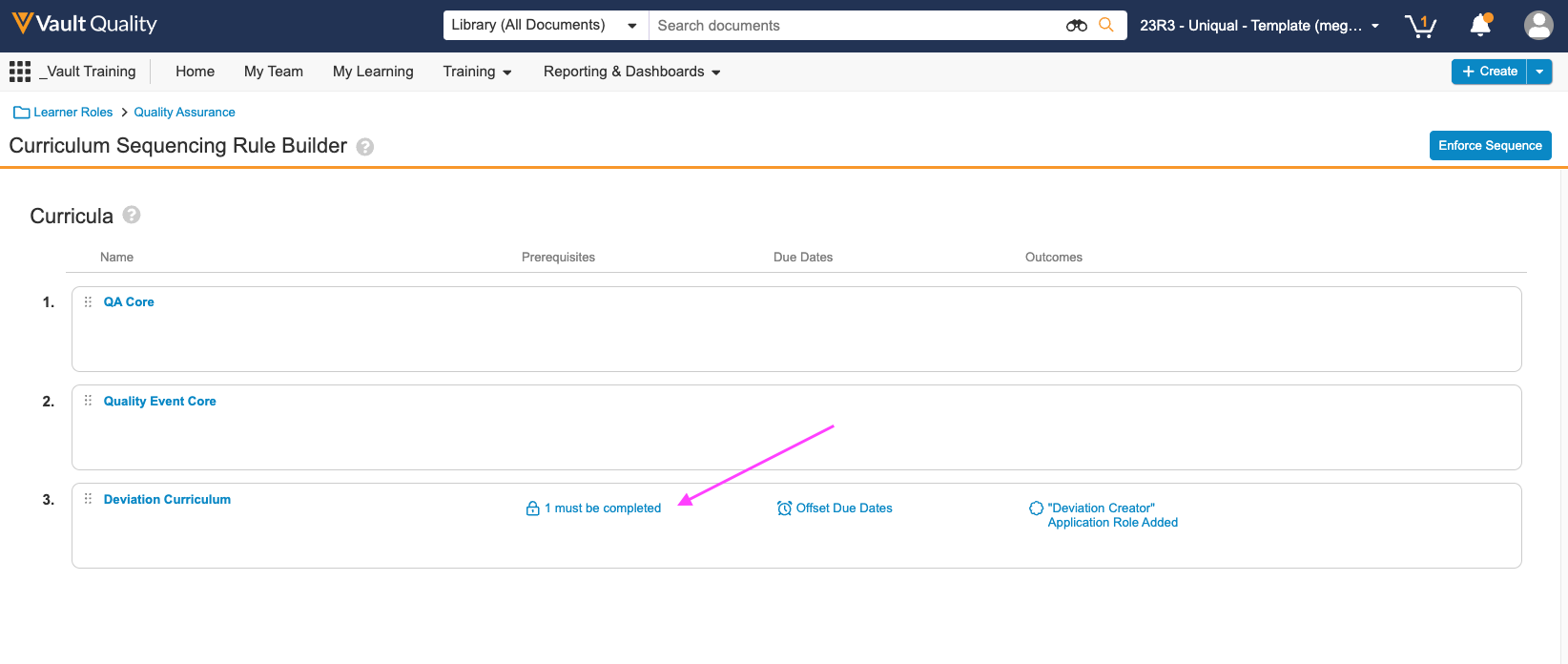
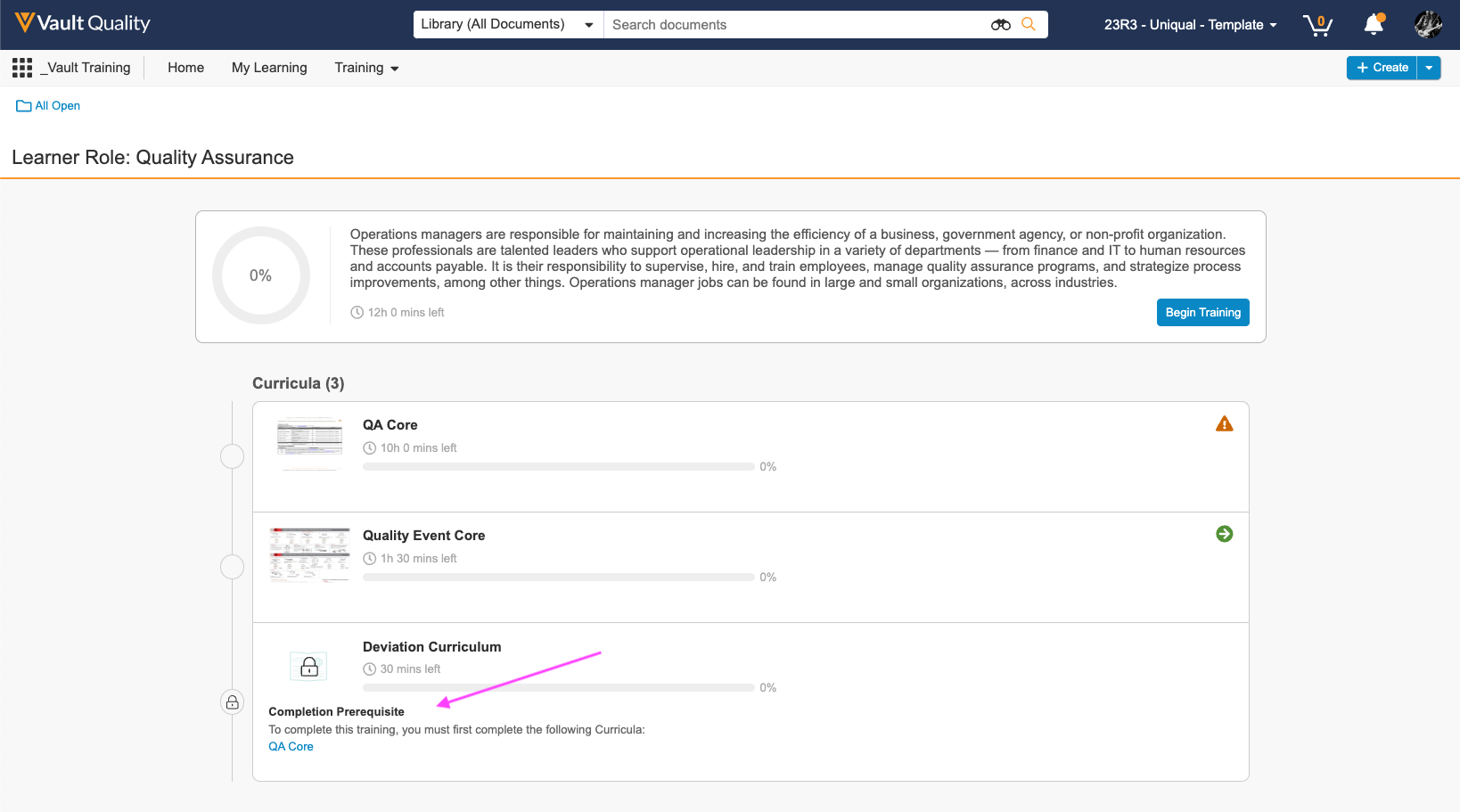
Curriculum Prerequisites
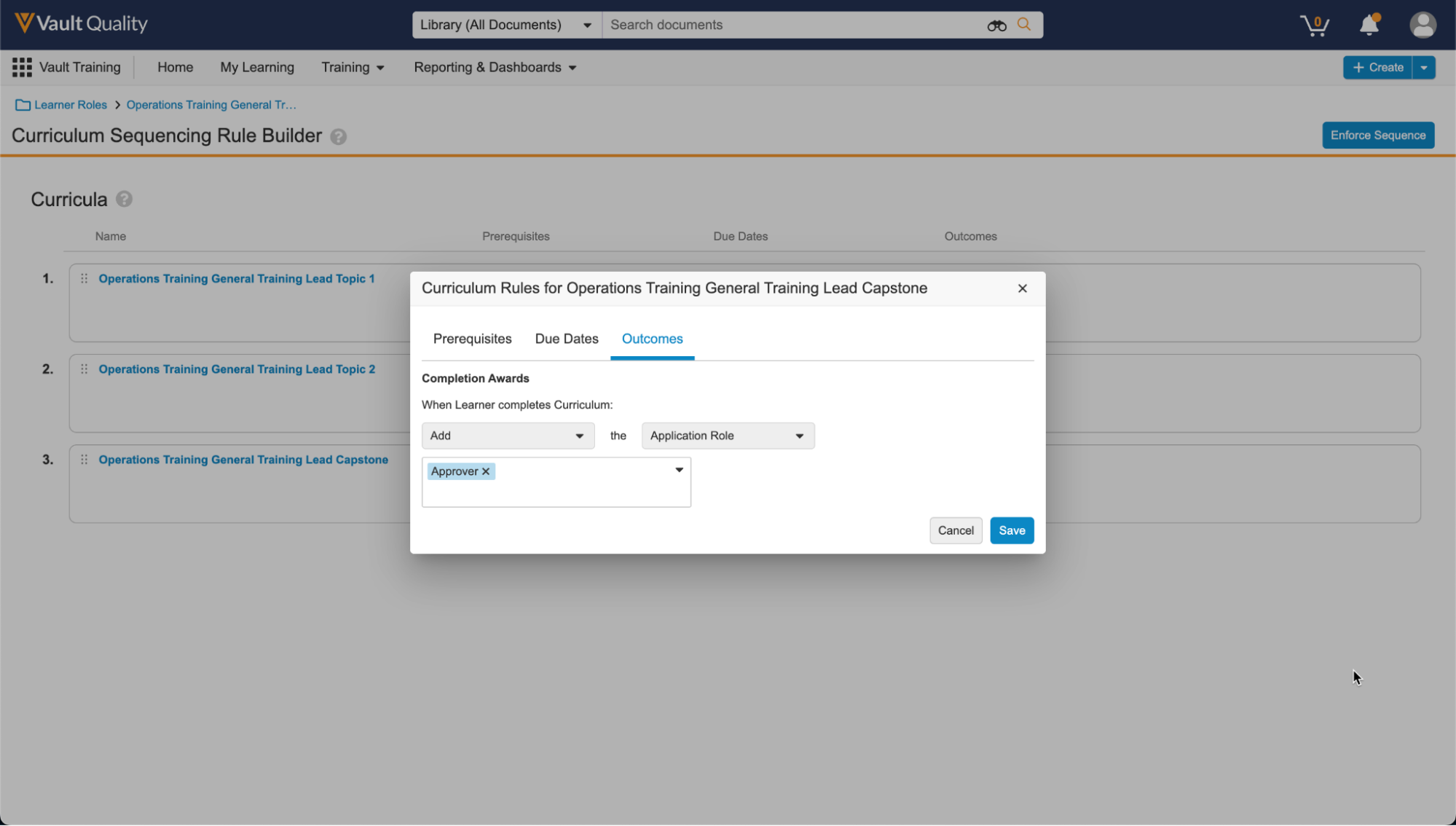
トレーニング管理者は、特定のカリキュラムにアクセスする前に完了する必要がある別のカリキュラムを示す、カリキュラム間のトレーニングルールの前提条件を作成できます。また、前提条件が不完全なままロックされたカリキュラムが期限切れにならないように、期日をオフセットする機能も導入されています。Curriculum 前提条件を完了すると、Vault はロック解除された Curriculum のトレーニング課題に期限を設定します。この機能は 23R3.2 で最初にリリースされ、23R3.4 で拡張され、代替のトレーニング要件を含むカリキュラムの前提条件ルールを定義できるようになりました。拡張機能には、Vault Loader または API を使用した前提条件ルールの移行のサポートも含まれます。
多くの場合、スキルは相互に構築され、特定の順序で完了する必要があり、これは習得型または能力ベースのトレーニングとしても知られています。たとえば、学習者はより困難なプロセストレーニングに進む前に、Safety 101 を受講する必要がある場合があります。Curriculum 前提条件は、特定の学習アプローチを適用し、手動のアサインメント時間を短縮します。
トレーニング管理者の画面:
学習者の画面:
LearnGxP ContentDirect
この機能により、LearnGxP コースライブラリに登録しているお客様に対するコースおよび更新の配信がシームレスに自動化されます。LearnGxP チームがコースを更新した時にその変更内容が自動的にお客様の Vault に反映されるため、手動介入が不要となり貴重な時間を節約できます。
Curriculum Completion Outcomes: Grant Additional Permissions
この機能では、カリキュラム完了時に学習者に追加権限を自動的に付与するルールを管理者が作成できるようになります。多くの場合、システムやビジネスプロセスへのアクセス (たとえば、ドキュメントを承認するための QualityDocs へのアクセス) は、トレーニングの完了に基づいています。このリリースでは、特定のカリキュラムを完了すると、Vault Training が学習者の User Role レコードを自動的に作成できます。
Supplemental Materials
Supplemental Materials は、トレーニング資料に追加できるオプションの参照トレーニング資料であり、トレーニング要件に追加して必要な資料を補うことができます。たとえば、ジョブ支援、ジョブマニュアル、フォーム、作業手順などのトレーニング資料は、必要な手順やポリシーの補足にすることができます。補足資料の更新には、再トレーニングやトレーニング要件インパクトアセスメントは必要ありません。
Quiz Support for Mobile Web & Mobile App
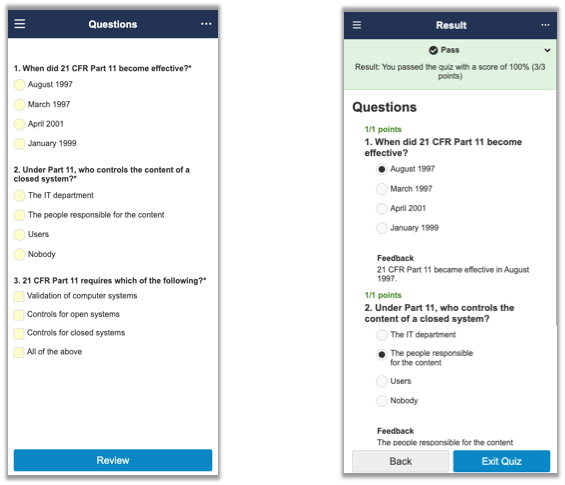
この更新により、クイズの表示形式がモバイル版ウェブおよびアプリケーションに対応するようになりました。これまでは、モバイルデバイスでクイズに入力する場合もデスクトップ版が表示されていました。今回の更新では、ユーザインターフェイスがモバイルデバイス用に最適化されました。
Assign, Pause, or Cancel Training Assignments Using Learner Role-Person Lifecycle
学習者ロール-パーソンライフサイクルとその他の設定により、Vault Training は、Learner Role-Person レコードのライフサイクル状態を使用して、トレーニング課題を割り当てるか、一時停止するか、キャンセルするかを決定できるようになりました。たとえば、New Hire Start Date フィールドは、学習者ロールの課題をずらすライフサイクルアクションをトリガーしたり、Clinical Study の Start Date と End Date によって課題の開始日と終了日を決定したりすることができます。
Add to Calendar Link: Training Assignment Link in Calendar Invite
Add to Calendar link を使用して Google または Outlook のカレンダーにクラスを追加した学習者は、会議の招待状から関連する Classroom Training 課題に直接アクセスできるようになりました。
Curriculum Page: User Interface Updates
Curriculum Page には、学習者が Self-Enrollment Curriculum Page の Card ビュー (Open タブ) または Explore タブから自己登録のトレーニング課題から登録を解除できる機能など、使いやすさのためのユーザインターフェースの更新がいくつか含まれています。
ユーザは、Begin Training ボタンをクリックする代わりに、トレーニング課題へのハイパーリンクをクリックしてトレーニングを開始できるようになりました。さらに、新しい Curriculum Page のブレッドクラムと All Open アイコンにより、ナビゲーションが簡単になりました。
Learner Homepage: User Interface Updates
次の場合、学習者ホームページの Card ビューの Begin Training ボタンラベルが View Training と表示されるようになりました。
- Evaluation Training Assignment が In Evaluation または In Evaluation Preparation 状態の場合
- External Training Assignment が In Verification 状態の場合
また、学習者が名簿に追加されず、自己登録が許可されていないクラスルームトレーニング課題については、以前はリストビュー、学習者ロール、カリキュラムページでハイパーリンクされていました。これらのクラスルームトレーニング課題は表示されますが、ハイパーリンクは表示されなくなりました。
Training Requirement Prerequisites: User Interface Updates
前提条件のある Training Assignments をナビゲートして完了するユーザエクスペリエンスが、以下のように改善されました:
- 学習者ホームページの Curriculum ビューと Learner Role ビュー: Training Assignment に前提条件となる Training Requirement がある場合、Vault は期日をグレーではなくステータスに応じた色で表示します。また、ロックされた Training Assignments への青色のハイパーリンクは無効になります。
- 学習者ロールページ: Training Assignment に不完全な前提条件の Training Requirement がある場合、Vault はそれをロックアイコンで示します。また、ロックされた Training Assignments への青色のハイパーリンクは無効になります。
- 学習者ホームページの List ビュー: Training Assignment に不完全な前提条件の Training Requirement がある場合、ロックされた Training Assignment への青色のハイパーリンクは無効になります。
- 前提条件である Training Requirement または Curriculum の結果としてロックされたすべての Training Assignments は、黒字で表示されます。さらに、ユーザーがこれらの課題にカーソルを合わせると、「前提条件が不完全なため、このコースはロックされています」にテキストが更新されます。
前提条件のある Training Assignments のリストで、Vault は完了した順にそれらを表示し、最後に完了した課題をリストの最後に表示します。以前は、Vaultは現在の課題をリストの最後に表示していました。
Evaluation Training Assignment: Create Checklists for Learners without a Vault User
学習者の Person レコードの Person is not a Vault user フィールドに示されている、Vault ユーザではない学習者のトレーニング課題に対して評価チェックリストを作成できるようになりました。
Document Training Assignments: Show Additional Document Toolbar Options
Training Assignment またはライブラリからドキュメントを表示する際に、一貫したユーザエクスペリエンスを維持するために、フルスクリーン、注釈の表示、および表示オプションのすべてのアイコンが、Vault ドキュメントの Training Assignment から使用できるようになりました。
Training Assignment State Change: Ignore State Of Evaluation Checklist Design
トレーニング課題の状態が更新されると (例: キャンセル済み)、この機能により、Vault は関連する Evaluation Checklist Design レコードのライフサイクル状態を無視することができます。以前は、Evaluation Checklist Design レコードが Draft のときに、 Training Assignment レコードの状態を変更できませんでした。
Training Requirement Impact Assessment: Copy Training Impact from Document
この機能は、Training Impact フィールドの値をドキュメントから Training Requirement Impact Assessment (TRIA) レコードに自動的にコピーします。これにより、Training Impact = No の場合に、ビジネスプロセスに基づいて TRIA レコードを自動的に終了することができます。
Auto-Manage Persons: Exclude System Users from Auto-Creation
Java SDK サービスアカウントやアプリケーション所有者などのシステムユーザアカウントは、パーソンの自動管理による処理から除外されます。
Curriculum SmartMatch Renamed to Curriculum Matching
このリリースでは、以前 Curriculum SmartMatch と呼ばれていた機能が、Quality アプリケーション設定、オブジェクトフィールド、アプリケーション管理などを含む Vault UI で、Curriculum Matching にラベル変更されました。
Curriculum Matching Rules に基づいて Curricula のサブセットを Learner Roles に割り当てる方法の詳細については、カリキュラムのマッチング設定を参照してください。
Instructor-Led Training: Auto-Cancel Workflow Tasks when Instructor is Updated
この機能は、インストラクターが更新された場合に以前のインストラクターのワークフロータスクを自動的にキャンセルします。この機能が導入される前は、これらのワークフロータスクは手動でキャンセルする必要がありました。
Instructor-Led Training: Improved Error Message when Adding Instructor
ユーザアカウントが無効なインストラクターがクラスに追加されると、エラーメッセージが明確かつアクション可能になります。
Training Matrix & Other Limits Updated
Vault のパフォーマンスを維持するために、Vault Training データモデル内のほとんどのトレーニングマトリックスオブジェクトはこれまでデフォルト制限の対象であり、Veeva サポートから選択制限の増加をリクエストするオプションがあります。この機能は、お客様の機能の導入、使用状況、フィードバックに基づいて、すべての Training Vault にわたるほとんどのトレーニングマトリックスオブジェクトおよび関係の制限を引き上げます。これらの制限が今後引き上げられる場合は、Vault のリリースを通じて行われます。たとえば、1 つのカリキュラム レコード内のトレーニング要件に対して定義できるトレーニング ルールの前提条件の最大数は 25 でした。24R1 ではこの制限がなくなり、トレーニング管理者は特定のカリキュラム内の任意の数のトレーニング要件に対して前提条件ルールを定義できるようになりました。
Data Model: New Fields & Objects
このリリースには、よく使用されるフィールドを標準化し、オブジェクトとその制限を Platform データモデルと調和させるための Training データモデルの更新が含まれています。これらの新しいオブジェクト、オブジェクトタイプ、フィールド、選択リストにより、Vault Training および Study Training のお客様と Veeva のベスト プラクティスとの一貫性が保たれます。詳細については、24R1 Quality データモデルの変更をご覧ください。
Standard Action Layouts for Training Objects
Learner Role、Curriculum、および Training Requirement オブジェクトの標準アクションレイアウトが追加されました。この各種レイアウトには、データをトレーニングマトリックスオブジェクトの各セクションおよびフィールドに整理する上でのベスト プラクティスが含まれています。管理者はこの各種テンプレートをコピーし、各自の用途に合わせてカスタマイズできます。
QMS
QRM: リスクビルダー: 表示する列
この機能により、組織はリスクビルダーのアセスメントリスク列の表示方法をより柔軟に選択できるようになります。
システム管理者は、リスクビルダーのデフォルトの列と、列の順序を設定できるようになりました。Assessment Risk コントロールの新しいダイアログウィンドウには、画面下部にフィールド選択が含まれており、選択したリスク評価オブジェクトタイプに対してデフォルトで表示される Assessment Risk フィールドを指定します。
システム管理者は、Assessment Risk セクションで、Allow users to override columns チェックボックスを使用して、ユーザが表示される列を変更できるようにするかどうかも設定できます。このオプションをオンにすると、ユーザは Edit Columns メニューにアクセスできるようになり、リスクビルダーに表示される列と、列の順序を変更できるようになります。
詳細については、リスクビルダーでアセスメントリスクを表示するアセスメントリスクコントロールの設定をご覧ください。
QRM: リスクビルダーの機能強化
このリリースでは、お客様からのフィードバックに基づいて、いくつかの新しいリスクビルダーの機能強化が導入されています。
Assessment Risk 緩和の管理
ユーザは、Assessment Risk を緩和するレコードをリスクビルダー内で直接表示、割り当て、削除できるようになりました。リスクを緩和するために使用できるレコードには次のものがあります。
- 緩和アクション
- Quality Event
- CAPA Actions
- 変更管理
- 継続的な改善
- MedTech CAPAs
以下のスクリーンショットは、リスクを緩和するために複数のレコードが割り当てられた Assessment Risk を示しています。割り当てられたリスク軽減レコードの横にある X をクリックすると、そのレコードは Assessment Risk から削除されます。プラス記号 (+) をクリックすると、ユーザはリスクを緩和するために、1 つ以上の既存のレコードを割り当てるか、新しいレコードを作成できます。各ユーザがリスクビルダーで列を閲覧するためには、システム管理者がアセスメントリスク緩和オブジェクトを有効化する必要があります。
Assessment Risk の削除と関連する Quality Team の削除
23R3 の時点で、ユーザは、チームメンバーが割り当てられた Quality Team がある場合に、その Assessment Risk レコードを削除することができません。このリリース以降、Vault では、Quality Team が定義されている場合でも、ユーザがリスクビルダーから Assessment Risk レコードを削除できるようになります。
その他の UI 機能強化
Vault では、Risk Assessment のヒートマップの Severity/Occurrence および Detectability 軸のラベルが自動的に折り返されるようになりました。これによって、ラベルが読めなくなるのを防ぎます。
QRM: 定期リスクレビュー
組織は、Vault QMS アプリケーションの品質リスク管理機能を使用して、事業および業務プロセスにおけるリスクを事前に特定します。リスク評価は、プロセス失敗モードと効果分析 (pFMEA) など、いくつかの一般的なリスク管理法に基づいてリスクを特定し、特徴付けるために使用されます。各 Risk Assessment には、Assessment Risk と呼ばれるリスクが含まれており、故障モード、結果、原因、制御、評価方法に基づくリスクのその他の特性などを説明します。Assessment Risk は、Risk Matrix に基づいて、Severity、Occurrence の可能性、および Detectability スコアを把握するために、評価前後のフィールドでスコアリングされます。
組織のビジネス環境が変化すると、事業および業務プロセスに生じるリスクも変化します。企業は Risk Assessments を定期的に見直し、現在の状態に基づいてリスクに適切に対処していることを確認する必要があります。
定期的な見直しを実行する場合、組織は Risk Assessment とその Assessment Risk のスナップショットの取得を希望します。これにより、企業はリスクが時間の経過とともにどのように変化したかを理解し、リスクにどのように対処し続けるかについての洞察を得ることができます。履歴情報を取得する現在のアプローチは、Vault のフォーマットされた出力機能を使用しており、これはリスク評価のスナップショットをリスク評価レコードの添付ファイルとして PDF 形式で保存します。過去のリスクデータを PDF 添付ファイルに保存すると、組織の次の能力が制限されます。
- 特定の時点でのリスクに対処するために、どの緩和品質プロセス (CAPA など) が使用されたかを確認する。
- リスク評価のリスクが一定期間にわたってどのように変化したかを効果的に確認する。
24R1 Release では、システム管理者が有効化できる新しい設定可能な機能を導入することでこれらの制限に対処し、組織が Risk Assessment とその Assessment Risk (リスクスコアと緩和アクションを含む) に関する履歴データを PDF ではなく構造化された形式で保存できるようにします。その結果、ユーザは、以前承認された Risk Assessment とその Assessment Risk を特定の時点で簡単に表示できます。これには次のようなものが含まれます。
- リスク評価概要
- 評価リスク
- アセスメントリスクスコア
- アセスメントリスク緩和アクション
企業は、Risk Assessment が定期的に見直されて改訂される際に、過去のリスク情報に簡単にアクセスできるという利点があります。
24R1 以降、Risk Assessment が承認されると Vault はスナップショット情報を自動的に取得します。
長期にわたって定期レビューが実施されて Risk Assessment が変更されると、ユーザはリスク評価レコードやリスクビルダーからその履歴を表示できるようになります。
このリリースでは、過去のリスクデータに加えて、定期レビュープロセスのための新しい機能が導入されています。ユーザは、承認済み Risk Assessment の定期レビューワークフローを手動で開始できるようになっています。
QRM: Recompute Risk Matrix
品質リスクマネジメント (QRM) を使用すると、組織は、リスク評価を使用する企業プロセスと運用プロセスに関連したリスクを管理できます。QRM の重要な要素は、次の主なリスクディメンションを定義するリスクマトリックスです: 重大度 (例: 小規模、中規模、大規模)、頻度 (例: 希少、時々、頻発)、および 検出可能性 (例: 低、中、高)。
リスクマトリックスは、定量的または定性的です。定量的リスクマトリックスは、リスクディメンションに数値を割り当て、リスクディメンションの可能な組み合わせごとにリスクスコアを自動的に計算します。計算されたリスクスコアは、リスクディメンションの積です。定量的リスクマトリックスでは、リスクスコアを分類できるように、しきい値のリスクレベルを定義することもできます。定性的リスクマトリックスはリスクスコアを計算することはありませんが、リスクレベルの手動の課題をサポートします。
リスクマトリックスが承認されると、リスク評価で使用され、初期リスクスコアと残余リスクスコアおよびリスクレベルが割り当てられます。22R2 より前に QRM を使用しているお客様は、関連するリスクマトリックスから初期リスクスコアおよび 残余リスクスコア (標準フィールド) を入力しない リスク評価を有する可能性があります。また、23R3 Releaseより前に作成されたリスク評価では、リスクマトリックスで定義された重大度スコアおよびレベルを入力しません。これは、お客様が 23R3 で導入されたヒートマップ機能を使用してリスク評価をグラフィカルに視覚化したい場合に問題になります。ヒートマップ機能は、Risk Level および Criticality Level フィールドを使用する Assessment Risk レコードを含むリスク評価に依存します。ただし、そのマトリックスを使用してリスクマトリックスとすべてのリスク評価を更新することは困難です。組織が手順を自動化する方法を提供することで、この機能における問題は解消します。
管理者は、リスクマトリックスのライフサイクル状態におけるユーザアクションを設定して、Risk および Criticality フィールドをリスクマトリックス、およびリスクマトリックスが使用されるリスク評価内で、すべての Assessment Risk レコードを非同期的に更新します。以下のスクリーンショットは、リスクスコア および リスクレベルの自動化に関するユーザアクションの内容を示しており、Assessment Risk レコードの Risk Score および Risk Level フィールドが、該当するリスクマトリックスで定義されたリスクスコアとリスクレベルに基づき入力されます。
監査実行のチェックリストの機能強化
このリリースでは、Vault QMS での監査の実行のサポートに特化したチェックリストが機能強化されています。ユーザは、新しい監査チェックリストタイプのチェックリストを利用して再利用可能な構造化された質問票を定義し、詳細な回答や添付ファイルを取得し、所見や観察の直接提出をサポートできるようになりました。質問への回答とそれらの回答に関連する所見は、チェックリストを通じて、または直接所見として、監査に自動的にリンクされます。
この直接入力により、監査自体が Vault QMS 内のチェックリストとしてサポートされ、監査の実行における Word 形式のドキュメントの必要性が軽減または排除されるため、監査人が監査結果をコピーして Vault QMS に貼り付ける必要性が排除または大幅に軽減されます。
さらに、監査人がこれらのチェックリスト内で監査の実行状況を把握するため、組織は、潜在的な所見を明らかにするのに最も効果的な監査 (さらには監査内の特定の質問や手順) について分析または報告することで、さらなる利益を得ることができます。
Vault のチェックリスト機能に慣れているユーザは、シンプルで直感的な新しいアクションが追加されているため、簡単に操作できます。
ユーザは、監査チェックリストが監査記録の監査タイプ、方法、カテゴリ用に設定されている場合、監査チェックリストを開始できます。ユーザがチェックリストの完了を失念したり他のことに気を取られたりしないようにタスクを作成できます。
監査チェックリストは監査に関連付けられており、監査の閲覧者または報告ユーザが直接利用できます。
簡単なアクションで、チェックリスト内の任意の質問から所見を直接提出できます。
所見の提出はチェックリスト内で実行され、組織の所見の定義、関連するメタデータ、設定に基づいて動的に行われます。質問またはステップごとに複数の所見がサポートされます。
最後に、チェックリスト内の各質問または手順に関連付けられたすべての所見が保存され、チェックリストインターフェース自体と Vault の両方に表示されます。
この機能を使用するには、新しいエンティティがユーザに確実に表示されるようにするため、監査チェックリストの設定と、監査および所見の関連ページレイアウトの更新が必要です。詳細については、監査チェックリストをご覧ください。
関連イベントアプリセクション
このリリースでは、Quality Event またはスタンドアロン Quality Event (逸脱またはクレームなど) の種類に関係なく、関連する Quality Event レコードを管理および表示するためのシングルストップショップが導入されました。この新しいアプリセクションにより、Vault QMS は、名前、タイトル、タイプ、ライフサイクル状態、関連レコードからのレコードチェック結果などの重要な情報を簡潔かつ詳細な形式で表示できるようになりました。この機能強化は、高品質イベント用のスタンドアロンデータモデルを導入済みの組織、導入中の組織、または導入を計画している組織にとって特に価値があります。例:
この機能が導入される前は、基本的な Vault オブジェクトレコード詳細ページの関連レコードセクションでは、関連レコードを視覚化する最適なレイアウトは少数の表が配置されたレイアウトであり、すべてのデータを表示するには水平スクロールが必要でした。
この機能を使用すると、データが収集および要約されます。レコードがどのタイプの品質イベントであるかに関係なく、関連するレコードに関する重要なデータがいくつかの列に表示されます。
適切な権限を持つユーザは、ページレイアウト上の 1 つのセクションから、データモデルに関係なく、表示しているレコードに関連する Quality Event レコードを識別して管理できるようになりました。
新しいセクションでは、リンクされたレコードの作成、削除、変更が可能です。Related Events アプリケーションセクションは、Quality Event オブジェクトまたはスタンドアロン Quality Event オブジェクトの任意のページレイアウトに追加できます。また、現在のレコードに関連するオブジェクトのレコードを視覚化し、それらとの対話を可能にしたり、その逆の操作を可能にしたりします。このアプリケーションセクションを介してレコードをリンクするユーザは、単一の関係レコードを作成するだけです。Vault は逆リンクレコードを作成し (削除の場合は管理し)、ユーザがどのレコードを表示しているかに関係なく、リンク後に、このセクションで関係を確認できます。
Related Events グリッドでのレンダリングをサポートし、他のイベントタイプと一致させるために、Change Plan オブジェクトにはオブジェクトに追加された新しい標準フィールド、Title が存在します。
この機能には設定が必要です。変更がユーザに表示される前に管理者がアプリケーションセクションを目的のページレイアウトに追加する必要があるからです。詳しくは、関連イベントセクションの設定をご覧ください。
Enable Adverse Event Reportability Assessment in QMS
Vault QMS のお客様は、報告可能性評価を完了することで、苦情を保健当局に報告する必要があるかどうかを評価できるようになりました。これは、Vault Product Surveillance のお客様のみが利用できる機能でした。Complaint オブジェクトの Pharma Complaint または MedTech Complaint オブジェクトタイプのいずれかに接続する Reportability Assessment オブジェクトを使用すると、組織は苦情の重大度を判断するための質問を設定し、保健当局への有害事象の報告可能性を判断できます。重大度と報告可能性は、Complaint を扱うユーザによって提供される報告価値評価の回答に基づき決定されます。
この機能により、QMS のお客様は苦情の重大度、報告決定ルール、および 製品の市場に基づいて、そのイベントが報告可能であるかどうか、どの保健当局に報告するかを決定することができます。有害事象ドキュメント生成の自動化、または有害事象の提出をサポートするツールに関心をお持ちの場合、Vault QMS の姉妹品である Vault Product Surveillance アプリケーションを確認することをお勧めします。
Quality Incidents: Create a Quality Incident from a QR Code
組織は、従業員や受託業者が日常業務を遂行する際に、品質に関する文化を確立するために懸命に取り組んでいます。これには、製品またはプロセスの品質に影響を与える可能性のあるインシデントを報告する必要性を重視することが含まれています。これは、製造オペレーションに関与する領域では特に重要です。
Vault QMS を使用すると、ユーザはインシデント発生時に既知の情報を取得する Quality Incident レコードを作成できます。また、適切な担当者がインシデントを優先順位付けして本格的な品質イベントプロセス (逸脱、不適合など) が必要かどうかを判断できるライトなプロセスも含まれています。従業員が Quality Incident を迅速かつ簡単に作成できるようにすることは、それらの確実な報告のために不可欠です。このリリースより前は、Quality Incident の作成は、標準の Vault QMS ユーザインターフェースを介してのみ可能でした。このリリースでは、モバイルデバイスを使用して Quality Incident を作成する、簡単で負荷の少ない方法が導入されています。
システム管理者は、印刷して従業員や受託業者が作業する場所に配置できる QR コードを設定できるようになりました。Vault QMS ユーザは、モバイルデバイスのカメラで QR コードをスキャンし、遭遇したインシデントに関する必要な情報を収集するスマートなページに記入できます。担当者は Vault QMS のライセンスを持っている必要があり、アクティブなセッションが確立されていない場合は、Vault にログインを求められます。この機能はモバイルデバイスのブラウザを利用します。詳細については、サポートされているブラウザをご覧ください。
これは、Verteo Biopharma の Vault から自動的に生成されてダウンロードされた PDF ファイル内の QR コードの例です。同社は PDF ファイルを印刷し、ダブリンの施設の製造エリア内の目立つ場所に掲示しました。
QMS ユーザがモバイルデバイスで QR コードをスキャンして Vault にログインすると、次のような画面が表示されます。組織、施設、および部門の値は QR コードからデフォルトで設定されます。ユーザは必須の Date Occurred フィールドと Title フィールドに値を入力し、オプションで Summary を入力します。Quality Incident フィールドに記入することに加えて、ユーザは最大 5 つの添付ファイルを追加できます (添付ファイルあたり最大 20 MB)。Quality Incident 添付ファイルには、通常、遭遇したインシデントを示す写真または短いビデオが含まれます。
Quality Incident 情報を提供したら、ユーザは Submit をタップします。以下のスクリーンショットは、ページが更新されて、Vault で Quality Incident が作成され、ユーザが必要に応じて別の Quality Incident ファイルを作成できるようになる様子を示しています。
Vault に品質インシデントレコードが存在するようになったため、ユーザは標準の Vault QMS ユーザインターフェースを使用してトリアージプロセスに参加できます。以下のスクリーンショットは、作成直後の Vault の Quality Incident レコードを示しています。詳細については、品質インシデントの使用をご覧ください。
システム管理者は、品質インシデント取り込みレイアウトを設定して、ページに表示される最大 10 個の品質インシデントフィールドとその表示順序を指定します。
次に、ビジネス管理者が、組織の業務のさまざまな分野に関連付けられた QR コードを表す Quality Incident Configuration Data レコードを作成します。各 Quality Incident Configuration Data レコードは、Quality Incident Intake Layout 設定を参照します。QR コードは、Quality Incident レコードの次のフィールドの任意の組み合わせの特定の値に関連付けることができます。
- Country (上記のサンプルのスクリーンショットには表示されていません)
- Organization
- Facility
- Department
上の例では、Verteo のシステム管理者が Quality Incident Intake Layoutを作成し、それには、Organization、Facility、Department、Date Occurred、Title、Summary の各フィールドが含まれています。Verteo の企業管理者は、このレイアウトを使用して、次のフィールドにデフォルト値を指定することで、ダブリン施設の Verteo の製造エリアで使用する QR コードを作成しました。
- Organization = Verteo Biopharma
- Facility = Dublin
- Department = Manufacturing
企業管理者が QR コードを生成するユーザアクションを実行すると、Vault によってダイアログに QR コードが表示されます。企業管理者は、ダイアログ画面を使用して QR コードの静的 URL をコピーできます。静的 URL をユーザと共有すると、デスクトップブラウザ経由で同じ品質インシデント作成ページにアクセスできます。
詳細については、品質インシデント QR コードの設定と管理をご覧ください。
External Notifications Enhancements: Multiple Recipients & Add Document from Library
組織は、多くの場合、外部通知を使用して、製品の苦情について社外の関係者とメールで連絡します。対応には、初回報告者、訴人、医療機関など、Complaint レコードに特定された複数の人物が関与することがよくあります。
このリリースより前は、ユーザはレコードで識別された Person ごとに 1 つの External Notification を開始する必要がありました。24R1 Release では、管理者はユーザが 1 つのメールをレコードの複数の Person に送信できるようにする External Notification を設定できます。たとえば、以下のスクリーンショットは、Vault ユーザが Complaint レコードの Complainant フィールドおよび Reporter フィールドで特定された個人に苦情を受け取ったという確認通知を送信できるようにする外部通知設定を示しています。
上記の外部通知設定は、Complaint レコードを操作しているユーザが外部通知を送信するときに次の画面が表示され、送信者が複数の外部の個人を選択できることを意味します。
外部関係者とのやり取りでは、Vault で作成、確認、承認された複数のドキュメントを添付する必要があることがよくあります。このリリースより前は、External Notification は、レコードのフィールドで特定されたドキュメントを含める設定のみ可能でした。24R1 を使用すると、システム管理者は、ユーザがドキュメント上のフィールドを介したレコードを参照するドキュメントを送信できるようになります。
たとえば、ドキュメントフィールドで特定された特定の Complaint レコードに対して書かれた承認済みの通信文書を考えてみましょう。
システム管理者によって有効にされている場合、外部通知を送信するユーザは、レコードを参照するドキュメントを含めることができます。たとえば、上記のドキュメントを Complaint レコードの外部通知に含めることができます。
この機能強化により、システム管理者は、必要な受信者とドキュメント添付ファイルのさまざまな組み合わせを処理するために必要な External Notification 設定の数を減らせるようになります。その結果、組織はユーザに提供する External Notification メニューオプションを減らし、すべての通信要件に柔軟に対応できるようになります。
設定の詳細については、外部通知をご覧ください。
5 Whys Analysis: Support for Dynamic Reference Constraints
23R2 では、なぜなぜ分析の静的参照制限をサポートするための機能強化が行われました。
このリリースでは、動的参照制限をサポートする 5 Whys Analysis ツールの改善を進めています。これにより、ユーザは Root Cause オブジェクトの別のフィールドの選択に基づいてオブジェクト参照フィールドをフィルタリングできるようになります。
このツールの強化も行われ、リッチテキスト、数値、ルックアップのデータ型がサポートされるようになりました。さらに、セキュリティが強化され、Root Cause Analysis Lifecycle の Atomic Security Relationships 設定に基づいて、Why カードを追加または削除できるかどうかをお客様が定義できるようになりました。また、ツール自体内でユーザが表示および実行できる内容は、5 Whys analysis 関連オブジェクト (Root Cause Analysis、Root Cause Analysis Item、および Root Cause) に対するユーザ権限によって決定されることが多くなります。
Duplicate Check: Check Lifecycle State Processing Enhancement
このリリースでは、QMS Duplicate Check タイプのレコードチェックが強化され、レコードが重複として識別されても識別されたレコードの状態が変更されない一連の状態を示すようになりました。この設定可能なフィールド「Do not transition complaints in the following lifecycle states」を使用すると、組織は、特定の Duplicate Check 設定に対して、以前にキャンセルされたレコード、クローズされたレコード、または必要に応じて、ほとんどの処理済みレコードを現在のライフサイクル状態で保持しながら、識別されたレコードを重複として取得し、リンクできます。
この機能を有効にするには設定が必要であり、Duplicate Check タイプのレコードチェックにのみ適用されます。
Duplicate Check: Update Summary Page to Accurately Reflect Lifecycle Transitions
このリリースでは、お客様ベースからのフィードバックに基づいて、Duplicate Check の概要ページに機能強化を加えています。自動的に有効になるこれらの機能強化は、Duplicate Check 中に実行されたアクションの結果として、どの操作がどのレコードに影響を与えるかをより明確にすることを目的としています。
Record Check Enhancements: Capture Suggested Terms & UI Enhancements
このリリースでは、QMS レコード チェックが強化され、一致用語と推奨用語に関するサポートが向上しました。一致用語 (管理画面で「Fields on object to compare in text search」として表示) を活用するレコードチェックについては、チェック時に使用される推奨用語が、レコードチェック実行の一環として生成される Record Check Result および Record Check Rule Detail レコードで取得され、保存されるようになりました。これにより、一致の確認に使用された用語に関するより明確な履歴が提供されます。
この動作は、一致用語をすでに利用しているレコード チェックに対して自動的に有効になりますが、これらの新しい推奨用語の取得は、このリリース以降に実行される新しいレコード チェックでのみ使用可能になります。
推奨用語の取得と UI 機能強化に加えて、ユーザがレコードチェックを完了しても、レコードチェックの結果フィールドおよびレコードチェックの結果状態フィールドは更新されなくなりました。この変更は、ユーザが再帰性チェックの完了直後にフィールドを編集しようとした場合のエラー排除を目的としています。
Quality Teams 関連オブジェクトセキュリティ
QMS では、Quality Teams を使用して、ユーザを特定のプロセスレコードのロールに手動で割り当てます。プロセスレコードには、Quality Team が有効になっていない他のオブジェクトとの関係もあります。現在、これらの関連レコードが Quality Team メンバーによって作成された場合に誰がそれらを変更できるかを確実にする方法はなく、作成したユーザのみが編集または削除できます。
ユーザが Quality Team メンバーでなくなった場合、すべてのユーザがオブジェクトを変更できるようにする、よりオープンなセキュリティモデルが実装されない限り、他のユーザはその関連レコードを削除または編集できません。
たとえば、Deviation レコードで、所有者が関連するアセットレコードを Deviation - Asset セクションに追加し、その 2 日後、逸脱がまだレビュー中である間にその所有者が会社を辞めたとします。
Quality Team の新しく割り当てられた所有者が関連するアセットレコードを削除または編集する必要がある場合、その所有者には Deviation - Asset レコードの共有設定のロールがないため、削除または編集することはできません。
この新機能は、レコードロールのオーバーライドを利用して、Quality Team によって管理されていないレコードへの動的なアクセスを実現します。これにより、Quality Team メンバーは、安全かつスケーラブルな方法で、Quality Team 有効化プロセスに関連付けられた関連レコードを動的に編集または削除できるようになります。
前述の例では、管理者が Quality Team の設定でレコードロールのオーバーライドを設定しているため、新しい所有者に関連オブジェクトレコードのロールが自動的に割り当てられます。これにより、退職した前の所有者によって作成された古い関連レコードを編集または削除できるようになります。
セキュリティ保護の関連オブジェクトは、次のオブジェクトに対して構成可能です: APQR、APQR項目、評価リスク、評価、監査、監査者プロファイル、監査者ロール、CAPA アクション、変更アクション、変更アクション テンプレート、変更管理、苦情、封じ込め、継続的改善、是正、逸脱、有効性チェック、延長要求、調査結果、影響評価、調査、問題エスカレーション、ラボ調査、MedTech CAPA、緩和措置、不適合、組織、製品返品、提案された監査、品質イベント、QMR、QMR 項目、認定、品質インシデント、リスクイ ベント、リスク登録、根本原因分析、SCAR、SCN 影響評価、サプライヤー変更通知、および テスト計画。
上記のオブジェクトに関連する標準オブジェクトとカスタム オブジェクトも、保護する関連オブジェクトとして使用できます。品質チーム関連のオブジェクト セキュリティについて詳しくご覧ください。
Cascade Delete Quality Team Members
自動的に有効になるこの機能は、チームチーム有効化レコードのシングルアクション削除を有効にすることで、Quality Teams の設定 (通常は Sandbox または QA 環境) でのレコード削除の必要性を効率化します。この機能が導入される前は、削除するレコードに Quality Team メンバーが関連付けられている場合、レコードの削除は不可能であり、先に Power Delete Quality Team Members レコードアクションを実行し、その後で問題のレコードの削除を試みる必要がありました。この 2 段階のプロセスは、特に大規模な QA 環境または Sandbox 環境でテストデータを設定しようとする場合、煩雑になる可能性があります。さらに、このような操作はチームメンバーの継承が子レコードに対して設定されている設定では複雑になり、削除するレコードの「ツリー」を下まで分析し、それぞれに対してパワー削除アクションを実行し、最後に自分でレコードを削除する必要があります。
この機能により、詳細な分析要件が排除され、チーム有効化レコード削除の作業を制御する設定自体への依存が導入されます。チーム有効化レコードを削除する権限を持つユーザは、関連付けられたチームメンバーの存在によってブロックされなくなります。関係の設定が子レコードのカスケード削除の関係削除ルールを示している場合、削除対象のレコードからチームメンバーシップを継承する子レコードも削除できます。トップレベルチーム有効化レコードが削除され、Set field to blank when related record is deleted の関係削除ルールが設定されたサブプロセスレコードが存在する場合、これらのサブプロセスレコードの割り当て (およびレコード自体 - null に設定されている関係フィールドを除く) は、削除の影響を受けません。
この機能は、チーム有効化レコードの削除が設定によって許可されている設定とユーザにのみ影響します。
Quality Components Configuration Migration Improvements
この機能は、QMS Vault の拡張機能として自動的に有効化される、管理者にとって歓迎すべき機能です。Vault Package (VPK) 移行の実施に慣れている Vault 管理者は、Sibling Check Related Record State Change などのアクションに関するいくつかの課題にも精通している可能性があります。状態に属するアクションとして、別のライフサイクル状態を参照する可能性があり、循環依存関係が作成され、VPK の移行を元に戻すのが非常に困難になる場合は特にあてはまります。
この機能では、Vault QMS のコンポーネントアクションの依存関係を考慮した新しい VPK 機能が導入されています。VPK のインポート中に既存の Allow deployment of blocked steps パラメータを使用すると、Vault が適切なプレースホルダーコンポーネントを作成できるようになり、この種のエントリアクションを含む VPK のインポートはエラーなしで成功します。
さらに、適切な名前を持つプレースホルダコンポーネントの自動作成をサポートする変更が導入され、後続の VPK または手動設定によって更新できるため、次のコンポーネントのいずれかを含む VPK 操作が効率化されます。
qualityrecordcheck__vqualityrelatedrecordmatchfield__vqualitymatchtier__vqualityrelationshipstatemap__vqualityrecordcheckrule__vqualityteam__sysqualityteamrole__sysqualityteamrolemembershiprestriction__sysqualityteamrelatedobjectsecurity__sys
Standard Action Layout on Deviation Object
今回のリリースでは、Vault アクションレイアウト機能が導入されました。Vault アクションレイアウトは、Vault をご利用のお客様が、Vault レコード操作時に堅牢、動的かつ柔軟なユーザインターフェイス エクスペリエンスを提供することを目的とした、幅広い機能の集合体です。この有用な機能を学びたいと希望している Vault QMS 管理者をサポートするため、QMS Vault に Deviation オブジェクト用の 2 つの新しい標準アクションレイアウト、GMP Deviation Detail Page (Full View) と GMP Deviation State Layout が追加されました。この 2 つのアクションレイアウトは、既存の設定やエンドユーザと干渉しないよう、無効ステータスで展開されます。このアクションレイアウトは、管理者が Vault のレコードベースソリューション上でレイアウト規則、レイアウト表現および表示エフェクトに則り、関連するメタデータのみを適切なタイミングでユーザに表示できるよう、ベスト プラクティスに基づくガイドや例の役割を果たすものとして事前設定されています。このようなサンプルレイアウトは、Vault QMS のアクションレイアウトで実現できる内容の一部にすぎません。
開始するには、まず管理者は無効ステータスになっている上記アクションレイアウトのコピーを作成し、そのコピーを使用して Vault 上でアクションレイアウトに利用できる機能を確認することができます。このレイアウトはすべての Vault にプロビジョニングされますが、設定管理者以外のユーザが閲覧できるようにするには設定が必要です。
Surveillance
Regulatory Updates for eMDR
近年 (2023 年 8 月 /9 月) FDA が eMDR の提出処理に加えた変更を考慮して、Vault Product Surveillance は、規制に準拠した中断のない eMDR およびフォーム 3500A 生成をお客様に保証するためにいくつかの変更を導入しました。変更および影響の概要:
| 規制変更 | 影響 |
|---|---|
| フォームコードが FDA によって受け入れられなくなった | フォームコードが、有害事象オブジェクトで非アクティブ化され、フォーム生成から削除されました。 |
| 患者のジェンダーと患者の性別が更新され、3500A で区別されるようになった | ジェンダーと性別の区別に対応し、関連する選択リストの値を追加するため、苦情オブジェクトのフィールドが更新されました。 |
| FDA アクションコードの長さは最大 50 文字をサポート | AER オブジェクトの FDA アクションコードの最大文字長が 50 文字に増加しました。 |
| 3500A セクション H10 のサポートの追加: このフィールドにより、送信者は同じイベントに関する 1 つ以上の他の MDR (製造元 MDR と同じイベントを参照するユーザ施設または任意の MDR など) を識別できるようになります。 | 新しい Related Adverse Event Reports オブジェクトと Related eMDR セクションが追加され、フォーム生成にマッピングされました。 |
| 7 桁のシーケンス番号を持つ製造元および輸入業者の MDR のサポート | レポート番号でサポートされる最大桁数が増加しました。 |
| セクション H3 が簡略化された | セクション H3 を 4 部から 1 部に減らしました。 |
有害事象報告の UI の機能強化
このリリースには、有害事象報告 (AER) 機能への継続的な投資が含まれています。これにより、Vault Product Surveillance で有害事象に関連するフィールドを編集する際に、明確性が高まり、冗長性が低くなります。全般的な概要として、関連レコードのフィールドが VPS 内の AER ページに追加され、そのページから直接追加または管理できるようになりました。一般に、これらの影響を受けるセクションの多くは、関連するデータ、アクティビティ、またはコンテキストの複数のエントリを AER に関連付けるサポートを中心に展開されます。現在、eMDR および EU MIR レポートのいくつかの変更が利用可能です。
eMDR AER では、主に、さまざまなセクションでのこのデータの取得と管理を中心とした変更が行われています。例:
セクション B6 (上で強調) は、複数のテストの追加をサポートし、AER とのインラインでのそれらのテストのレンダリングをサポートするようになりました。D10 の併用医療製品と治療日、および H6 の有害事象コードにも同様の処置があります。
また、EU MIR AER は、これまで埋もれていた関連情報リストの視覚化から、eMDR エクスペリエンスに似た関連項目の表示と管理や、セクション 4.3.c の類似のインシデントの時間範囲の定義についての改善された新しい処理方法に至るまで、さまざまな更新を受けています。以下にいくつかの例を示します。
セクション 2.3 p (上記) と 2.5 (図示せず) の両方に、それぞれ、指定機関製品 (選択された製品に基づく)、および市販品表 (苦情からの製品バリエーションに基づく) からの関連情報が表示されるようになりました。列は保健当局のフォームのフィールドに基づいています。
セクション 2.6、3.2 a、3.3 a、4.2 e、f は、すべて、それぞれのフィールドの複数の値のインライン表示、追加、管理をサポートするようになりました。
EU MIR のセクション 4.3.c も機能的に改善され、類似のインシデントの時間枠を定義するために必要なクリック数が減り、それぞれの時間枠で地域ごとに発生した類似のインシデントの数がより明確になりました。
類似のインシデントの時間枠と類似のインシデントの概要は、利用と管理を容易にするために表形式で表示されます。
類似のインシデントの時間枠は、AER 自体の中から直接、改良された新しいポップアップ UI で個別に編集できます。
Feedback-Based Enhancements
このリリースには、Vault Product Surveillance の早期アダプタープログラムの参加者からのフィードバックに直接基づいた、いくつかの機能強化が含まれています。これらの変更により、既存のプロセスが強化され、いくつかの問題点と冗長な要素がソリューションから削除され、有害事象レポートの管理プロセスがよりシンプルで信頼できるものになりました。
まず、関連する ACK2 メッセージの前に ACK3 メッセージを FDA から受信するという特定の eMDR 送信シナリオがより明確に処理されるようになりました。ACK3 は ACK2 が存在するかどうかに関係なく保存されるとともに、「孤立した」ACK3 は新しい Vault ジョブによって処理され、ドキュメンテーションの証跡が明確になります。
次に、報告価値評価と再評価のロジックが更新されました。以前のリリースでは、有害事象レポートの完了から終了タイプへの移行が自動的にフォローアップレポートをトリガーしていました。このリリースでは、AER の実行中に報告価値の再評価が実行された場合、この移行中にのみフォローアップレポートがトリガーされます。この変更により、AER の終了中に処理が必要となる無関係または価値の低いフォローアップレポートが減少します。
さらに、eMDR のセクション G3 内で、報告者の詳細が Complaint オブジェクトと MedTech Complaint オブジェクトとの共有から統合されました。それにより、ユーザは、Complaint オブジェクトの関連するすべての報告者の詳細 (職業、連絡先情報、施設など) を検索できるようになりました。その結果、AER オブジェクトのDate Received by Mfg (date_received_mfg__v) がアクティブになり、セクション G3 に使用されます。
Validation Management
Printable View for Test Scripts
24R1 では、Vault Validation Management は、テスト手順ファイルの添付ファイルとして保存される、関連オブジェクトレコードや実行者から収集された証拠 (スクリーンショットなど) を含むテストスクリプトの印刷可能ビューを生成する方法を提供します。印刷可能ビューがユーザのアクションから開始されると、テストスクリプト、テスト手順、不一致などの関連オブジェクトレコードに関する情報が含まれるため、ブラウザから PDF に印刷して監査人などの外部関係者と共有できます。
新しい印刷可能ビュー設定を使用すると、管理者はテストスクリプトのライフサイクル状態の設定にどの印刷可能ビューを使用できるかを定義できるようになります。XML テンプレート設定ファイルでは、管理者は、関連するオブジェクトレコードが印刷可能ビューに表示される順序や、表、1 列形式、2 列形式で表示されるフィールドを設定できます。その他の設定可能なオプションには、ヘッダーロゴ画像、ヘッダーテキスト、フッターテキスト、45 度の角度で表示される透かしテキストがあります。ヘッダーおよびフッターのテキストにトークンを使用して、レコード番号 (name__v)、印刷可能ビューを生成したユーザ名、印刷可能ビューが作成された日付/時刻などの属性を含めることができます。ヘッダーとフッターのテキストに自由テキストを追加できます。
Template Requirements
24R1 では、Vault Validation Management に要件および要件セットのテンプレート機能が導入されています。有効にすると、ユーザ要件、機能要件、設計仕様を単一のテンプレート要件レコードから作成することや、複数のテンプレート要件のセットをインポートすることができます。これにより、組織は部門やグローバル施設全体でプラクティスを調和させ、一般的に使用される要件と仕様のライブラリを確立し、新しいシステム、機器、プロセス、その他の GxP システムやプロセスが本番環境に導入されるときにインポートできるようになります。
この機能は、組織が業務でグローバルに使用できる要件の中核拠点を確立し、検証方法に関係なく、さまざまな種類のエンティティで使用できるテンプレート要件の承認済みリポジトリを確保するのに役立ちます。
Family Validation
24R1 では、Vault Validation Management でエンティティをエンティティファミリーへとグループ化する方法が追加されました。これによりファミリーの共通要件が、同一ファミリー内のすべてのエンティティ間で適用され、新しいエンティティがそのファミリーに追加された際に自動共有されるようになります。今回追加された機能強化には、エンティティをアセットオブジェクト (asset__v) のアセットに関連付ける機能や、成果物を同一ファミリー内のエンティティ間で共有できるようにする新たな Validation Deliverable Entity Version オブジェクトが含まれます。Validation Requirement オブジェクト (val_requirement_svo__v) に、複数のフィールドが追加されました。この各種フィールドは、この機能を Vault に導入する際にオブジェクトタイプ別に有効化する必要があります。
Re-order & Copy Support for Additional Prompts
24R1 では、Vault Validation Management は追加プロンプトのサポートを拡張し、ドラッグアンドドロップを使用してテストスクリプト作成 UI からプロンプトの順序を直接操作できるようにします。セットアップ手順 または実行手順がコピーされると、追加プロンプトのあるセットアップ手順および実行手順も含まれるようになりました。
Standard Action Layouts for Validation Management
24R1 では、Validation Management オブジェクトとオブジェクトタイプ用に、標準アクションレイアウトおよびレイアウト規則が追加されました。この各種アクションレイアウトには、データを標準フィールドに則った標準 Validation Management オブジェクトの各セクションおよびフィールドに整理する上でのベスト プラクティスが含まれています。管理者はこの各種テンプレートをコピーし、各自の用途に合わせてカスタマイズできます。標準アクションレイアウトはすべて、デフォルトでは無効に設定されていることに注意してください。
LIMS
Stability Study Management
この機能により、LIMS アプリケーションに次のような安定性治験を管理する機能が追加されます。
- 保管条件の詳細、時点の計画、方向の設定、仕様のテストと評価を含むスタディ デザイン。
- 承認されたスタディ デザインテンプレートから複数のスタディを開始する機能。
- 個々のスタディと検査室のリソースを最適に管理するため、1 つまたはすべての、計画されて進行中のスタディ全体にわたる今後の時点の認識。
- 安定性サンプルの分析のための既存のテスト実行機能との統合
- バッチリリーステストを使用できることで、安定性治験の時点ゼロ基準が評価されます。
LIMS Change Analysis: Merging via Multi-Record Workflow
LIMS 変更分析の機能が拡張され、マルチレコードワークフローとの互換性が追加されたとともに、必要な場合に同一ワークフロー内にある複数バージョンから単一ドラフトを作成できるようになりました。
Aggregate Testing
この機能により、再テスト時に元の臨床検査を保持することができ、新しい Aggregate Testing 機能が追加されます。これには、Sum、Average、Min、Max、Median、Mode、CountA、 StDevP、StDevS の計算関数が含まれ、多数の臨床検査から選択された結果に適用されます。これらの結果は、個別のオブジェクト、特に、Lab Sample および Spec Execution に関連付けられたすべての臨床検査から収集されます。
Modifying Test Inputs
テスト入力値が、セルから離れると即時保存され、テスト手順の入力後も、結果変更サインオフ処理と同様の手順で変更できるようになりました。
Instrument Integration Foundation
Vault Connections を使用して、サードパーティ データシステムと機器を統合するための基盤を実装します。機器を Vault 接続にマッピングし必要な統合マッピングパラメータを取得するため、新しい LIMS 統合および LIMS 統合パラメータオブジェクトが追加されました。
Bulk Sample Printing
この機能により、BarTender 接続を介して一度に複数の LAB サンプル用ラベルを一括印刷できるようになりました。これは、新しい一括レコードアクションとして実装されます。
24R1 Early Adopter Customer Feedback
仕様実行の所有者フィールド
仕様実行オブジェクトに、標準の所有者フィールドが追加されました。このフィールドは自動入力され、テンプレート入力からのドキュメント生成アクションで使用できます。
変更分析の所有者フィールド
変更分析オブジェクトに、標準の所有者フィールドが追加されました。このフィールドは自動入力され、テンプレート入力からのドキュメント生成アクションで使用できます。
仕様データマッピングの標準ライフサイクル
仕様データマッピング オブジェクトに、標準ライフサイクルが追加されました。これにより、仕様データマッピングレコードにワークフロー (レビューワークフローなど) を設定できるようになりました。
ラベル印刷の入力アクション
入力アクションが、サンプル、テストおよび消耗品の各 LIMS オブジェクトのラベル印刷で使用できるようになりました。
テスト実行の UI
テスト実行の UI にある「テストセットにテストを追加」セレクタの機能が強化され、チェックボックスでダイアログ内にあるすべてのテスト (テストセット 1 つにつき最大 100 件) を選択できるようになりました。
テスト実行の UI にある「改訂」アイコンと「以前の規格外」アイコンに、ツールのヒントが追加されました。ユーザがこの 2 つのアイコンにカーソルをかざすと、アイコンの意味について説明が表示されます。
テスト実行 UI 内の最終セルフレビュー画面に移動する前の画面で、以前は「レビューする」となっていたボタンの表記を「確認する」に変更しました。
LAB 結果変更ダイアログ
情報明確化のため、ラボ結果変更ダイアログ内にサンプル名、テスト名、および結果名の各種フィールドを追加しました。
Quality Data Model Changes
24R1 Quality データモデルの変更をご覧ください。
Regulatory
以下の Release Notes に加えて、Vault RIM Registrations、Vault RIM Submissions & Submissions Archive、および Vault RIM Publishing Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモが提供されています。
Vault Connections セクションにリストされているいくつかの機能も、Regulatory アプリケーションファミリーに影響を与えます。
すべての RIM アプリケーション
RIM Standard Action Layouts
今回のリリースで、Application、Regulatory Objective および Submission の各種オブジェクト用の標準アクションレイアウトが追加されました。標準アクションレイアウトをコピーしてお客様固有のレイアウトを作成できるため、レイアウトデザインのベスト プラクティスの開始点として利用できます。標準アクションレイアウトを有効化する際には、推奨設定リストと照らし合わせて、さらにはお客様固有の用途に基づいて標準アクションレイアウトを確認する必要があります。
RIM Registrations
Bulk Create & Manage Event Details
このリリースでは、管理者は有効期限の変更や製造施設の変更などの変更タイプを設定し、ユーザのイベント詳細の作成を簡素化できます。ユーザは、イベントレコードからイベント詳細の作成アクションを実行し、そのイベントに適用する 1 つ以上の変更タイプを選択し、それらのタイプの変更に必要な情報を入力できます。完了すると、ユーザはプレビューを生成して、提案されたイベント詳細を作成前に調整できます。さらに、ユーザはイベントレコードからイベント詳細の管理アクションを実行してプレビューを生成し、新しい詳細を追加せずにイベントの既存の詳細を調整できます。
イベントの作成は規制プロセスの最初のステップとして推奨されます。なぜなら、イベントは、規制項目および提出を通じて影響を受ける市場全体に伝達される規制データの最初のエントリポイントとして機能するからです。イベントでは、数十、場合によっては数百のイベント詳細の作成が必要になる場合があります。この機能により、これらの詳細の作成と管理が効率化されます。
Relate Bundled & Split Regulatory Objectives & Submissions to Events
このリリースでは、バンドリングウィザードまたは分割ウィザードで作成された Regulatory Objective および Submission は、任意の選択されたアクティビティのイベントに直接関連しており、エンドツーエンドの可視性を向上させます。
Wizard Support for IDMP Manufacturing Details
Vault RIM データモデルを拡張し、既存メーカーの更新と新規メーカーの追加をより適切にサポートできるようにしました。次のフィールドが追加されました:
- Authorisation Effective Date (
authorisation_effective_date__v) - Confidentiality Indicator (
confidentiality_indicator__v) - Health Authority (
health_authority__v) - Manufacturing Authorisation Number (
manufacturing_authorisation_number__v) - Manufacturing Operation Start Date (
manufacturing_operation_start_date__v) - Manufacturing Operation Stop Date (
manufacturing_operation_stop_date__v)
これらの新しいフィールドは、Event、Application、Submission、および Regulatory Objective 関連オブジェクト (例: Event Product または Application Packaging): Product、Active Substance、Inactive Ingredient、および Packaging にあります。これらは、Vault RIM の一括作成および更新アクションによって完全にサポートされています。
この拡張により、組織は正確な製造に関する詳細を把握でき、より適切な製造変更を簡単に管理できます。
Enhanced Support for Packaging Shelf Life Field
この機能により、サブミッションウィザードが有効になっている場合、Packaging 結合オブジェクトのShelf Life フィールドが Registrations のウィザードでサポートされるようになりました。このフィールドは、提出ウィザードなしでも引き続きサポートされます。
Global-to-Local Site Role Mapping in Bundling
このリリースでは、Site Role 値のグローバルからローカルへのマッピングを重視するために、Bundling アクションが拡張されました。
ユーザがActivity または Regulatory Objective からバンドリングを実施する際に、Vault はイベント関連レコードのグローバル Site Role 値を、新しい Regulatory Objective 関連レコードの適切なローカル値に変換します。
これは、次のオブジェクトに適用されます:
- Regulatory Objective Product
- Regulatory Objective Active Substance
- Regulatory Objective Inactive Ingredient
- Regulatory Objective Packaging
この改善された機能は、ビジネス管理者が Site Role 値のグローバルからローカルへのマッピングを実行した後に使用できます。
この機能により、ユーザは Site Role 値をデフォルト値からローカル値に手動更新する時間を節約できます。
この機能強化は、23R2 Release で導入された Create Related Records アクションのための Site Role 値のグローバルからローカルへのマッピングを基に構築されています。
Enhanced User Messaging When Bundling Regulatory Objectives
規制項目からバンドルする場合、ウィザード内の更新されたメッセージは、選択したアクティビティに関連付けられたイベントからの関係のみを選択できる一方、バンドルされた規制項目のすべての関係も引き継がれることをユーザに明確に示します。
Enhancement to Product-Based Activity Scope Levels
アプリケーション関係が (アプリケーション関係の有効化設定で) 有効になっており、ユーザが Event から関連レコード作成ウィザードを実行すると、アプリケーションと製品および国、アプリケーション、製品のアクティビティ範囲レベルが Application Product レコード内の製品のアカウントに更新され、影響を受ける アプリケーションごとに作成するアクティビティを決定します。この自動オン機能強化により、データの整合性が向上します。
EUDAMED XSD Version 2.0.11 Support
この機能は、XSD バージョン 2.0.11 を使用する最新の実環境の更新状況に合わせて EUDAMED UDI Submission Generation & Validation を更新します。
External ID to be Unique on UDI Attribute Set
RIM Registrations データモデル更新 (23R3.2) の一部として、UDI Attribute Set オブジェクトの External ID フィールドで Values must be unique 設定が有効になっています。
Identification of Global Site Role Values That Could Not Be Mapped
バンドルでグローバルサイトロールとローカルサイトロールをマッピングする際、イベント関連レコードからのグローバル Site Role 値が新しい Regulatory Objective 関連レコードのローカル値と一致できない場合に Vault は Controlled Vocabulary レコードからのプレースホルダー値のあるレコードを作成します。バンドルでのグローバルからローカルサイトのロールマッピング機能 (23R3.2) のこの機能強化により、レコードが確実に作成されてユーザは値を更新できます。
RIM Registrations Data Model Updates
このリリースでは、Vault RIM データ モデルを拡張し、製品とデバイスの詳細をより適切に把握できるようにしました。新機能:
- 商号用の Regulatory Text ドキュメント: 新しい Regulatory Text (
regulatory_text__v) フィールドを Labeling (labeling__c) ドキュメントのサブタイプまたはその他の規制ドキュメントに追加して、ユーザが 1 つ以上の商号をドキュメントにタグ付けできるようにしました。 - Applicable Product Type オブジェクトフィールド: Applicable Product Type (
applicable_product_type__v) フィールドを次のオブジェクトに追加して、製品データ報告に関連した出力レコードの設定を容易にしました:- Medicinal Product (
medicinal_product__rim) - Registered Packaged Medicinal Product (
registered_packaged_medicinal_product__v) - Administered Product Route of Administration (
administered_product_route_of_admin__rim) - Product Cross Reference (
product_cross_reference__v)
- Medicinal Product (
- 更新された RIM オブジェクト マッピングルール: イベントからアプリケーションへのデータを生成するための Use for Registrations フィールドは、サブミッションウィザードが非アクティブのときにユーザが Create Related Records アクションを実行する際に重複が発生する可能性を回避するために更新されました。
- Use for UDI オブジェクトフィールド: Application Shelf Life or Condition (
application_shelf_life_storage__v) オブジェクトが更新されて、Use for UDI (use_for_udi__v) フィールドが含まれるようになりました。このフィールドは、提出ウィザードで新しい提出と Regulatory Objectives を作成する際に検討されます。 - Identifier Type 選択リスト値: Master UDI-DI (
master_udidi__v) 値を Identifier Type (identifier_type__v) 選択リストに追加しました。 - Container オブジェクトタイプ: Container (
container__v) オブジェクトタイプを次のオブジェクトに追加し、組織が詳細情報を取得して、パッケージコンテナに関連する変更を管理できるようにしました:- Application Shelf Life (
application_shelf_life_storage__v) - Submission Shelf Life (
submission_shelf_life_storage__v) - Regulatory Shelf Life (
regulatory_objective_shelf_life_storage__v) - Event Shelf Life (
event_shelf_life_and_storage__v) - Registered Shelf Life (
registered_shelf_life_and_storage__v)
- Application Shelf Life (
- SPOR Term のオブジェクトフィールド長: Controlled Vocabulary (
controlled_vocabulary__rim) オブジェクトのSPOR Term (spor_term__v) フィールドの長さは、名前が非常に長い SPOR 値に対応するために 1500 文字に増加しました。これにより、IDMP の FHIR メッセージに完全な用語名を含めることが可能になりました。
RIM Registrations、RIM Submissions
Global Content Plan Dispatch by Country
この機能により、グローバルコンテンツプランを個々の国に送信できるようになります。ディスパッチダイアログは、国、ドキュメントセット (提出のグループ化) のいずれかの一部またはすべてをサポートします。ディスパッチダイアログで国が選択されている場合、ディスパッチはそれらの国の Activity レコードに基づいて行われます。
Generate Active Dossier from Global Content Plan Dispatch
この機能は、グローバルコンテンツプランのディスパッチ時に、ディスパッチされたドキュメントに基づいて Active Dossier レコードの自動生成をサポートします。対象となるアクティビティの Local Disposition によって、Active Dossier レコードが Dispatched として生成されるか Pending Current として生成されるかが決定されます。ディスパッチ時に生成することで、通常の提出および承認サイクル以外の規制上のユースケース (提出前または提出なしで実装できる変更など) に対して、より正確な Active Dossier が可能になります。さらに、この機能により、ディスパッチされて、同時変更が発生したときから次のドキュメントバージョンを構築するために内部で使用できる状態になっているドキュメントを早期に可視化できます。
Support Reattachment of a Global Content Plan to its Event Record
Event レコードから削除されるグローバルコンテンツプラン (GCP) を、再追加できるようになりました。以前は、Event レコードの Global Content Plan フィールドが、誤って/意図的にクリアされると、ユーザは Event レコードに GCP を再添付することができませんでした。今後は、フィールドがクリアされた場合でも、適切な GCP を Event フィールドで選択して、それを元の Event レコードに再度関連付けることができます。
Global Content Plan Synchronization Updates
この機能は、グローバルコンテンツプランを同期するための拡張機能で構成されています。
- 比較ビューアのヘルプが更新され、比較ビューア内で承認または拒否せずに移動したときにユーザの選択が保持されるようになりました。
- 対象の送信コンテンツプランセクションをマッチングするときに、不足している権限が検証されるようになりました。
- 対象の送信コンテンツプランでは、ディスパッチから作成された、新しく追加されたコンテンツプランとコンテンツプランアイテムのレコードは、同期後にテンプレート内の順序に基づいて並べられます。
- 比較ビューアは、ユーザの列幅の設定を保持します。
Global Content Plan Dispatch Update: Notify Submission Owners Upon Initial Submission Content Plan Creation
新しい通知 SCP Created from Dispatch は、新しいサブミッションコンテンツプランがグローバルコンテンツプランのディスパッチから最初に作成されたときに、提出者にアラートが送信されます。これは、比較が完了し、レビューの準備が整ったときに所有者に通知される比較準備完了通知に似ています。この新しい通知は、エンドユーザーに利便性を提供し、提出者がシステムを監視したり、注意が必要な新しく作成された提出コンテンツプラン用のメッセージを送信したりする必要がなくなります。
RIM Registrations、RIM Submissions、RIM Submissions Archive
Active Dossier Hovercard Update and Viewer Support for Dispatched Records
Dispatched Active Dossier 状態の選択リスト値は、23R2 で実装されました。このリリースでは、Active Dossier Viewer が更新され、Dispatched アイコン () が Dispatched 状態で Active Dossier Item Detail レコード用に表示されます。Active Dossier のホバーカードも Event と Activity フィールドを表示するために再フォーマットされます。
RIM Submissions、RIM Submissions Archive
Active Dossier Consolidated Sections (Enablement Update)
以前のリリース (23R3) では、Active Dossier テンプレートレコードから特定の製品関連トークンを削除するために、Active Dossier の統合セクション機能が導入されました。製品バリエーションと製造元 (3.2.A.1 では製品、製品バリエーション、有効成分) 用のこれらのトークンは、以前は繰り返し兄弟レコードセクションの作成を担当していました。24R1 より前は、既存の Active Dossier レコードのないお客様の Vault のみが自動的に更新され、有効な Active Dossier レコードを持つお客様には、管理者設定でレコードの移行を有効にするオプションが与えられていました。
24R1 では、すべての Vault が新しい Active Dossier テンプレートに移行され、管理者設定が削除されます。これらのアクションは、Vault に Active Dossier レコードがすでに含まれているかどうかに関係なく、自動的に実行されます。レコードの更新が完了すると、新規および既存の Active Dossier の要約された Active Dossier ビューアレイアウトがエンドユーザに表示されます。
Active Dossier Update to Support Common Countries
国がアクティブなドシエでサポートされるように、国コードを明確に定義する必要があります。コンテンツプランの目的で使用される共通 (EU) および 共通 (GCC) 国の 国コード は、それぞれ common_eu および common_gcc に明示的に設定されるようになりました。
これまで、共通 (GCC) は共通の国コードで識別され、共通 (EU) は空白の国コードで識別されていました。管理者は、country_code__rim フィールドを、これらの国で Active Dossier を適切にサポートするために、共通国レコードで更新する必要があります。
Cloning Enabled for RIM Views
コンテンツプラン、Active Dossier、または Submissions Archive の保存済みビューは、システム管理の RIM View (rim_view__v) オブジェクトによって管理されます。管理者の効率をサポートするために、RIM Vault のコピー作成時に RIM ビューオブジェクトもコピー作成されるように設定されます。
保存済みビューは、展開時に親 Vault から継承されます。共有設定やユーザ/グループは引き継がれません。Vault 所有者は、必要に応じてコピー作成されたビューにアクセスして共有できます。保存済みビューでコピー作成されていないオブジェクトレコードのフィルタは保持されません。
RIM Submissions
Expand Content Plan to Matched Document Level
コンテンツプランは、コンテンツプランアイテムで停止するのではなく、一致するドキュメントレベル (最大 2000 ドキュメント) まで拡張されるようになりました。この機能強化により、コンテンツプランレベルで Expand ユーザアクションを実行するときに、各コンテンツプランアイテムを個別に展開して一致するドキュメントを表示する時間と手作業が軽減されます。
Suggested Matching Enhancements: Reorder, Split, Update Expected Counts, & Bulk Match
サブミッションコンテンツプランで作業する場合、ユーザはドキュメント一致モード内から次のようなコンテンツプランアイテムのアクションを実行できるようになりました。
- コンテンツプランアイテムでの一致したドキュメントの順序変更
- コンテンツプラン項目の分割
- コンテンツプランアイテムの予想される固定状態数の更新
- すべて選択またはすべての選択を解除の使用によるドキュメントの一括一致
これらの機能強化により、コンテンツプランビューア内でドキュメントを管理し、手動で一致させるためのスムーズで効率的なフローがサポートされます。ユーザは、サブミッションコンテンツプランのドキュメントを手動で管理するために必要なすべての手順を 1 つの便利なインターフェース内で実行できるようになりました。
Improve Match Document Mode Performance
一致候補表示時に時々見られるパフォーマンス低下に対して永久的な解決策を提供するため、ドキュメント一致モードでも Vault ライブラリの仕様と同様に、おおよそのドキュメント数とページ番号が表示されるようになりました。ドキュメント選択で 25 件を超える一致可能性のあるドキュメントがヒットした場合、Vault では正確な合計数を計算する代わりに、「多数ヒットのうち 1 ~ 25 件」と表示されます。ヒット数をおおまかに算出することで、一致可能性のあるドキュメントの読み込みにかかる時間が大幅に短縮される一方で、ユーザは引き続き結果ページ間を移動できます。引き続き追加でフィルタリングを使用することにより、膨大な一致候補数を減らすこともできます。
Vault-Wide PDF Standardization
管理者は、デフォルトのレンディションプロファイルで PDF 標準化を Vault 全体の設定として行えるようになりました。24R1 より前は、この設定はカスタムレンディションプロファイルでのみ利用可能でした。
これをデフォルトのレンディションプロファイルに拡張することで、カスタムレンディションプロファイルを利用するための追加設定が必要なく、標準化をより包括的に適用できるようになります。
有効にすると、新しいバージョンの作成時にすべての新しいドキュメントが標準化されます。既存のドキュメントは、Vault Loader を使用して再レンダリングできます。
Improved Inactive Bookmark Handling During PDF Standardization
ソースファイル内の非アクティブなブックマークは、階層内の最初の子アクションブックマークに基づいて機能します。一部のお客様は、非アクティブなブックマークを利用してソースファイル内にブックマークのグループを作成します。
24R1 より前は、Vault は非アクティブなブックマークを 1 レベル上に調整し、グループ化が削除されていました。非アクティブなブックマークの動作を最初の子ブックマークに一致させると、ドキュメントビューアだけでなく PDF ソースファイルを操作する際にもユーザの一貫性が保証されます。
非アクティブなブックマークにアクティブな子ブックマークがない場合、それらのブックマークは標準化されたソース PDF およびドキュメントビューアでも削除されます。
Content Plan Creation to Respect DTD/XSD Version
この機能により、コンテンツプラン関連オブジェクトで 2 つの新しいフィールド、XML ICH DTD / XSD Version および XML Regional DTD / XSD Version が、Submission の XML Version フィールドに沿ったサブミッションコンテンツプラン作成プロセスへの更新と共に導入されます。これにより、RIM のお客様は、複数の異なる XML バージョンに適用されるコンテンツプランテンプレートのための、より関連性の高いコンテンツプランを生成できるようになりました。
この機能により、インデックスや STF などの eCTD 限定のコンテンツプランアイテムテンプレートで、XML ICH DTD / XSD バージョンの設定オプションを有効にし、これらのコンテンツプランアイテムが非 eCTD 用に作成されないようにできるようになりました。
RIM Submissions Archive
Sharing Submission Selection in the Viewer
提出アーカイブビューアのユーザーは、アプリケーション内の 1 つ以上の特定の提出用 URL を共有できるようになりました。以前は、適用される提出フィルターに関係なく、URL にはアプリケーション ID のみが含まれていたため、共有はアプリケーションビューに制限されていました。これで、ビューアの URL にはアプリケーション ID と提出 ID が含まれるようになりました。
ユーザが Submission 列で必要な提出物をフィルタ処理する際に、サブミッション ID が自動的に URL ストリングに追加されます。フィルタ処理された提出物がビューから削除されると、その提出 ID は URL から自動的に削除されます。ユーザーは、他のユーザーが開いたり、Web ブラウザに貼り付けたりできるように、URL をコピーして貼り付けることができます。URL 全体の文字数が 2083 文字を超えないようにしてください。これを超えると、ユーザーに警告が表示され、最大数を超えて追加された提出 ID は URL の文字列から削除されます。
埋め込みドキュメントビューア: モードの選択
24R1 Platform 機能 Doc Viewer: ユーザのデフォルトの改善を使用すると、Vault はすべてのドキュメント (Submissions Archive ビューア内のドキュメントを含む) を開きます。選択モードが自動的に有効になり、グラブモードは自動的に無効になります。
Submissions Archive では、この変更により、ビューアでビデオをレンダリングできるようになり、ユーザがビューアからテキストをコピーできるようになります。これにより、Submissions Archive ユーザがドキュメント、特にハイパーリンクのあるドキュメント内を移動する方法も変わります。選択モードがデフォルトで有効になることで、ビューアでハイパーリンクをクリックしたいユーザは、リンク上にマウスを移動し、表示されるポップアップを選択してリンクを開くことができます。ユーザはグラブモードを有効にすることもできます。それによって、ドキュメント内のリンクを直接クリックできるようになります。ヒント: PC ではキーボードショートカット「CTRL + Shift」、Mac では「Command + Shift」を使用してモードを切り替えることができます。
グラブモードを有効にしてハイパーリンクを直接クリックします:
リンクの上にマウスを移動してクリックすると、ハイパーリンクが選択モード (新しいデフォルト) で開きます:
Update Viewer Document Header
このリリースでは、Submissions Archiveビューアのドキュメントビューアヘッダーが更新され、ドキュメントのバージョンとドキュメントのライフサイクル状態が表示されます。
Leaf Source File Path Display in the Submissions Archive Viewer
この機能を使用すると、RIM Submissions Archive ユーザは、ビューアグリッドでリーフソースファイルパスフィールドのメタデータを表示およびフィルタリングできるようになります。
RIM Publishing
Document Type Definition for RLCP Published Output
この機能を使用すると、ユーザはContent Plan Item、Content Plan Item Template、Report Level Content Plan レコードでパブリッシュされた Report Level Content Plan (RLCP) コンテンツのドキュメントタイプを定義できます。それにより、RLCP のパブリッシュ後のサブミッションコンテンツプラン内での分類と自動一致が可能になります。
Section Level Merge for RLCP
この機能を使用すると、ユーザは、Content Plan セクション内の Content Plan Items のすべてのドキュメントを、公開時に 1 つの PDF に結合することができます。
この機能は現在、RLCP パブリッシュ済み出力のドキュメントタイプの定義をサポートしていません。セクションマージでマージされたセクションはすべて、サブミッション準備完了ドキュメントタイプとして出力されます。
Link Evaluator Enhancements
リンク評価機能に新たな機能が追加され、ユーザがリンク評価機能のインターフェイスから直接実行できるタスクの数が増えました。対応している各タスクでは、各ソースドキュメントに戻ることなく単一画面で完結できるため、ハイパーリンクを作成および確認する際の効率と生産性が向上します。改善点には以下が含まれます。
- リンク評価機能のインターフェース: ページの操作性が改善されました。また、リンク評価機能ページからサブミッションコンテンツプランに戻る操作や、サブミッションアーカイブにアクセスする操作がより簡単になりました。
- リンクの削除: 新しい Suppress Links アクションを使用すると、ユーザは有効なハイパーリンクまたは無効なハイパーリンクを、個別もしくは一括で削除できます。削除対象としてマークしたハイパーリンクは、Vault Submissions Publishing で抑制され、ブリッシュ済み出力上で通常の青色フォントから黒色へと変更されます。また、抑制済みリンクや再ターゲット済みリンクをフィルタリングできるようになりました。
- リンクの再ターゲット: Retarget アクションを使用する場合、ユーザはオプションで、ターゲットドキュメントに対して特定のレンディション (インポートされたレンディションなど) を選択できます。
- リンクの上書き: ユーザは、Retarget または Suppress リンクアクションを、新規リンクアクションによって個別または一括で上書きすることにより、元の状態に戻すことができます。上書き (抑制または再ターゲット) を適用する場合、ユーザはオプションで、その上書き処理をサブミッションレベルまたはアプリケーションレベルで適用するかを選択できます。サブミッションレベルで上書きを適用すると、現在のサブミッション内のパブリッシュ済みリンクが変更され、アプリケーションレベルで上書きを適用すると、アプリケーション内の (現在のサブミッションを含む) すべてのパブリッシュ有効サブミッションにあるパブリッシュ済みリンクが変更されます。この対象には、同じ一致するドキュメントおよび同じリンクを持つ今後のサブミッションも含まれます。
Jordan Multiple Application Number Support
JO でパブリッシュしているユーザは、Submission レコードから Update Administrative Information アクションを介して、eJDWS アプリケーション番号 (アプリケーション番号) とアプリケーション番号 (番号) の上書きを追加または削除できるようになりました。
Disable Auto Reference Leaf for Non-eCTD Submissions
Vault Publishing では、非 eCTD 提出で同じドキュメントが複数回一致した場合に、参照リーフが自動的に作成されなくなります。Vault Publishing では、非 eCTD 提出 (ICH/DTD が 3.0 または 3.2 ではない場合) に対して参照リーフ機能が無効になり、同じドキュメントを複数回発行できるようになります。
同じドキュメントが、提出コンテンツプラン内の複数のコンテンツプランアイテムに一致する場合、現在の発行動作では、eCTD ガイドラインに従って、単一ドキュメントの発行および提出の重複を防ぐために参照リーフが自動的に作成されます。XML バックボーンが欠如している非 eCTD 提出では、同じドキュメントが複数回一致した場合も、予想されるセクション内で発行および提出する必要があります。
RIM Publishing、RIM Submissions Archive
Increase RIM Vault Max File Size to 500GB
非常に大きなファイルを発行および提出するお客様をより適切にサポートするために、すべての RIM Vault で最大ファイルサイズ制限を 500 GB に増加しました。
Single Submission Package Potential Incomplete Export Notification
この機能により、提出書類が継続的発行用に設定されていて、エクスポートされたドシエパッケージが不完全な場合に、Submissions Archive のエクスポートメッセージが更新され、わかりやすくなりました。
RIM データモデルの変更
24R1 RIM データモデルの変更をご覧ください。
Safety
すべての Platform 機能を含む Safety 24R1 Release は、2024 年 4 月 18 日および 2024 年 4 月 26 日の暫定公開を予定しています。
注: 2024 年 4 月の 24R1 以降のすべてのリリースで、Vault Safety General Release のヘルプコンテンツは新しいサイトに移動します。新しいサイトは、Limited Release コンテンツからテストいただけます
Vault Connections セクションに記載されているいくつかの機能も、Safety アプリケーションファミリーに影響を与えます。
Safety
以下のリリースノートに加えて、Vault Safety および Vault SafetyDocs Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモを提供しています。
Sender-Based Inbound Validations
Vault Safety を使用すると、管理者は個々の送信プロファイル (ビジネスパートナー) に特定の検証基準グループ (ICH E2B(R3) など) を割り当てることができます。さらに、E2B 送信が AS2 ゲートウェイまたは Vault API を介してインボックスアイテムにインポートされると、Safety システムでは、インポートされたデータがトランスミッションプロファイルのルールに照らして検証されます。その後、症例が承認されたか却下されたかを示す確認応答が E2B 形式で自動的に生成されます。また、この検証の結果は、確認応答およびインボックスアイテムの検証結果セクションに追加されます。この更新以前には、Vault Safety では ICH E2B 検証ルールの固定コレクションを採用していました。この機能により、E2B 形式で受信したデータ、特にパートナーから送信されたデータの精度が大幅に向上します。その結果、Safety のお客様は、症例処理の包括的なプロセスを広範囲に自動化できます。
詳細
- 有効化: 送信者ベースの受信検証を有効にする
- 管理者ヘルプ: トランスミッションプロファイルの管理: 検証基準の追加
- ユーザヘルプ: E2B 送信を受信する
Safety Inbox Loader: Multi-CSV ZIP Data Import for VAERS, KAERS, and Custom Formats
23R3 では、インボックスアイテムへの複数症例 CSV インポートのサポートが Vault Safety に追加されました。今回のリリースにより、Vault Safety Inbox Loader では、複数 CSV ZIP ファイルからインボックスアイテムへの一括インポートがサポートされるようになりました。Vault Safety では、圧縮されたすべての CSV ファイルからデータが自動的に抽出されて集積され、症例ごとにインボックスアイテムが作成されます。この機能を使用すると、管理者はエンコーディングや国などの形式メタデータを指定できます。この機能強化により、複数 CSV ZIP ファイルから配布された症例の症例処理効率とデータ品質が向上します (VAERS 症例用の米国の FDA、韓国の KAERS など)。
詳細
- 有効化: Safety インボックスローダによる表形式の複数症例データのインポートを有効化
- 管理者ヘルプ: Safety インボックスローダによる表形式の複数症例データのインポートを管理
- ユーザヘルプ: インボックス項目のインポート: 複数症例の表形式データのインポートについて
Case Parent Information Support for JSON Intake
今回のリリースにより、Vault Safety では JSON 取り込み用の症例親情報のインポートがサポートされるようになりました。
詳細
Two-Way Relationships Between Multi-Case and Single-Case Files
Vault Safety では、複数症例ファイル (XML、CSV、ZIP) とその個々の単一症例ドキュメントの間の双方向関係が円滑になりました。この機能強化により、インボックスアイテムとそのソースドキュメントの間のシームレスなリンクが可能になります。以前は、この関係は容易には実現できず、インボックスアイテムをソースドキュメントに関連付けることは困難でした。
詳細
- ユーザヘルプ: インボックス項目のインポート
Product Ranking Suggestions for Imported Inbox Items
今回のリリースにより、Vault Safety では、あらゆるソース (E2B、JSON、Inbox Loader、Connections) からの Product Ranking Suggestions for Imported Inbox Items が強化されています。治験製品の一致がある場合、Vault Safety では、疑わしい薬剤ロールを持つ最初の一致にランク 1 が割り当てられます。または、治験製品プレースホルダーの一致がある (盲検名が存在する) 場合、Vault Safety では、疑わしい薬剤ロールを持つ最初の一致にランク 1 が割り当てられます。最後に、治験製品または治験製品プレースホルダーの一致がない場合、システムでは、疑わしい薬剤ロールを持つ最初に一致した自社製品に対してランク 1 の割り当てが試行されます。ランク 1 がソースデータに存在する場合は、製品のソースデータパネルにも表示されます。
詳細
Prevent New Info Date Update During Merge to Current
インボックスアイテムをインフライト症例に移行するときに、システムでは症例の最新情報入手日付が更新されなくなりました。この変更では、準拠した送信期限日の生成がサポートされています。
詳細
- ユーザヘルプ: 受信箱項目をインフライト症例にマージする
Case Compare: Informative Message for Custom Objects with Duplicate Names
インボックスアイテムをインフライト症例にマージする際、または Follow-up Case に変更する際に、一意の名前が必須であるにもかかわらずカスタムオブジェクトの名前が重複している場合は、症例比較ページに明確な情報メッセージが表示されるようになりました。このメッセージには、マージまたはコピーできないレコードが一覧表示されます。症例処理担当者は、処理をキャンセルしてレコードを修正するか、あるいはカスタムレコードをアクションから除外して続行するかを選択できます。これまでは、この設定ではシステムエラーが生じ、管理者が一意の値を必須として設定した各カスタムレコードについて情報が表示されませんでした。
詳細
Document References on Domestic and Localized Follow-Up Cases
国内症例とローカライズ済み症例のフォローアップを作成する場合、Vault Safety では、以前の症例バージョンからドキュメントリンクがコピーされるようになりました。これには、症例評価予測可能およびローカライズ済み症例評価レコードのデータシートドキュメントフィールド内、およびローカライズ済み症例ドキュメントレコードのドキュメントフィールド内のリンクの設定が含まれます。
Non Re-Transmittable Document Classification
Vault Safety では、E2B 送信に含めてはいけない症例ドキュメントに対して、新しいドキュメント分類である Case > Source > Non Re-Transmittable Document が導入されています。Initial Case および Follow-up Case の作成時に、新しく作成された症例バージョンが、Case > Source > Non Re-Transmittable Document classification を持つすべてのドキュメントの Case フィールドに追加されます。
Case Access Group Security: Inbox Items by Country
このアップデートにより、症例アクセスグループセキュリティを使用して、インボックスアイテムの閲覧者を国別に制限できるようになりました。User Access Group Assignment レコードで国が指定されている場合、そのグループ内の各ユーザは指定された国のインボックスアイテムのみを閲覧できます。これまでは、ユーザは報告者の国に関係なく、すべてのインボックスアイテムを閲覧できる状態でした。この機能強化により、Inbox Item ビューのコントロールが Case ビューと一致するようになりました。
詳細
Case Access Group Security: Support for Localization on Inbound Transmissions
Vault Safety では、関連付けられている症例またはインボックスアイテムのローカライゼーションに基づいて、受信伝送での症例アクセスグループセキュリティをサポートするようになりました。これにより、既存のセキュリティ制御が拡張され、ユーザの受信伝送アクセスが確実にローカライゼーション別に許可されるようになりました。
詳細
- 管理者ヘルプ: 症例アクセスグループセキュリティの管理: ユーザの伝送アクセス
QC Checklist Generation
今回のリリースにより、Vault Safety では Vault チェックリストを利用できるようになりました。このリストは、プロセス中および終了後の品質管理 (QC) アクティビティのために自動的に生成できます。Vault Safety では、これらのチェックリストは通常、QC ガイダンスと、症例に対して (通常は QC プロセス中に) 実行する必要がある特定のフィールド検証を提供するために使用されます。管理者は、さまざまな使用例 (自発症例、臨床症例、製品固有の症例など) に合わせて特定のチェックリストデザインを設定できます。症例の属性 (レポートタイプ、製品、重症度、データ入力ユーザ、ソースなど) に基づくチェックリストルールに基づいて、適切なチェックリストデザインを自動的に選択できます。このルールでは、サンプリングレート (すべての症例、日付範囲に基づいてランダムに選択された症例など) に従ってチェックリストを生成する頻度を制御することもできます。
品質管理は、症例処理時間に大きく影響します。QC チェックリストの自動生成により、QC アクティビティが最適化され、オープン症例と終了済み症例の処理の品質と効率が向上します。
詳細
- 有効化: QC チェックリストの生成を有効にする
- 管理者ヘルプ: QC チェックリスト生成ルールの設定
- ユーザヘルプ: QC チェックリストの生成
Clinical Trials: Isolate Blinded Product Information
この機能は、Vault Safety で臨床試験症例におけるすべての盲検化製品情報を完全隔離するためのの基盤となり、盲検解除された症例製品の独立した評価をサポートするものです。このアプローチにより、SUSAR を倫理委員会や治験施設に配布する際の臨床試験のデータ整合性が促進されます。さらに、盲検解除済み製品情報を隔離することで、お客様が下流工程でレポート作成ハブを用いて正確なレポート作成を円滑に行えるようになります。
注: この機能は、現在テスト用途でのみご利用いただけます。詳細については、Veeva 担当者にお問い合わせください。
詳細
- 有効化: 臨床試験の有効化: 盲検化製品情報の隔離
- ユーザヘルプ: 盲検化臨床試験情報の隔離
Combination Product Updates Applied to Case Names
症例組み合わせ製品の更新が、Vault Safety で生成された症例名に反映されるようになりました。以前は、組み合わせ製品の更新は症例名に影響を与えませんでした。より正確でわかりやすい症例名により、症例の迅速な参照と特定が可能になります。
詳細
- 管理者ヘルプ: デフォルトの症例オブジェクト命名規則について
Inactivate Product & Study Registrations
今回のリリースにより、Vault Safety では Product and Study Registrations を Deleted 状態タイプに設定できるため、Safety Rule Engine では症例の報告価値を判断する際にそれらが考慮されなくなりました。以前は、登録が無効なライフサイクル状態に移行していたため、フォローアップ状況で問題が発生することがありました。この新しい製品および治験登録機能により、ダウンストリームの ICSR レポートがより正確になります。
詳細
- 有効化: 製品および治験登録の無効化を有効にする
- 管理者ヘルプ:
Datasheet Expectedness by Age and Sex
Vault Safety の予測可能評価では、年齢層と性別の基準がサポートされるようになりました。この機能により、特定の統計情報に影響を与える有害事象について、より正確なデータシートを作成できます。これにより、自動計算された予測可能値をオーバーライドする必要がなくなり、症例処理担当者の時間が節約され、精度が向上します。
詳細
- 有効化: 年齢と性別によるデータシートの予測可能を有効にする
- 管理者ヘルプ:
Manage Custom Dose Forms, Routes of Administration, and Units of Measurement
このリリースで、管理者は、Vault Safety 内でカスタム用量フォーム、投与経路、測定単位値を管理できるようになりました。システムが提供する値への柔軟な拡張により、管理者は必要に応じてカスタム値を追加、編集、削除できます。MedDRA、WHODrug、および IMDRF 辞書が管理されているのと同じビジネス管理領域で利用できるCoding Dictionary Loader を使用すると、カスタム値の更新プロセスが簡単で効率的になります。カスタム値がロードされると、インテーク、症例処理、EDQM マッピングにすぐに使用できるようになります。
詳細
- 有効化: カスタム用量フォーム、投与経路および測定単位の管理を有効にする
- 管理者ヘルプ: カスタム用量フォーム、投与経路および測定単位の管理
MedDRA Browser and Search Improvements
今回のリリースにより、Vault Safety では MedDRA 検索が強化され、より良い結果が返されるようになりました。単一語または複数語の用語を検索すると、検索用語に完全に一致するか、または検索用語を含む下層語 (LLT) が結果に表示されます。さらに、MedDRA ブラウザによって返される検索用語の結果の制限が 5000 件に増加しました。これらの改善により、MedDRA ブラウザで検索するユーザが正しい MedDRA 用語をコーディングできる可能性が高まります。
詳細
- 管理者ヘルプ:
Standard MedDRA Query (SMQ) Hierarchy Update
標準 MedDRA クエリ (SMQ) を Vault Safety にインポートすると、システムでは基本語 (PT) レベルで MedDRA 用語への参照が作成されるようになりました。以前は、下層語 (LLT) レコードへの参照が作成されていました。この更新では、SMQ を使用して報告価値を判断する移動先への考えられる過少報告に対処します。
詳細
- 管理者ヘルプ: 標準 MedDRA クエリのインポート
Medical History Label Update
今回のリリースにより、Vault Safety では、廃止予定の病歴テキストフィールドのラベルが「病歴*」に更新されます。この変更は、管理者が Vault に現在の病歴テキストフィールドが設定されていることを確認するのに役立ちます。最新の病歴テキストフィールドには、E2B 送信に有効な空欄の理由値の選択リストが含まれます。症例ページレイアウトで廃止予定の病歴* フィールドが使用されている場合は、それを削除してから、準拠した病歴テキストフィールドを追加します。
詳細
- 管理者ヘルプ: 病歴テキストフィールドの設定
Create and Export Case and Safety Report Collections
症例のグループのレビューとレポートをサポートするために、Vault Safety では症例コレクションと、複数の E2B および CIOMS I フォームの一括エクスポートが導入されています。エクスポートの際、フォームはバインダーに整理されるため、安全性レポートを簡単にダウンロードして、パートナーや関連する保健当局と共有することができます。さらに、Vault Safety では、すべての終了済み症例の CIOMS I フォームを毎日自動的に生成できます。以前は、症例のグループを確認して複数のフォームを一度に生成するには、手動プロセスが必要でした。この機能により、フォーム生成プロセスが合理化され、症例管理に対するより一貫したアプローチが提供されるため、効率が大幅に向上します。
詳細
- 有効化: 症例および安全性レポートコレクションの作成およびエクスポートを有効にする
- 管理者ヘルプ: 症例終了ジョブで症例フォームの自動生成を設定する
- ユーザヘルプ: 症例および安全性レポートのコレクションを管理する
Transmission Profiles for FDA E2B(R3) Submissions and Distributions
FDA E2B(R3) 形式での FDA への送信をサポートするために、Vault Safety では 3 つの新しいトランスミッションプロファイルが導入されています。これらは CDER 治験、CBER 治験、および CDER IND 免除です。この機能は、Vault Safety の今後の FDA E2B(R3) 機能を拡張する上での構成要素です。
Update to Seriousness Picklist
デバイスおよびコンビネーション製品のケースをサポートするために、管理者は重大度選択リストを更新して、「恒久的な障害/損傷 (デバイス) を防ぐために必要な介入」を含めることができるようになりました。これまで、Vault Safety にこの値はありませんでした。この機能には、[重大度] 選択リスト値のみが含まれます。今後の機能強化には、レポート生成のための値のマッピングが含まれます。
詳細
- 有効化: 重症度選択リストの更新の有効化
- 管理者ヘルプ: 外部症例の移行
Support Device Seriousness on MedWatch 3500A, CIOMS I, and E2B
Vault Safety では症例フォーム生成時に、重大度選択リストの「恒久的な障害/損傷 (デバイス) を防ぐために必要な介入」の値を、以下のレポートとセクションにマッピングするようになりました。
- MedWatch 3500A: B.2 有害事象を原因とする結果
- CIOMS I:
- I. 7 + 13 反応の説明
- I. 8-12 すべて確認
- E2B:
- E2B(R3): E.i.3.2f その他の医学的に重要な状態
- E2B(R2):
- A.1.5.1 重症
- A.1.5.2 重症度基準
詳細
Reporting Rules Configuration for Non-Vault Owners
必要な権限を持つ Vault Safety ユーザーは、カスタム報告規則を編集できるようになりました。以前は、Vault 所有者として指定されたユーザーのみが報告規則を変更できました。この向上により、管理者によるカスタム報告規則の管理で柔軟性が増しました。
詳細
- ユーザヘルプ: レポート作成ルールセット: カスタム安全性ルールセットの作成
Safety Rule Engine Troubleshooting Tool
Vault Safety の管理者は、Safety Rules トラブルシューティングツールを使用して、提出規則と配布規則に関する問題を診断できるようになりました。管理者は、このツールを使用して、特定のケース ID に対して Vault Safety Rule Engine を実行できます。その後、どの規則がパスし、どの規則がパスしなかったかなど、そのケースに対する規則エンジン実行の詳細なログを取得できます。以前、Vault Safety は各提出と配布が生成された理由を説明する送信メッセージを作成していました。ただし、規則をパスしなかった場合、システムは情報を記録しませんでした。この透明性の向上により、Vault Safety の管理者は、追加の設定検証、トラブルシューティング、Vault Safety の報告規則の検証を実行することができます。
詳細
- 管理者ヘルプ: 安全性ルールのトラブルシューティング
Rule Engine Update for Safety Measure and Research Reports
このリリースでは、PMDA 安全対策および研究報告書の安全性報告規則が大幅に強化されています。報告規則エンジンは、評価規則で指定されていない限り、特別報告分類の症例を除外するようになり、PMDA 以外の機関への過剰報告の問題に効果的に対処します。以前は、報告対象外の症例に対しても提出物が生成され、それを抑制するために手動による介入が必要でした。この更新により、報告プロセスが合理化され、手作業が削減され、より正確で効率的な提出が可能になり、時間とリソースを節約しながらコンプライアンスが強化されるようになりました。
詳細
- ユーザヘルプ: レポート作成ルールパラメータの参照: 特別レポート分類
Japan: Substance Matching for CPR Generation (Cross Report)
Vault Safety は、過大報告と過少報告の問題を排除するために、日本の PMDA 提出のための症例製品登録 (CPR) 生成のサポートを拡張しました。この機能強化により、治験薬登録のための物質の完全一致を優先するために、CPR 生成ロジックが改訂されますが、完全一致が利用できない場合は市販後の製品登録で部分一致が使用されます。また、複合製品 (複数の物質を含むケース) の CPR は、ケース製品がこれらの複数の物質と正確に一致した場合にのみ生成されます。この更新により、PMDA 提出物の過大報告や過少報告を排除することで、症例処理の効率が大幅に向上し、コンプライアンスが確保され、CPR の適切な生成とより正確な報告プラクティスが実現します。
この機能は、PMDA ローカル症例処理の自動化が有効になっている Vault で自動的にオンになります。
詳細
Japan: One Last Time Reporting
「最後にもう一度レポート作成」の要件に準拠するために、Vault Safety では、日本の PMDA 提出に対する報告価値の変更の評価がサポートされています。
この機能は、日本の症例製品登録ベースの期日計算を使用しているお客様に対して自動的にオンになります。この機能では、Vault で日本の「最後にもう一度レポート作成」を設定するための追加オプションが管理者に提供されます。
詳細
- 有効化: 日本の最後にもう一度レポートを有効にする
- ユーザヘルプ: 日本 (PMDA) 版の最後にもう一度レポート作成
Japan Periodic Reporting: Post-Market Non-Serious (NUPR)
今回のリリースにより、Vault Safety では、市販後非重篤未記載定期レポート (NUPR) による初の医薬品医療機器総合機構 (PMDA) 集積レポート機能が導入されています。
詳細
- 有効化: 日本の定期レポート作成: 市販後非重篤 (NUPR) を有効にする
- ユーザヘルプ: PMDA 市販後集積レポートの作成
DSUR and PBRER: Cumulative Tabulations for all IMP, Comparator, Placebo and Blinded Case Study Products
今回のリリースにより、Vault Safety では、「DSUR and PBRER 治験薬の因果関係」機能 (23R2 でリリース) が強化されています。これは、主要症例治験製品のみに焦点を当てるのではなく、重篤な副作用 (SAR) において症例に関連するすべての治験薬、プラセボ、実対照薬、および盲検化治験製品を含むように範囲を拡張することで実現されています。さらに、重篤な有害事象 (SAE) 表における症例数の適切な列を決定する際には、主要症例治験製品に限定されることなく、症例に関連するすべての治験薬、プラセボ、実対照薬、および盲検化治験製品が考慮されます。
この機能は、DSUR and PBRER 治験薬の因果関係が有効になっている Vault で自動的にオンになります。
詳細
- ユーザヘルプ:
Safety Data Validation Exception Handling
Vault Safety の管理者が安全性データ検証の評価中に発生したエラーを診断できるように、Vault Safety では、個別の症例と提出および個別の検証基準に関する検証エラーログが提供されるようになりました。これにより、サポート要請を提出することなく、既存の検証概要結果レコードとともに、カスタム安全性データ検証の設定、トラブルシューティング、および管理に必要な情報が管理者に提供されます。
詳細
- 有効化: 安全性データ検証の例外処理を有効にする
- 管理者ヘルプ: 検証基準のトラブルシューティング
Validation Query Builder: Inbound Object Reference Expressions
Vault 式エンジンを使用するカスタム検証基準では、インバウンドオブジェクト参照を使用して、同じチェック内の複数のオブジェクトレコードを検証できるようになりました。また、式エンジンでは症例ドキュメントを検証できます。以前は、この機能では単一オブジェクトレコードのデータの検証のみがサポートされていました。この機能強化により、Veeva プロフェッショナルサービスとお客様は、Veeva テクニカルサービスの支援なしに安全性データ検証を管理できます。
詳細
- 管理者ヘルプ: カスタム検証基準ルール数式の作成
Supporting AS2 Connection Adoption
このリリースにより Safety Gateway には、AS2 NMPA CDE 接続のパフォーマンスおよび安定性の強化、ならびに接続用 AS2 Vault URL に関する検証が実装されました。
Lifecycles for Case Assignment Objects
Vault Safety では、ケース割り当てオブジェクトのデフォルトのライフサイクルが導入されています。この機能強化は、23R3 で Vault Safety に追加されたケース割り当てチーム機能をサポートします。
Safety Rule Set Configuration Report: Function Families
このリリースでは、Vault Safetyは 関数ファミリをルール セット構成レポートにエクスポートすることをサポートします。これにより、23R2 でVault Safety に追加された Safety Rule Set Config Report 機能が強化されます。
Japan: PMDA ICSR Reporting Enhancements
このリリースでは、Vault Safety は PMDA への提出のサポートを強化します。この機能により、症例製品登録のスナップショット、ローカライズされたケース評価期限の計算、および期待値の計算が改善されます。さらに、このリリースでは、準拠した提出をサポートするために PMDA E2B(R3) データ マッピングが更新されます。
Aggregate Reports: Assessment Result (Override)
このリリースでは、重篤な副作用 (SAR) テーブルの DSUR and PBRER 間隔ライン リストと CIOMS II ライン リストに、評価結果の値ではなく、評価結果 (オーバーライド) の値が存在する場合はそれが表示されます。
Product Types Populated in Company Product Browser
Vault Safety 製品ブラウザに、各製品の製品タイプと製品登録が表示されるようになりました。
詳細
- ユーザヘルプ: 製品の自動コード化と閲覧: 製品閲覧について
External Product Substance Creation Improvements
WHODrug 辞書を使用して外部製品をコーディングする場合、Vault Safety によって自動的に作成される製品成分の数が増えるようになりました。作成できる製品成分の数の制限は、すべての製品物質名の合計文字数に基づいて 1,500 文字です。
詳細
- ユーザー ヘルプ: WHODrug 用語のコード化
Safety Feature Enablement Updates
今回のリリースでは、以下の機能で有効化の更新が適用されます:
Enablement Update: PMDA Local Reporting Details Generation and Submission Linking
今回のリリースにより、PMDA ローカルレポートの詳細情報の生成と提出のリンク付け機能が自動オンになりました。この機能は、当初、サポート付きで 23R3 に導入されました。この機能は自動オン仕様ですが、一部のコンポーネントでは追加の設定が必要です。
この機能の詳細については、以下の記事をご確認ください。
Safety & SafetyDocs
Redacted Document Tracking
今回のリリースにより、新しい標準選択リストの編集フィールド、および編集済みと未編集との関係が、Vault Safety に追加されました。これにより、ユーザは個人識別情報 (PII) データが編集されたドキュメントを識別し、未編集のソースドキュメントに関連付けることができます。編集済みのドキュメントの追跡により、データプライバシー規制への準拠が容易になります。
詳細
- ユーザヘルプ: Vault Safety の操作: ライブラリ
Create Inbox Item from Literature Article
今回のリリースにより、Vault SafetyDocs では文献症例処理がよりシンプルになり、安全性管理との統合が強化されます。Vault SafetyDocs で処理され、ICSR を含む文献記事は、Vault Safety インボックスに送信して症例として処理することができます。これにより、手動でデータを入力する必要がなくなり、ミスが起こる可能性が低下して、文献症例処理のための効率的なエンドツーエンドソリューションが提供されます。
注: この機能を使用するには、Vault Safety と Vault SafetyDocs の両方のライセンスが必要です。
詳細
- 有効化: 文献記事からのインボックスアイテムの作成を有効にする
- ユーザヘルプ:
Aggregate Reporting: Combine Tabulations and Authoring
今回のリリースにより、Vault Safety の集積レポートセクションレベルで集積レポートのラインリストと表を生成できるようになり、レポート作成プロセスが合理化されます。この機能を使用すると、Vault Safety の表生成機能と Vault SafetyDocs のレポート作成機能が統合され、お客様は、計画からデータ収集、作成、承認、提出追跡に至るエンドツーエンドの集積レポートプロセスを単一のシステムで管理できます。
注: ラインリストと表を生成するには、Vault Safety のライセンスを取得する必要があり、Aggregate Report を作成するには、Vault SafetyDocs のライセンスを取得する必要があります。
詳細
SafetyDocs
Multiple Logbook Entries per Major Version of PSMF Documents
今回のリリースにより、Vault SafetyDocs では、PSMF ドキュメントの主要バージョンごとに最大 10 個のログブックエントリを作成および表示できるようになりました。この機能強化以前は、Vault SafetyDocs では主要ドキュメントバージョンごとに 1 つのログブックエントリがサポートされ、表示されていました。この機能強化は、EMA 規制 (GVP モジュール II) に準拠しており、外部システムからのログブックレコードの移行をより適切にサポートします。
詳細
PSMF: Document Deletion
ユーザーが PSMF コンテンツドキュメントを削除すると、Vault SafetyDocs は対応する PSMF ログブックと関連するログブックエントリを自動的に削除できるようになりました。このリリース以前は、ドキュメントを削除すると、関連するログブックレコードで参照されていたためにエラーが発生し、ユーザーは最初にレコードを手動で削除する必要がありました。この機能により、Vault SafetyDocs は、関連する PSMF ログブックレコードを PSMF コンテンツドキュメントとともに削除することで、手動作業を排除して生産性を向上させます。
詳細
- 有効化: PSMF ドキュメントの削除を有効にする
- ユーザヘルプ: PSMF バインダーおよびログブックの管理: PSMF ログブック
Exclude Obsolete Documents from the PSMF PDF
Vault SafetyDocs では、マージされた PSMF PDF から、PSMF コンテンツまたは PSMF 生成ドキュメントのサブタイプを含む古いドキュメントが除外されるようになりました。古い PSMF ドキュメントは、ログブックおよびログブックエントリへの追跡可能性を維持するために PSMF バインダーに含まれることがよくあります。この機能を使用すると、ユーザは古いドキュメントを含めずに、関連するログブックおよびログブックエントリを PSMF PDF に含めることができます。マージされた PDF から古いドキュメントを除外すると、よりクリーンで準拠性の高い PSMF が提供されます。
詳細
Auto-Translation of Imported Literature Abstracts
このリリースで、Vault SafetyDocs は、データベースから一括でインポートされた英語以外の文献の抄録を自動翻訳できるようになりました。以前は、一括インポートされた抄録をユーザーが手動で送信して翻訳していましたが、このプロセスが自動化され、効率が向上しました。
この機能は、文献抄録の自動翻訳が有効になっている Vault で自動オンになります。
詳細
PVA: Document Distribution via Email
このリリースでは、Vault SafetyDocs はドキュメント配布 (集計レポート、PSMF、RMP、文献レビュー、シグナルレポートなど) の追跡を拡張し、電子メールでの配布も可能になりました。この機能は、PVA アクティビティまたは PVA アクションアイテムレコードからドキュメントをディスパッチする新しいアクションを追加します。このアクションが開始されると、受信者が関連ドキュメントの閲覧に使用できるリンクを含む Vault 追跡電子メールメッセージが生成および送信されます。この機能は、PVA 指令への準拠を実証するのに役立ちます。
詳細
- 有効化: メールを使用して PVA ドキュメント配布を有効にする
- ユーザヘルプ:
Document Distribution to Partners via Vault Task
Vault SafetyDocs では、Vault タスクを使用したパートナーへのドキュメント配布がサポートされるようになりました。このタイプの配布により、ドキュメントの正しいバージョンが確実に送信され、手動タスクが削減されて、トレーサビリティが強化され、PVA 指令への準拠が実証されます。
注: パートナーは、タスクが割り当てられている Vault にアクセスできる必要があります。
詳細
MedDRA Hierarchical Browser for Product-Event Combinations
このリリースでは、MedDRA ブラウザが拡張され、MedDRA 階層のどのレベルでも製品とイベントを組み合わせた MedDRA 用語のコーディングが許可されるようになりました。以前は、製品とイベントの組み合わせの MedDRA 階層用語ブラウザでは、すべての階層レベルでコーディングできるわけではありませんでした。MedDRA ブラウザには、システムが検索する MedDRA 階層レベルを管理者が指定できる新しい選択リストが追加され、MedDRA コーディングプロセスが合理化および改善され、シグナルを追跡する際の粒度が向上しました。
詳細
- ユーザヘルプ: シグナル管理: 製品とイベントの組み合わせの作成
Aggregate Report Task Creation Enhancements
集計レポートタスクを作成する際に、それらに添付されたドキュメントは、関連する集計レポートと集計レポートタスクの名前の一部を組み合わせた新しい命名規則に準拠するようになりました。このロジックにより、名前が最大文字数制限を超えたことによるドキュメント作成時のエラーが防止されます。
Aggregate Reporting: Section Role Assignment
Vault SafetyDocs ユーザは、集積レポートセクションを他のユーザに割り当ておよび再割り当てできるようになりました。システムでは、割り当てられたユーザがセクションレコードおよび関連するレポートドキュメントに確実にアクセスできます。この機能により、共同編集が容易になり、集積レポートのコントリビュータの生産性が向上します。
詳細
Update Label of Generate Aggregate Report Section Creation Dialog
今回のリリースにより、機能をより適切に表すために、集積レポートタスクの名前が集積レポートセクションに変更されました。集積レポートタスク生成ダイアログが、集積レポートセクション生成ダイアログに更新されました。その他の集積レポートタスクのラベルは設定可能であり、この用語に合わせてお客様が直接更新することができます。
Aggregate Reporting Feature Configuration
今回のリリースにより、Vault SafetyDocs では集積レポートの全設定を使用できるようになりました。これには、以前にリリースされた次の機能が含まれます。
詳細
- 有効化: 集積レポートの管理を有効にする
Safety Data Model Changes
24R1 データモデルの変更: Safety をご覧ください。
QualityOne
以下の Release Notes に加えて、QualityOne Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモを提供しています。
ドキュメント管理
External Collaboration: External Review & Approval of Documents
この機能を使用すると、既存の QMS External Collaboration 機能が Document Control に拡張され、ユーザはドキュメントのレビューと承認のタスクを外部の人に割り当てることができます。この機能は、組織と外部関係者 (サプライヤーなど) の間の共同編集プロセスを合理化するのに役立ち、電子メールや共有ドライブなどの効率の悪い共同編集メカニズムを置き換えることができます。
ドキュメントのレビューと承認のための External Collaboration プロセスは、次のように行われます。
- ユーザがドキュメント上で、レビューまたは承認のためにそのドキュメントを外部の人に送信するかどうかを指定します。yes の場合、ユーザは組織を選択し、その組織の承認済みの連絡先から外部コラボレーション者を選択できます。
- ユーザが関連するドキュメントワークフローを開始すると、Vault によってユーザアカウントが作成または有効化され、適切な権限がプロビジョニングされて、外部ユーザライセンスが要求されます。
- Vault では、情報とログインの詳細が記載された適切なウェルカムメールが外部コラボレーション者に送信されます。
- 外部コラボレーション者は、必要に応じてドキュメントに注釈を追加し、裁定して電子署名を付けることができます。
- 外部コラボレーション者が Vault 内のオープンタスクを完了し、ドキュメントが固定状態に達すると、外部コラボレーション者にさようならメールが送信され、ユーザアカウントは自動的に無効になります。外部ライセンスが再び有効になります。
この機能は、アグリーメントやその他のドキュメントのレビューおよび承認のためのアクセスを頻繁に必要としない外部関係者を対象としています。
External Collaboration 機能でドキュメントのレビューおよび承認を行うについて詳しくご覧ください。
QMS (QualityOne)
Supplier Periodic Review (SPR)
今回のリリースにより、QualityOne QMS では定期レビュー機能が拡張されて、サプライヤー定期レビュー (SPR) がサポートされ、年次製品品質照査 (APQR) プロセスの既存のサポートに追加されています。
サプライヤー定期レビュー (SPR) 機能を使用すると、ユーザはデータを取得し、サプライヤーの定期評価に関連するプロセスを完了できます。SPR プロセスの一環として、お客様はデータ編集タスクの割り当て、サプライヤー組織の属性 (リスクや監査日付など) の設定、レポートからのドキュメントの自動生成、収集され記録に保存されたデータに基づく最終 SPR レポートの生成を行うことができます。
QMS 定期レビュー機能により、ユーザは各定期レビュープロセスに合わせたカスタム機能を備えた共有機能を使用して、Vault で製品 (APQR) とサプライヤー (SPR) の定期評価を管理できるようになりました。
詳細については、定期レビューおよび SPR の実施をご覧ください。
HACCP Flow Diagram Enhancements
この機能では、HACCP フロー図に以下の改善が加えられました。
- 新しいグリッドビュー表示オプションにより、ユーザが処理手順と処理手順接続を位置合わせしやすくなりました。
- 図上に重要管理が表示されるようになりました。
- 図上の材料処理手順に原料を追加できるようになりました。
- プロセスステップに新しい Actions メニューが追加され、処理手順を編集、削除、および任意または必須としてマークする各種オプションが追加されました。
- 図上に処理手順接続ラベルが表示されるようになりました。
- 処理手順接続の入力ハンドルと出力ハンドルを選択できるようになりました。
詳しくは、HACCP Flow Diagram をご覧ください。
注: 現在、この機能を利用できるのは、アーリーアダプタープログラムに登録されているお客様のみです。詳細については、Veeva 担当者にお問い合わせください。
HACCP Flow Diagram: Grouping Process Steps
この機能を使用すると、ユーザは特定のビジネスまたは運用目標を達成する HACCP 計画内で処理手順のグループを作成できます。処理手順グループが作成されると、HACCP Flow Diagram に表示されます。
詳しくは、HACCP Flow Diagram をご覧ください。
注: 現在、この機能を利用できるのは、アーリーアダプタープログラムに登録されているお客様のみです。詳細については、Veeva 担当者にお問い合わせください。
COA Email Intake Enhancements
この機能により、COA ファイルを含む同じ電子メール通知に複数のユーザが応答することがなくなり、COA ファイルの電子メール取り込みが改善されます。これらの機能強化強化により、Vault では適切な数の COA インスペクションレコードが確実に作成されます。また、電子メール取り込みと Vault の相互作用を使用するときに COA ファイルが既に正常に受信されているかどうかがユーザに通知されます。
この機能は、分析証明書の自動電子メール取り込みが既に設定されている Vault で自動的にオンになります。
詳細については、メール取り込みによる COA ファイルのアップロードをご覧ください。
注: 現在、この機能を利用できるのは、アーリーアダプタープログラムに登録されているお客様のみです。詳細については、Veeva 担当者にお問い合わせください。
Preliminary COA Variant Setup Panel
この機能は、ドキュメントライブラリから COA バリエーションを直接設定するための統合ビューをユーザに提供する基盤となります。これにより、ユーザエクスペリエンスを合理化し、COA ファイルを取り込む準備に必要な時間を短縮することができます。完全なバリエーション設定エクスペリエンスは、複数のリリースにわたって提供されます。
詳細については、COA ファイル分析のバリエーション定義をご覧ください。
注: 現在、この機能を利用できるのは、アーリーアダプタープログラムに登録されているお客様のみです。詳細については、Veeva 担当者にお問い合わせください。
QMS (QualityOne) および HSE
Auditor Profile & Qualification Management
この機能により、ユーザはすべて Vault 内で、監査人に関する重要な統計情報を取得し、さまざまなタイプの監査で特定のロールを実行するために監査人に必要な資格を管理し、監査人の監査履歴と、監査ロールの資格取得に向けた進捗状況を追跡することができます。その結果、次のような利点が得られます。
- チームの監査ロールとして選択できるのは、権限および資格のある監査人のみです。
- 必要な資格取得に向けた監査人の進捗状況を追跡します。
- 監査人の資格が明確化し、組織内の資格のある監査人のプールに対する監視が強化されます。
- 資格を満たす監査人に、特殊なアクセス権とロールの資格が自動的に付与されます。
監査人のプロファイルと資格の管理について詳しくご覧ください。
QualityOne Audit Checklist Mobile Enhancements
この機能は、Android と iOS 両バージョンの QualityOne 監査チェックリストモバイル アプリケーションに、シームレスなエンタープライズ展開に不可欠な機能強化と、お客様からのフィードバックに基づいた重要な更新を提供します。
QualityOne 監査チェックリストモバイルの 24R1 リリースは、2024 年 4 月 30 日に暫定公開を予定しています。
Android Audit Checklist Enhancements
この機能により、監査チェックリストを実行する監査人にとってのユーザエクスペリエンスがさらに合理化され、データ入力のスピードアップが可能になるほか、監査チェックリストの特定種類の質問への回答に必要なクリック数を削減できます。
iOS Audit Checklist Enhancements
ユーザはこの機能を使用して、iOS 版の QualityOne 監査チェックリスト モバイルアプリケーションから直接、監査チェックリストセクションを作成できます。これによりモバイルアプリケーションを終了したり Vault にアクセスすることが不要となり、監査人はその場で監査対象コンテンツをより柔軟に構成できます。
この機能により、SSO SAML プロバイダーを使用するお客様は、ユーザによる単一の認証情報を使った QualityOne 監査チェックリストモバイルへのアクセスを許可できるようになるため、より安全な認証方法が実現します。
さらにこの機能により、監査チェックリストを実行する監査人用の監査チェックリストが機能強化され、データ入力のスピードアップが可能になるほか、監査チェックリストの特定種類の質問への回答に必要なクリック数を削減できます。
QualityOne Data Model Changes
24R1 データモデルの変更: QualityOne をご覧ください。
RegulatoryOne
すべての Platform 機能を含む RegulatoryOne の 24R1 Release は、2024 年 4 月 30 日に暫定公開を予定しています。
以下の Release Notes に加えて、RegulatoryOne Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモを提供しています。
コンプライアンス管理
Formulation Composition Viewer: Bulk Create Qualitative Assessments
この機能により、ユーザは Formulation Composition Viewer から、1 つの配合剤について最大 15 個の定性的評価を同時に作成できるため、定性的評価をより迅速かつ少ないクリック数で作成できるようになります。これにより、組織の配合剤の承認時間と市場投入までの時間が短縮されます。
詳しくは、Formulation Composition Viewer をご覧ください。
規制ドキュメント、登録およびドシエの管理
Track Formulation Concentrations
この機能によって Formulation Concentration オブジェクトが追加され、ユーザは配合剤の物質濃度を追跡できるようになります。
レジストレーションおよびドシエ管理
Requirement Hierarchy Viewer: Run Record Actions
この機能により、ユーザは、Requirement Hierarchy Viewer 内で Requirement レコードアクションを実行できるようになり、各レコードの詳細ページに移動することなく、ビューア内で直接より多くのタスクを実行できるようになりました。
詳細については、Requirement Hierarchy Viewer をご覧ください。
Requirement Hierarchy Viewer: Add Matched Documents
この機能により、ユーザは EDL Items レコードに移動することなく、Requirement Hierarchy Viewer で直接、一致したドキュメントをドシエ内の EDL 項目に追加できるようになります。
詳細については、Requirement Hierarchy Viewer をご覧ください。
Requirement Hierarchy Viewer: Upload Documents
この機能により、ユーザは EDL Items レコードに移動することなく、Requirement Hierarchy Viewer で直接、ドシエ内の EDL 項目についてドキュメントをアップロードしたり、テンプレートからドキュメントを作成したりできるようになります。
詳細については、Requirement Hierarchy Viewer をご覧ください。
Requirement Hierarchy Viewer: Lock & Unlock Matched Documents
この機能により、ユーザは EDL Items レコードに移動することなく、Requirement Hierarchy Viewer で直接、ドシエ内の EDL 項目について、一致したドキュメントをドキュメントバージョンにロックおよびロック解除できるようになります。
詳細については、Requirement Hierarchy Viewer をご覧ください。
Requirement Hierarchy Viewer: Remove & Exclude Matched Documents
この機能により、ユーザは EDL Items レコードに移動することなく、登録階層ビューアで直接、一致したドキュメントをドシエ内の EDL 項目から削除または除外できるようになります。
詳細については、Requirement Hierarchy Viewer をご覧ください。
Requirement Hierarchy Viewer: Display EDL Item Data
この機能により、ユーザは、EDL 項目データ (予想されるドキュメントの数や定常状態のドキュメントの数など) をRequirement Hierarchy Viewer 内で表示できるようになりました。これにより、ユーザはさまざまな EDL Item レコードに移動することなく、より多くのデータをビューアで直接表示できるため、時間を節約できるようになりました。
Automatic Document Creation from Template for Dossiers
この機能を使用すると、ユーザは、ドシエ内の該当するすべての EDL 項目について、指定されたテンプレートからドキュメントを自動的に作成できるようになります。
詳細については、テンプレートからドシエドキュメントを自動作成するをご覧ください。
Generate Requirements Enhancement: Default Field Values from Requirement Library
この機能により、管理者は Requirement Library レコードから、Requirement、EDL、および EDL Item の各オブジェクトで定義されたドシエ要件の詳細への、オブジェクトとフィールドのマッピングを定義し、オブジェクトマッピングを Generate Requirements アクションにリンクできるようになります。このアクションが実行されると、Vault では、オブジェクトマッピングにマッピングされたデフォルトのフィールド値が、生成されたレコードに自動的に入力されるため、ユーザはこれらのフィールド値を入力する手間が省けます。
詳細については、要件生成アクションの設定をご覧ください。
RegulatoryOne Data Model Changes
24R1 データモデルの変更: RegulatoryOne をご覧ください。
Veeva Claims
すべての Platform 機能を含む Veeva Claims の 24R1 Release は、2024 年 4 月 30 日に暫定公開を予定しています。
以下の Release Notes に加えて、Claims Veeva Connect コミュニティでは、General Release に関するお知らせ、リリースの注目機能、主要な機能のデモを提供しています。
Veeva Claims
Preview Claims & Local Adaptations Before Generating Selective Records
この機能強化により、ユーザが新しい確認ページで Selectively Create Claims アクションと Create Selective Local Adaptation アクションの選択内容を確認できるようになり、Claims と Local Adaptations を選択的に生成するプロセスでエラーが発生しにくくなります。
以前は、これらのアクションにより、ユーザは Claims と Local Adaptations を選択して作成できましたが、新しいレコードを生成する前に、統合ビューで選択内容を確認することはできませんでした。このプロセスにより、ユーザは選択内容を確認および確定する前に Continue をクリックして、誤ってレコードを生成する可能性がありました。この機能を使用すると、混乱が少なくなり、不要なレコードが生成されなくなります。
詳細については、選択的クレームの作成および選択的ローカル適応の作成をご覧ください。
Dynamic Deep Copy Project Enhancement: Record Selection
この機能により、Clone Project アクションが強化され、ユーザは各関連レコードセクションからレコードを選択して、新しくコピーされたプロジェクトにコピーできるようになります。さらに、ユーザは新しい Project レコードを作成する前に、確認ページで選択内容を確認できます。
プロジェクトレコードをコピーして新しいプロジェクトを開始することは、Veeva Claims ユーザの間で一般的な使用例です。以前は、Clone Project を使用すると、ユーザは新しいプロジェクトを作成し、指定されたオブジェクトグループのすべてのレコードを自動的にコピーできました。ただし、これにより、それらのグループ内の不要なレコードのコピーも含まれる場合がありました。この機能により、ユーザは不要なレコードを作成することなく、プロジェクトのコピープロセスを高速化できるようになります。
詳細については、プロジェクトのコピー作成をご覧ください。
Comment Thread Viewer Enhancement: Timestamp & Sort
この機能により、2 つの新しい改善が導入され、コメントスレッドビューアの使用に関する全体的なエクスペリエンスが向上しました。まず、各コメントに作成日時が表示され、会話に貴重なコンテキストが提供されるようになりました。次に、元のコメント作成日または最新のコメントと返信の作成日に基づいて、コメントと返信を並べ替えることができ、よりパーソナライズされた効率的なエクスペリエンスを提供できるようになりました。
詳細に関しては、using comments をご覧ください。
Remove Statement & Associated Records from Project: UI Enhancements
この機能では、Remove Statement and Associated Records from Project アクションの全体的なユーザエクスペリエンスを向上させるためのいくつかの機能強化が導入されています。ユーザは、各関連レコードセクションのすべてのページですべてのレコードを選択し、各セクションヘッダーで選択したレコードの数を確認できるようになりました。
詳細に関しては、プロジェクトからステートメントと関連レコードを削除をご覧ください。
Element Validation for Pack Copy
この機能では、ユーザがローカル要素をグローバル パックコピーに追加すること (およびその逆) を制限する検証チェックが導入されました。さらに、管理者がこれらの新しい検証に違反する可能性のあるパックコピー を特定して、それらのレコードを簡単に修正できるようにするための設定可能なジョブを提供します。この機能強化により、組織はデータの整合性を保護し、不正確な情報の意図せぬ作成を防止し、パックコピーの開発時の正確性と信頼性を向上させることができます。
詳細については、パックコピー管理、および一致しないパネル要素レコードのレビューをご覧ください。
Unify Admin Experience for Selective Project Automations
このリリースの時点で、Selectively Create Claims via User Input および Create Local Adaptations via User Input アクションは廃止されサポートされなくなったため、Vault での設定はお勧めしません。これらのアクションは、Selective Project Automation Enhancements: Actions Run as System 機能で、23R2 で導入された Create Claims Run As System および Create Local Adaptations Run As System アクションに置き換えられます。新しいアクションを使用すると、ユーザーは、管理者が特定のフィールドレベルの権限を割り当てることなくレコードを生成できます。
詳細については、選択的クレームの作成および選択的ローカル適応の作成の設定をご覧ください。
Veeva Claims Data Model Changes
詳しくは、24R1 データモデルの変更: Veeva Claims をご覧ください。